Full Answer
What are elements in Group 7 halogens?
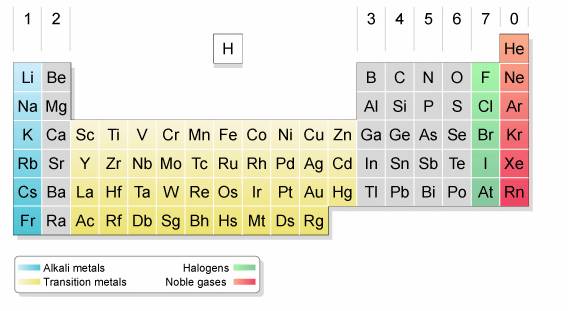
Group 7 (Halogens) The halogens are found in group 7 of the Periodic Table and are one electron away from a full outer shell of electrons. The halogens include the elements chlorine, bromine and iodine which all behave in similar ways due to similarities in their electron configurations.
Why are Group 7 elements called halogens?
Why is Group 7 called halogens? Group 7 elements form salts when they react with metals. The term 'halogen' means 'salt former', which is why Group 7 elements are called halogens. In general the halogens comprise the most reactive group of non-metals. The halogens are so reactive that they cannot exist free in nature.
What number group is halogens?
The halogens are the elements that form group 17 of the periodic table. They are reactive nonmetals and include fluorine, chlorine, bromine, and iodine. Halogens are highly reactive non-metals. These elements greatly resemble in property with each other.
What are the characteristics of Group 7 elements?
- They are inert gases located on the right of the periodic table.
- They have a full-set of valence electrons, so they're stable.
- They are colorless, odorless and tasteless.
- They have low melting and low boiling points.
- They can be found in small amounts in the Earth's crust and the Earth's atmosphere.
Are halogens Group 7 or Group 17?
halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).
Is Group 17 called halogens?
The group 17 elements include fluorine(F), chlorine(Cl), bromine(Br), iodine(I) and astatine(At) from the top to the bottom. They are called “halogens” because they give salts when they react with metals.
Is Group 7 the halogens?
The halogens are found in group 7 of the Periodic Table and are one electron away from a full outer shell of electrons.
Is group 17 and 7A the same?
Fluorine is a halogen, which is a group of non-metals located on the right side of the periodic table that includes fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Most halogens are electron-hungry, like fluorine. Halogens can also be referred to as group 7A, group 17, or group VIIA elements.
Why is Group 7A called halogens?
The name "halogen" means "salt former", derived from the Greek words halo- ("salt") and -gen ("formation"). The Group 7A elements have seven valence electrons in their highest-energy orbitals (ns2np5).
What are group 17 elements known as?
Group 17 elements are collectively called halogens (In Greek: halo means salt and genes mean producing, so collectively salt producing) and it consists of fluorine, chlorine, bromine, iodine, and astatine. The similarity to this extent is not found in other groups of the periodic table.
What is group 7 called?
The Group 7 elements are called the halogens. They are placed in the vertical column, second from the right, in the periodic table . Chlorine, bromine and iodine are the three common Group 7 elements. Group 7 elements form salts when they react with metals. The term 'halogen' means 'salt former'.
Why are the halogens in Group 17?
Why are the elements in Group 17 categorised together? Group 17 is the halogens, they have 7 electrons in their outermost shell. They are all non-metals, and form anions with a -1 charge. Reactivity decreases down the group, with fluorine being the most reactive.
What is the trend of group 7 halogens?
The non-metal elements in Group 7 - known as the halogens - get less reactive as you go down the group. This is the opposite trend to that seen in the alkali metals in Group 1 of the periodic table .
What does 7A mean on the periodic table?
Families: 1A - alkali metals. 2A - alkaline earth metals. 7A - halogens.
Why are group 7A 17 elements found in many compounds but not group 8a 18?
Each uncharged atom of these elements requires 1 more valence electron to achieve a stable noble gas electron configuration. The lack of an incomplete valence shell means that main group 7A elements will readily participate in chemical reactions that form various compound products.
Which of the following is not involved in Group 17?
Argon is a gas from group 18 called noble gases which are highly unreactive. The Halogens are the elements belonging to Group 17 in the periodic table....Detailed Solution.Element NameSymbolAtomic NumberFluorineF9ChlorineCl17Bromine Br35IodineI531 more row
Why is it called halogens?
The name halogens are from Greek halo (sea salt) and gens (producing formation) and thus means 'sea salt former'. Group 17 contains fluorine, chlorine, bromine, iodine which form salts. Hence they are called halogens. Was this answer helpful?
Why are the halogens in group 17?
Why are the elements in Group 17 categorised together? Group 17 is the halogens, they have 7 electrons in their outermost shell. They are all non-metals, and form anions with a -1 charge. Reactivity decreases down the group, with fluorine being the most reactive.
What are halogens and why are they so called?
