Is energy endergonic or exergonic?
The most known definitions of exergonic and endergonic reactions is exergonic refers to a reaction that gives off energy, while endergonic reactions take in energy. Both reactions require for an input of activation energy and use enzymes to lower the activation energy need to complete the reaction.
What are the examples of exergonic reaction?
Examples of exergonic reactions. Combustion. Metal oxidations. Catabolic reactions of the body. Others. References. A exergonic reaction It is one that occurs spontaneously and that, in general, is accompanied by a release of energy, either in the form of heat, light or sound. When heat is released, it is said that we are facing an exothermic ...
What are some examples of endothermic and exothermic reactions?
- Melting ice cubes
- Melting solid salts
- Evaporating liquid water
- Converting frost to water vapor (melting, boiling, and evaporation, in general, are endothermic processes
- Making an anhydrous salt from a hydrate
- Forming a cation from an atom in the gas phase
- Splitting a gas molecule
- Separating ion pairs
- Cooking an egg
- Baking bread
Is an exothermic reaction endothermic or exothermic?
When a chemical reaction combines two or more things and makes a chemical bond, energy is released, so it is an exothermic reaction. These reactions usually feel hot because heat is given off. If a reaction breaks one or more bonds, energy is needed, or consumed, so it is an endothermic reaction.

Is an exothermic reaction exergonic?
Exergonic reactions occur spontaneously (no outside energy is required to start them). Examples of exergonic reactions include exothermic reactions, such as mixing sodium and chlorine to make table salt, combustion, and chemiluminescence (light is the energy that is released).
Whats the difference between exergonic and exothermic?
“Exothermic” literally means “outside heating” while “exergonic” literally means “outside work.” Some examples of exothermic reactions are: combustion of fuels such as wood, coal, oil, and petroleum, condensation of rain from water vapor, and mixing of alkalis and acids.
Is exergonic endothermic?
Exergonic and exothermic? The terms seem to be very similar, but not identical; is there a difference? Endergonic and exergonic refer to free energy changes (delta G). Endothermic and exothermic refer to the changes in internal energy of molecules, measured as heat given off or taken up, delta H.
Is endergonic the same as endothermic?
Exo/Endothermic represents the relative change in heat/enthalpy in a system, whereas Exer/Endergonic refers to the relative change in the free energy of a system.
What is meaning of exergonic?
exergonic in British English (ˌɛksəˈɡɒnɪk ) adjective. (of a biochemical reaction) producing energy and therefore occurring spontaneously.
Can an exothermic reaction be endergonic?
No. Exergonic reactions may be both exothermic or endothermic. Endergonic reactions are endothermic only. Endergonic and exergonic relate to changes in free energy (delta G), while endothermic and exothermic are related to changes in enthalpy (delta H).
What is endergonic vs exergonic?
Endergonic and exergonic reactions Exergonic reactions are also called spontaneous reactions, because they can occur without the addition of energy. Reactions with a positive ∆G (∆G > 0), on the other hand, require an input of energy and are called endergonic reactions.
What is the difference between exergonic and endergonic reaction?
Exergonic and endergonic reactions result in changes in Gibbs free energy. In exergonic reaction the free energy of the products is lower than that of the reactants; meanwhile in endergonic the free energy of the products is higher than that of the reactants.
Is exergonic positive or negative?
Exergonic is a type of reaction that has a negative Gibbs free energy. This indicates that an exergonic reaction is a spontaneous reaction since spontaneous reactions have a negative value for Gibbs energy when calculated using the thermodynamic relationship given below.
How can an endothermic reaction be exergonic?
For a exergonic reaction constraint is (from Gibbs-Helmholtz eqn): ΔG<0⇒ΔH−TΔS<0⇒ΔH
Definition. An endergonic reaction is one that requires free energy to proceed. An example of an endergonic reaction of biological interest is photosynthesis. Photosynthetic organisms conduct this reaction by using solar photons to drive the reduction of carbon dioxide to glucose and the oxidation of water to oxygen.
Exergonic reactions release energy; endergonic reactions require energy to proceed.
In the exergonic reaction, the reactants are at a higher free energy level than the products (reaction goes energetically downhill). In the endergonic reaction reaction, the reactants are at a lower free energy level than the products (reaction goes energetically uphill).
exergonic: What's the difference? In biochemistry, an endergonic chemical reaction is one that requires the input of energy (end- is a variant of endo- that means “inside” or “within”). An exergonic reaction is one that releases energy (ex- means “out”).
Exergonic reactions release energy; endergonic reactions require energy to proceed.
Photosynthesis and cellular respiration provide examples of how organisms store energy and release energy in biochemical systems. Photosynthesis is a type of endergonic reaction where energy is stored, and cellular respiration is a type of exergonic reaction where energy is released.
As opposed to exergonic reactions, which deal with the change in free energy of any chemical reaction known as Gibbs free energy, the former deal with the enthalpy changes in any chemical process evaluated in terms of heat in a closed system. Energy is released in both processes, however the sort of energy released varies.
