Isotopic analysis can be used to understand the flow of energy through a food web, to reconstruct past environmental and climatic conditions, to investigate human and animal diets in the past, for food authentification, and a variety of other physical, geological, palaeontological and chemical processes.
What is isotope analysis?
Isotope analysis is the identification of isotopic signature, the abundance of certain stable isotopes and chemical elements within organic and inorganic compounds. Isotopic analysis can be used to understand the flow of energy through a food web, to reconstruct past environmental and climatic conditions,...
What are the methods of isotopic analysis in the ocean?
Isotopic Analysis 1 Nitrogen Isotopes in the Ocean. The isotopic analysis of N relies on the generation of a stable gas, typically N2, as the analyte for isotope ratio mass spectrometry. 2 Marine Biogeochemistry. ... 3 Alberta Oil Sands. ... 4 Isotope Tracer Methods for Studies of the Marine Nitrogen Cycle. ... 5 Oxygen. ...
What are isotopes used for in archaeology?
A wide range of archaeological materials such as metals, glass and lead-based pigments have been sourced using isotopic characterization. Particularly in the Bronze Age Mediterranean, lead isotope analysis has been a useful tool for determining the sources of metals and an important indicator of trade patterns.
Why are isotopes important in food webs?
Certain isotopes can signify distinct primary producers forming the bases of food webs and trophic level positioning. The stable isotope compositions are expressed in terms of delta values (δ) in permil (‰), i.e. parts per thousand differences from a standard.

What can isotopic analysis tell us?
Stable isotope analysis, the study of the nuances of elements in archaeological materials, can unlock all sorts of secrets about climate, diet, and the geographical origins of bones and other materials. Stable isotopic analysis looks at the isotopes—atoms with extra or missing neutrons—of different elements.
How are isotopes used in forensics?
For several years, forensic scientists have been able to use isotopes found in human hair as markers that can indicate a region of the country where a person was living because many water supplies have unique isotopic signatures that are captured in hair.
How do you read isotopic data?
Reading Isotope Data The resultant isotopic signature of a sample is expressed using a delta (δ) followed by the isotope number and the symbol of the element being measured. Oxygen isotope measurements are read as δ18O, or delta oxygen eighteen, and carbon is read as δ13C, or delta carbon thirteen.
How are isotopes used in anthropology?
By examining multiple isotopes in different tissues, anthropologists can reconstruct changes in residence patterns. For example, isotopes in enamel bioapatite (mineral fraction) do not change throughout life, so teeth provide a record of where a person lived when a particular tooth formed.
How can isotope testing help investigators find a missing person?
But unlike DNA analysis, which can provide a near perfect match with an individual, stable isotope analysis cannot identify people. What it can reveal instead are subtle clues about diet, geography and movements of a person in the months and years before their death.
What are the three main kinds of isotope tissue samples that are collected from the human body?
Prior studies evaluating animal ecology using stable isotopes have relied on the use of three broad types of samples: (1) Primary samples (i.e., muscle, hair, feces) collected from live animals or animals with a known time of death; (2) Found samples (i.e., salvaged road kill, scat, hair or feathers) with an unknown ...
How do you measure isotopes?
The isotopic ratio is measured using an isotope ratio mass spectrometer (IRMS). There are two main types, or modes, of IRMS, dual inlet (DI) and continuous flow (CF) each with their own advantages to different applications. Stable isotope ratios are expressed in delta values relative to international scales.
What is isotope method?
isotope dilution, radiochemical method of analysis for measuring the mass and quantity of an element in a substance. The procedure involves adding to a substance a known quantity of a radioisotope of the element to be measured and mixing it with the stable isotope of the element.
What is isotope identification?
Radioisotope Identification Devices (RIIDs) are instruments that are designed to determine the identity of radioactive materials by measuring the energy of the emitted gamma rays.
Why are isotopes used in Archaeology?
Archaeology: From Dig to Lab and Beyond Stable isotopes have a stable nucleus that does not decay. Their abundance, therefore, stays the same over time, which allows for many useful applications in archaeology (and other disciplines like ecology or forensic science).
What is isotope analysis in Archaeology?
Stable isotope analysis is a scientific technique which is used by archaeologists and other scholars to collect information from an animal's bones to identify the photosynthesis process of the plants it consumed during its lifetime.
Who invented isotope analysis?