The group 17 elements include fluorine(F), chlorine (Cl), bromine(Br), iodine(I) and astatine(At) from the top to the bottom. They are called “halogens” because they give salts when they react with metals.
What is the term halogen mean?
halogen. / (ˈhæləˌdʒɛn) / noun. any of the chemical elements fluorine, chlorine, bromine, iodine, and astatine. They are all monovalent and readily form negative ions.
What are the elements in the group 7?
The Group 7 elements are called the halogens. They are placed in the vertical column, second from the right, in the periodic table. Chlorine, bromine and iodine are the three common Group 7 elements. Group 7 elements form salts when they react with metals. The term ‘halogen’ means 'salt former'.
What is the purple vapour that forms when it is warmed?
Iodine forms a purple vapour when it is warmed.
Where are halogens found in the periodic table?
The halogen elements are located in group VIIA of the periodic table, which is the second-to-last column of the chart. This is a list of elements that belong to the halogen group and the properties that they share in common:
How many electrons are in a halogen atom?
They are highly reactive nonmetals. Atoms of belonging to the halogen group have 7 electrons in their outermost (valence) shell.
What are halogens used for?
Their high reactivity also makes these elements important components of some types of bleach. Halogens are used in incandescent lamps to make them glow at a higher temperature and with a white color. The halogen elements are important drug components, as they aid drug penetration into tissues.
What color is halogen?
The halogens are colorful, even as gases. Fluorine is the palest element, but even as a gas it has a distinct yellow color.
Why do halogens need more electrons?
These atoms need one more electron in order to have a stable octet. Halogens are highly electronegative, with high electron affinities. The melting and boiling points of the halogens increase as you increase atomic number (as you move down the periodic table).
What is the element Br?
Bromine is element 35 with symbol Br. It is a liquid at room temperature and pressure.
Is chlorine a halogen?
Fluorine, chlorine, bromine, iodine, and astatine definitely are halogens. Element 117, which has the placeholder name of ununseptium, might have some properties in common with the other elements. Even though it is in the same column or group of the periodic table with the other halogens, most scientists believe element 117 will behave more like ...
What is the name of the group of halogens in the periodic table?
This page introduces the Halogens in Group 7 of the Periodic Table. Group 7 is also known by its more modern name of Group 17.
Which halogen displaces the less reactive one from one of its salts?
The more reactive halogen displaces the less reactive one from one of its salts.
How is hydrogen chloride gas produced?
Hydrogen chloride gas is produced explosively when the mixture is exposed to light. In the lab, you have to be very careful if you mix gas jars of hydrogen and chlorine to keep them well away from sunlight.
How many protons does chloride have?
In a chloride ion, the outer electrons feel a pull of 17 protons in the nucleus, partially shielded by 10 electrons - a net pull of 7+. In a bromide ion, the outer electrons feel a pull of 35 protons in the nucleus, partially shielded by 28 electrons - a net pull of 7+.
Is fluorine reactive?
Fluorine is extremely reactive and I have never even seen it, let alone used it. There is an interesting bit of video from the University of Nottingham Periodic Table series which shows that I am not alone. You are unlikely to need this for exam purposes. I include it simply because it is interesting.
Is lithium chloride a white solid?
The video skips over the fact that lithium chloride is a white solid. This is masked by the reaction between lithium and the glass producing black solids.
What Are the General Properties of Halogens?
Just like other groups in the periodic table of elements, the six elements in column 17 have similar chemical and physical properties . This is mainly because of their atomic structures and electron configuration.
How many valence electrons are in a halogen?
Halogens have seven valence electrons in their outermost energy level, which can be denoted by ns 2 np 5. Additionally, they all have an ‘ s’ (spherical shape) and a ‘ p’ (dumbbell shape) orbital in the outermost shell. All halogens are highly electronegative due to the high effective nuclear charge of the elements.
What is the name of the group of elements that are highly reactive?
The halogens are the six nonmetallic, highly reactive elements under Group VIIa (column 17) of the periodic table. They’re very strong oxidising agents, which means they take electrons from other substances. They’re also very reactive and usually form salts with Group 1a, i.e. the alkali metals. The name halogen literally means salt-producing. Halogens in elemental form do not exist in nature because they’re so highly reactive.
What are halogens used for?
Halogen compounds have a wide range of applications, including water sanitation, paper production, plastic polymer synthesis, and petroleum refining.
What are some examples of interhalogen compounds?
Some examples include bromine fluoride (BrF), iodine monochloride (ICl), and chlorine monofluoride (ClF).
Why are halogens so reactive?
All halogens are highly reactive because of their electron configuration. They’re also corrosive and poisonous, making them ideal disinfectants…but also potential chemical weapons.
How many elements are in Group VIIA?
Group VIIa (17) has five naturally-occurring elements and one synthetic element. While the halogens have similar chemical and physical properties, the synthetic member of the group, tennessine, has some properties that are dissimilar to the halogens. For example, it’s solid and semi metallic at room temperature.