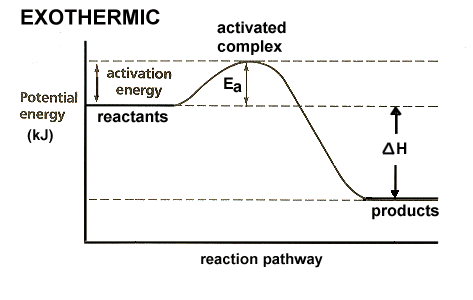
When two or more reactants rearrange their molecules in an exothermic reaction, energy is released in the form of heat or even light (a change in enthalpy H is also negative) as a result of the formation and breaking of chemical bonds. Joules are used as a unit of measurement here (the unit of heat).
An Exergonic reaction releases energy into the surrounding environment by rearranging the molecules of two or more reactants and creating and breaking chemical connections. This energy may then be employed to do work. It is also measured in joules since the quantity of energy consumed to do the job is the same as the amount of work done.
Exergonic processes are mostly bioenergetic, while exothermic reactions are primarily thermodynamic.
When it comes to their Gibbs free energy change (G and H both are negative), both exothermic and exergonic processes have enough energy to pass the activation energy barrier. However, there is a tiny variation in the amount of power they release.
In High School I learned that an exothermic reactions releases energy, while an endothermic reaction needs energy to occur. Now I learned that there is a separate, somewhat similar classification scheme of exergonic and endergonic reactions.
This is a very useful metric for predicting what compounds will form under certain conditions and the TOTAL potential energy changes however.. the 2nd law of thermodynamics tells us that we cant use ALL of the energy in a chemical reaction to do work, only a small amount of it.
Energy of the reaction system decreases relative to that of the surounding, i.e. the surrounding becomes hotter.
Energy of reactants is greater than that of products. Energy of the reaction system decreases relative to that of the surounding, i.e. the surrounding becomes hotter. Exergonic reaction. Energy released, has a special name called Gibbs energy or Gibbs free energy. Energy reactants is greater than that of the products.
Yes, all exergonic reactions are exothermic. Consider a reaction occurring spontaneously we know that energy would be released i.e. ' Δ H is negative' (since a reaction or process absorbing energy makes it non-spontaneous) and according to the second law of thermodynamics, entropy (or disorder) of the system must increase.
The difference between exergonic reactions and exothermic reactions lies in the processes through which they release energy. During exergonic reactions, the breaking of weak bonds releases the energy scientists refer to as free energy. Exothermic reactions release energy to their surroundings, usually in the form of heat.
Exothermic refers to spontaneous chemical reactions where something releases energy into the surroundings, usually in the form of heat. When oxygen combines with iron or a metal that contains iron, the result is extra energy. This energy then transfers to its surroundings in the form of rust.
Endothermic chemical reactions are ones that need an activation energy to begin the absorption of energy. Their opposites, exothermic reactions, are spontaneous and involve the process of transferring energy. One example of endothermic reactions is baking bread.
Their opposites, endergonic and exothermic reactions, also have their place in helping to understand thermodynamics. Reactions that absorb energy do so to create stronger products.
Exothermic reactions release energy to their surroundings, usually in the form of heat. This article will discuss what exergonic and exothermic mean, as well as their counterparts, endergonic and endothermic reactions. We will then explore how the processes work and look in their environment.
Exothermic reactions release energy, usually in the form of heat, into their surroundings. These chemical reactions can also transfer light or sound energy. The system releases the energy in order to keep the energy the same within the system.
Various chemical reactions can occur naturally or with added energy, and exergonic and exothermic are the two types of energy-releasing reactions. These chemical reactions are key components of thermodynamics, as they involve the transfer of energy with heat as a product.
It is a reaction process wherein energy is released from a system usually in the form of heat and light such as explosions, flames, and sparks. An exothermic reaction may also come in the forms of electricity and sound.
An exergonic reaction is a type of chemical reaction that releases free energy to the surroundings. The final state of this reaction is less than its initial state.
Some examples of exergonic reactions are: catabolism, respiration, and the breakdown of sugars through cellular metabolism.
Marcellin Berthelot was the person who coined the term “exothermic.”. Exothermic processes are commonly applied in the physical sciences wherein there is an involvement of chemical reactions, particularly the conversion of chemical bond energy to heat or thermal energy.
Some examples are: the combustion of fuels such as wood, coal, oil, and petroleum, condensation of rain from water vapor, the mixing of alkalis and acids, and the setting of cement and concrete.
She has taught science courses at the high school, college, and graduate levels. Endergonic and exergonic are two types of chemical reactions, or processes, in thermochemistry or physical chemistry. The names describe what happens to energy during the reaction.
Endothermic reactions offer good examples, as they absorb heat. Mix together baking soda (sodium carbonate) and citric acid in water. The liquid will get cold, but not cold enough to cause frostbite.