The existence of isotopes was first suggested in 1913 by the radiochemist Frederick Soddy, based on studies of radioactive decay chains that indicated about 40 different species referred to as radioelements (i.e. radioactive elements) between uranium and lead, although the periodic table only allowed for 11 elements ...
How do forensic scientists use analysis of hydrogen and oxygen isotopes to assist in solving crimes?
How do forensic scientists use analysis of hydrogen and oxygen isotopes to assist in solving crimes? The human body is made up of water. Forensic scientists examine the presence of oxygen and hydrogen isotopes that are found in our body tissues, including our hair through chemical and radiological analysis.
Which of the following is an example of forensics?
Forensics is the application of science in a legal setting. An example of modern forensics evidence is the use of DNA profiling. Sources of DNA include blood, hair, semen, saliva, bone and tissue. Fingerprints can be detected and used for forensic purposes.
How do isotopes end up in hair?
There are different concentrations of these isotopes in different regions of the world. When people drink the water in a given region, the isotopes end up in their hair in the same concentrations.
What can we determine about someone just by looking at the chemical makeup of their hair?
Hair analysis is very useful for drug testing, especially when looking at long-term usage. It's often used to test for the presence of marijuana, cocaine, amphetamines, and heroin. It may even be able to detect chronic alcohol abuse.
Why can’t we use isotopic dating techniques with sedimentary rocks?
An important assumption that we have to be able to make when using isotopic dating is that when the rock formed none of the daughter isotope was present (e.g., 40 Ar in the case of the K-Ar method). A clastic sedimentary rock is made up of older rock and mineral fragments, and when the rock forms it is almost certain that all of the fragments already have daughter isotopes in them. Furthermore, in almost all cases, the fragments have come from a range of source rocks that all formed at different times. If we dated a number of individual grains in the sedimentary rock, we would likely get a range of different dates, all older than the age of the rock. It might be possible to date some chemical sedimentary rocks isotopically, but there are no useful isotopes that can be used on old chemical sedimentary rocks. Radiocarbon dating can be used on sediments or sedimentary rocks that contain carbon, but it cannot be used on materials older than about 60 ka.
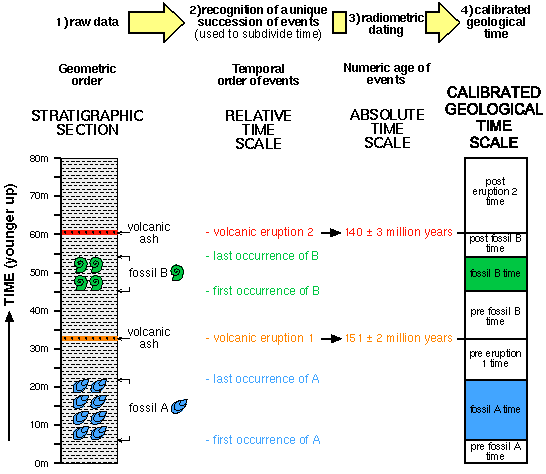
Why do fossils only provide relative ages?
Originally fossils only provided us with relative ages because, although early paleontologists understood biological succession, they did not know the absolute ages of the different organisms. It was only in the early part of the 20th century, when isotopic dating methods were first applied, that it became possible to discover the absolute ages ...
What is radiocarbon dating?
Radiocarbon dating (using 14 C ) can be applied to many geological materials, including sediments and sedimentary rocks, but the materials in question must be younger than 60 ka. Fragments of wood incorporated into young sediments are good candidates for carbon dating, and this technique has been used widely in studies involving late Pleistocene glaciers and glacial sediments. An example is shown in Figure 8.16; radiocarbon dates from wood fragments in glacial sediments have been used to estimate the time of the last glacial advance along the Strait of Georgia.
Can sedimentary rocks be dated isotopically?
If we dated a number of individual grains in the sedimentary rock, we would likely get a range of different dates, all older than the age of the rock. It might be possible to date some chemical sedimentary rocks isotopically, but there are no useful isotopes that can be used on old chemical sedimentary rocks.
How to analyze isotopic N?
