
How can we separate azeotropic mixture by fractional distillation?
We cannot separate the compounds of azeotropic mixture by fractional distillation. Azeotrope is a constant boiling mixture. When boiled, the composition of vapour is same as that of unboiled mixture.
What happens when distillation cannot alter a fraction of a liquid?
When distillation cannot alter a fraction of a liquid, it results in azeotropes. An azeotropic compound is a mixture of two substances which distil at the same temperature. An azeotrope is a mixture of compounds with the same composition in the vapour as in the liquid.
Do azeotrope mixtures have a higher or lower boiling point?
These mixtures can either have a lower boiling point or a higher boiling point of the components. An azeotrope is a mixture that, at a given pressure (the azeotropic pressure), boils at a constant temperature (the azeotropic temperature), and has the same composition (azeotropic composition) in the equilibrium vapour and liquid phases.
How do you separate azeotropes?
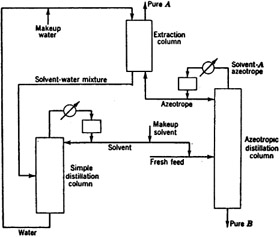
We can separate the azeotropes using extractive distillation, azeotropic distillation, or pressure swing distillation. What is a binary azeotrope?
Can we separate azeotropes by fractional distillation?
We cannot separate the compounds of azeotropic mixture by fractional distillation. Azeotrope is a constant boiling mixture. When boiled, the composition of vapour is same as that of unboiled mixture.
Can you distillation an azeotropes?
Azeotropic distillation (AD) is a process to break azeotrope where another volatile component, called the entrainer, the solvent, or the mass separating agent (MSA), is added to form a new lower-boiling azeotrope that is heterogeneous.
What can be separated by fractional distillation?
Fractional distillation is a method for separating a liquid from a mixture of two or more liquids. For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points.
Can you purify an azeotrope using distillation?
Breaking an azeotrope For low boiling azeotropes, the volatile component cannot be fully purified by distillation. To obtain the pure material one must "break the azeotrope", which involves a separation method that does not rely on distillation. A common approach involves the use of molecular sieves.
How do you separate an azeotrope?
Azeotropes can be separated by distillation (azeotropic distillation, extractive distillation, pressure swing distillation). In the case of azeotropic distillation a solvent is added which forms a lower boiling azeotrope (e.g. separation of aliphatics-aromatics using acetone.
Is it possible to separate the components of an azeotropic mixture by simple distillation explain?
Solution : No, we cannot separate the components of an azeotropic mixture by distillation because at a particular composition. Both the components boil at the same temperature.
Which of the following Cannot be separated by fractional distillation?
Which of the following cannot be separated from air by the process of fractional distillation ? Explanation: Hydrogen cannot be separated from the mixture of air as it is not present in the free state.
Which of the following mixtures Cannot be separated by fractional distillation?
Therefore, we can say that ,Benzene and toluene are the mixtures which cannot be separated by fractional distillation.
Which of the following is not separated by distillation?
Which of the following is not separated through distillation process? Explanation: Milk and water are not separated through the distillation process. All the other options can be separated through a distillation process.
Is azeotrope mixture can be separated?
An azeotropic mixture cannot be separated into its components by fractional distillation because, when an azeotrope is boiled, the vapor has the same proportions of constituents as the unboiled mixture.
Why Cannot distillation be used to separate azeotropes?
The reason why azeotropes cannot be separated by simple distillation is because when azeotropes are boiled, the vapours have the same proportions of the constituents as the unboiled mixtures.
How does azeotrope affect distillation?
An azeotrope (/əˈziːəˌtroʊp/) or a constant heating point mixture is a mixture of two or more liquids whose proportions cannot be altered or changed by simple distillation. This happens when an azeotrope is boiled, the vapour has the same proportions of constituents as the unboiled mixture.
Why is it impossible to separate an azeotropic mixture?
An azeotropic mixture cannot be separated into its components by fractional distillation because, when an azeotrope is boiled, the vapor has the same proportions of constituents as the unboiled mixture.
Why can distillation be used to separate azeotropes?
An azeotrope is a mixture that exhibits the same concentration in the vapor phase and the liquid phase. This is in contrast to ideal solutions with one component typically more volatile than the other; this is how we use distillation to separate materials.
What is meant by azeotropic distillation?
Definition: A method of separating mixtures based on differences in their volatilities in a boiling liquid mixture.
Do azeotropes obey Raoult's Law?
Solution : Second part: Azeotropic mixture does not obey Raoult's law.
What is azeotropic distillation?
Azeotropic distillation is the addition of a third component to generate a new, lower-boiling azeotrope.
What is an azeotrope?
An azeotrope is a constant-boiling mixture in which the composition of the vapour is the same as that of the liquid.
How to regenerate sieves?
The sieves can be regenerated by heating in a vacuum oven.
How to alter azeotrope?
Alter the azeotrope by changing the ambient pressure. Add a third component to remove one component a (e.g., ethanol, water, benzene), leaving the other. Use another mechanism for separation (membrane permeation).
Can you isolate azeotropeic mixtures?
Of course, the problem is that you really have no choice in determining which of the two components you can isolate, as that depends entirely on the starting composition of the mixture. In practice, you can often separate azeotropeic mixtures by modifying the distillation process in various ways.
Is a distillate richer than a residue?
This being understood, we at least separate one component from the mixture as the residue (for positive azeotropes) and the distillate (for negative azeotropes) is richer in one component than the other since the remaining part approaches the azeotropic concentration thereby increasing the concentration of one of the components depending on which side of the azeotropic "point" are we on the graph of boiling temperature and composition.