For example, it takes energy to start a fire, but once combustion starts, the reaction releases more light and heat than it took to get it started. Endergonic reactions and exergonic reactions are sometimes called reversible reactions.
Endergonic reactions absorb energy from their surroundings.
If the temperature of the surroundings decreases, the reaction is endothermic.
You cannot tell how quickly a reaction will occur based on whether it is endergonic or exergonic. Catalysts may be needed to cause the reaction to proceed at an observable rate. For example, rust formation (oxidation of iron) is an exergonic and exothermic reaction, yet it proceeds so slowly it's difficult to notice the release of heat to the environment.
The change in the standard Gibbs Free Energy (G) of an endergonic reaction is positive (greater than 0). The change in entropy (S) decreases. Endergonic reactions are not spontaneous. Examples of endergonic reactions include endothermic reactions, such as photosynthesis and the melting of ice into liquid water.
An exothermic reaction is a chemical or physical reaction that releases heat. It gives net energy to its surroundings. That is, the energy needed to initiate the reaction is less than the energy released.
This was because in the classical practice of chemistry, chemists saw energy flowing into and out of reactions in terms of hate therefore was called exothermic or endothermic, however the thought was not that it was so much heat flowing in and out but that it was energy required to go in and out. Today, especially in fields related to biology such as biochemistry, chemists are rec
They both mean that heat is absorbed, but the difference is that endothermic is a relative change in enthalpy, whilst endergonic refers to the relative change in free energy of a system. So in an endothermic reaction, the temperature of the surroundings decrease. In an endergonic reaction, energy is absorbed from the surroundings.
An exothermic reaction is a chemical reaction that releases energy by light or heat. It is the opposite of an endothermic reaction.
Δ H is the heat absorbed or released by the system at constant T and P. If heat is released, Δ H < 0 and the process is exothermic. If Δ H > 0 heat is absorbed and the process is endothermic. (Therm refers to heat.)
The suffix -ergonic refers to work because the Gibbs function refers to the amount of work a process can perform if the free energy change is negative or the work required to make the process go if the free energy change is positive.
They are not necessarily the same. An exothermic reaction requires an input of heat to occur, whereas an endergonic reaction requires an input of some form of energy, not necessarily heat energy, to occur.
Which type of reaction is endergonic?
Does an exergonic reaction release energy?
What is the difference between exergonic and endergonic reactions?
What is endergonic vs exergonic?
Do exergonic reactions release energy?
What are examples of endergonic and exergonic reactions?
Exothermic Vs. Exergonic
Exothermic
Exergonic
Difference Between Exothermic and Exergonic
Conclusion
What is the difference between an endothermic and an exothermic reaction?
What law of thermodynamics tells us that we cant use all of the energy in a chemical reaction to?
What happens to the energy of the reaction system relative to that of the surrounding?
Is the energy of reactants greater than that of products?
Is an exergonic reaction exothermic?
What is the difference between exergonic and exothermic reactions?
What is exothermic energy?
What is an endothermic reaction?
What are the opposites of exothermic reactions?
What is the energy released by exothermic reactions?
How does exothermic energy transfer?
What are the two types of energy releasing reactions?
What is exothermic reaction?
Why do exergonic reactions not require energy?
What are some examples of exergonic reactions?
Who coined the term "exothermic"?
What are some examples of combustion?
What is an endergonic and exergonic reaction?
How to absorb heat from an endothermic reaction?
What is the term for the reaction that releases more light and heat than it took to start a fire?
What type of reaction absorbs energy from its surroundings?
What happens when the temperature of the surroundings decreases?
Is rust an exergonic reaction?
Is endergonic entropy positive?
What is exothermic reaction?
Why is it called exothermic or endothermic?
What is the difference between endothermic and endergonic?
What is the opposite of an endothermic reaction?
What is the term for heat absorbed or released?
What does the suffix "ergonic" mean?
Is an exothermic reaction the same as an endergonic reaction?

Understanding The Meanings of Exergonic and Exothermic
Identifying Exergonic and Exothermic Reactions
- The idea of absorbing and releasing energy to maintain equilibrium within systems is a fairly abstract one. Being able to connect the definitions to pictures will make these reactions more concrete.
Differences in Reactions That Release Or Absorb Energy
- Even though exergonic and exothermic reactions seem very similar, they do have their differences as well. They not only have different qualities from their counterparts but from their opposite reactions as well. Exergonic reactions are spontaneous chemical reactions that create stronger bonds from breaking weaker ones. The energy from the weaker bonds then releases or transfer…
Final Thoughts
- Exergonic and exothermic reactions may seem exactly the same, but, in reality, they have their own distinct properties. Exothermic reactions are exergonic reactions, but exergonic refers more to a process and exothermic to a more specific reaction. There are also differences between reactions that transfer energy and reactions that absorb it. Under...