
- The density of water at 40C is maximum and it is in liquid state at this temperature. ...
- Icebergs float on water but 90% volume of an iceberg remains underwater. ...
- The “competition” between hydrogen bond formation and thermal motion of water molecules is the reason behind the maximum density of water at 40C.
What temperature does water reach its densest point?
The temperature at which water is most dense is 3.98 degrees Celsius (39.164 degrees Fahrenheit, 277.13 Kelvin). At this temperature water’s density is about 999.9720 kilograms per cubic meter. At room temperature (20 degrees Celsius) water has a density of 998.2071 kilograms per cubic meter.
What temperature is the highest density of water?
Water is the densest at 39 degrees Fahrenheit (or 3.98 degrees Celsius). At this temperature, ice floats on water. Water's density decreases as its temperature increases.
Is hot water less dense than room temperature?
When water is cooled, the water molecules move slower and get closer together. This makes cold water more dense than room temperature water. Since cold water is more dense, it sinks in the room temperature water. When water is heated, the water molecules move faster and spread out more. This makes hot water less dense than room temperature water.
How does temperature affect the density of water?
Temperature plays a major role in affecting the density of water. As the temperature increases the molecules gain energy and move randomly and rapidly so there will be a difference of mass per unit volume ratio I.e density. We can say that temperature is inversely proportional to density! Hope you understood.
Why is water so dense?
As water cools it becomes more dense. At 39°F (or 3.98°C to be exact) water is the most dense. This is because the molecules are closest together at this temperature. The closer the molecules, the heavier. The farther apart the molecules, the lighter.
Why is water considered a solid?
In other words, the molecules are farther part even though the water is solidifying into ice. This seems counter-intuitive because as most elements solidify, they become heavier. Water is unique in this sense, because as it becomes a solid – it becomes lighter. Think about a cup of ice water.
Why isn't ice more dense?
So then if water is heavier the colder it gets, why isn’t ice more dense? This is because the molecular structure and hydrogen bonding of the water takes up more space when it is a frozen solid rather than when it is a warmer liquid.
Why does ice float on water?
The ice floats on the water because ice is less dense than water. It’s also why you’d rather carry a cooler of ice than a cooler or water. And why lakes freeze at the top rather than from the bottom.
Water Density
If you're still in school, you've probably heard this statement in your science class: " Density is the mass per unit volume of a substance". On Earth, you can assume mass is the same as weight, if that makes it easier.
Water's density varies with temperature
Growing up with an older brother was difficult, especially when he had his friends over, for their favorite activity was thinking of ways to antagonize me. I was able to use water density once to at least play a trick on them, though. One hot summer day they climbed the huge hill next to our house to dig a hole to hide their bottle-cap collection.
Ice is less dense than water
If you look at this picture you can see that some of the iceberg is below the water level. This is not a surprise, but actually almost all of the volume of an iceberg is below the water line, not above it. This is due to ice's density being less than liquid water's density. Upon freezing, the density of ice decreases by about 9 percent.
We said ice floats on water, but what about "heavy ice"?
We already said ice floats on water because it is less dense, but ice of a special kind can be denser than normal water. "Heavy ice" is 10.6 percent denser than normal water because the ice is made from "heavy water".
Water Density
If you're still in school, you've probably heard this statement in your science class: " Density is the mass per unit volume of a substance". On Earth, you can assume mass is the same as weight, if that makes it easier.
Water's density varies with temperature
Growing up with an older brother was difficult, especially when he had his friends over, for their favorite activity was thinking of ways to antagonize me. I was able to use water density once to at least play a trick on them, though. One hot summer day they climbed the huge hill next to our house to dig a hole to hide their bottle-cap collection.
Ice is less dense than water
If you look at this picture you can see that some of the iceberg is below the water level. This is not a surprise, but actually almost all of the volume of an iceberg is below the water line, not above it. This is due to ice's density being less than liquid water's density. Upon freezing, the density of ice decreases by about 9 percent.
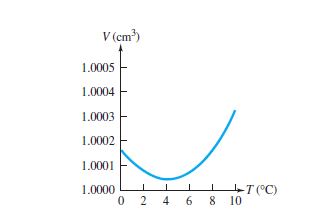
Why does the maximum density of water occur at 4 °C?
1 Answer. The maximum density of water occurs at 4 °C because, at this temperature two opposing effects are in balance.
What happens to water molecules as the temperature of warm water decreases?
As the temperature of warm water decreases, the water molecules slow down and the density increases. At 4 °C, the clusters start forming. The molecules are still slowing down and coming closer together, but the formation of clusters makes the molecules be further apart.
How do clusters affect the density of a liquid?
The clusters still have empty spaces, so they decrease the density of the liquid. The molecules of the water are closer together, and this increases the density of the liquid. Now, let's cool the water. As the temperature of warm water decreases, the water molecules slow down and the density increases. At 4 °C, the clusters start forming.
Which effect is bigger, cluster formation or density?
Cluster formation is the bigger effect, so the density starts to decrease.
What happens when ice melts to liquid water?
When the ice melts to liquid water, the structure collapses and the density of the liquid increases. At temperatures well above freezing, the molecules move faster and get further apart. The density decreases as temperature increases. At temperatures near 0 °C, the water still contains many ice-like clusters.
What Is the Density of Water?
Density is the mass per unit volume of a substance. The density of water is most given as 1 g/cm3, but below is the density of water with different units.
What is the boiling point of water?
Below is a chart that shows the density of water (in grams/cm 3) at different temperatures, ranging from below water's freezing point (-22°F/-30°C) to its boiling point (212°F/100°C).
How to find the density of a liquid?
Calculating the Density of a Liquid With a Hydrometer. If you're trying to calculate the density of a liquid, you also can do so by using an instrument known as a hydrometer. A hydrometer looks like a thermometer with a large bulb at one end to make it float.
How to find the density of a substance?
You can find the density of any substance by dividing its mass by its volume. The formula for density is: ρ=m/v, with density represented by the symbol ρ (pronounced "rho").
How much density is 1g/1cm3?
So, 1g/1cm 3 = 1 g/cm 3 , giving water its easy-to-remember density. However, water's exact density depends on both the air pressure and the temperature of the area. These variations in density are very slight though, so unless you need to know very exact calculations or the experiment takes place in an area with an extreme temperature/pressure, you can continue to use 1 g/cm 3 for water density. You can look at the chart in the next section to see how water's density changes with temperature.
What is the density of aluminum?
So the density of aluminum is 2.7g/cm 3, and this is true for any piece of (pure and solid) aluminum, no matter what its size is.
How many grams does water weigh?
Density is mass divided by volume (ρ=m/v), and water was used as the basis for establishing the metric unit of mass, which means a cubic centimeter (1cm 3) of water weighs one gram (1g).