The isotopic analysis of N relies on the generation of a stable gas as the analyte for isotope ratio mass spectrometry. Online combustion to N2 is currently the standard method for the preparation of a N sample for isotopic analysis. With ‘off-the-shelf’ technology, a typical sample size requirement is 1–2 μmol N per analysis. Gas chromatography followed by combustion to N 2 is improving as a technique for specific organic compounds, amino acids in particular, although the polarity of many N compounds remains a challenge. Liquid chromatography is also being explored. There are standard methods of collection for most bulk forms of particulate N (PN) in the ocean. Shallow and deep samples of suspended PN are filtered onto glass fiber filters. Sinking PN is collected by sediment traps. Zooplankton can be picked from filtered samples or net tows, and particulates can be separated into size classes. In the case of dissolved forms of N, the species of interest must be converted selectively to a gas or other extractable form for collection. Since the 1970s, the δ15 N values of marine nitrate ( NO 3 − ), nitrite ( NO 2 − ), and ammonium ( NH 4 +) have been analyzed by conversion to ammonia gas and collection of the cationic ammonium form for subsequent conversion to N 2 (often referred to as the ammonia ‘distillation’ and ‘diffusion’ methods). Recently, more sensitive isotope analysis methods (requiring only 5–10 nmol of N per analysis) have been developed for nitrate and nitrite in which these species are converted to nitrous oxide (N 2 O), followed by isotopic analysis of this gas (the ‘bacterial’ or ‘denitrifier’ method and the ‘chemical’ or ‘azide’ method). The N 2 O produced by these methods (or naturally occuring N 2 O) is analyzed by a purge and trap system, followed by gas chromatography and isotope ratio mass spectrometry. The N 2 O-based methods also allow for oxygen isotope analysis of nitrate and nitrite, a measurement not previously possible in seawater. In addition, they provide a cornerstone for isotopic analysis of other dissolved forms of N, such as dissolved organic N (DON) and ammonium ( NH 4 + ), which can be converted to nitrate and/or nitrite. With respect to dissolved gases, methods of collection and isotopic analysis have been developed for N 2 and N 2 O, with recent progress on isotopomer analysis of N 2 O (i.e., distinguishing 15 N 14 N 16 O from 14 N 15 N 16 O).
What is the device used to measure oxygen isotopes?
Figure 6-1.29. Excimer laser device for O isotope analysis on silicate materials (after Rumble et al., 1997 ). The extraction system is directly connected to a mass spectrometer and oxygen isotopes are measured on O 2. The molecular sieve 5 Å is used to trap O 2 before release into the mass spectrometer.
What is the name of the laser used to determine oxygen isotopes?
Bao & Thiemens (2000) used a CO 2 laser system and BrF 5 as a fluorination agent for determination of oxygen isotopes ( 18 O, 17 O and Δ 17 O) in sulfates. Details of their method are given in Chapter 6-4.4.
What is the error on Hf isotopic data?
Error on Hf-isotopic data is 2s.e. in the sixth digit.
What are the conclusions of the isotopic analysis of Hoxnian carbonates?
The δ18 O composition of Hoxnian carbonates is compared with that of modern soil carbonates and carbonates formed under interglacials that were significantly warmer than the present day. This comparison indicates that climates during the Hoxnian may have been warmer than the present but not exceptionally warm by the standards of other interglacials. Human occupation during this interglacial, therefore, occurred during climates that were broadly comparable to those experienced during the Holocene. The isotopic studies show no evidence for the exceptional warmth that has been claimed for MIS 11 from a range of proxy evidence. This is consistent with other evidence from northwest and western Europe.
What are isotopes used for?
Therefore, isotope ratios can be used as a molecular signature to reflect an individual's habits and daily exposure to the environment.
Why do scientists use isotopes in human hair?
For several years, forensic scientists have been able to use isotopes found in human hair as markers that can indicate a region of the country where a person was living because many water supplies have unique isotopic signatures that are captured in hair. A person drinks the local water, and an isotopic record of that water is recorded in ...
What is the purpose of isotopes in hair?
Researchers investigate the potential of using isotopes in specific amino acids in human hair as a forensic tool to provide information about an individual's age, sex, race, body mass, genetic disorders, health, and region of origin. For several years, forensic scientists have been able to use isotopes found in human hair as markers ...
What are the different forms of the same chemical element that can be measured accurately and compared?
Isotopes are different forms of the same chemical element that can be measured accurately and compared. For forensic purposes, isotope ratios of different elements like carbon, nitrogen, oxygen, and hydrogen will vary based on a person's exposure to environmental factors such as drinking water and nutrients in food.
How do scientists determine where humans are from?
corn). Scientists can roughly determine human regions of origin or residence by measuring carbon isotope ratios in relation to dietary plants and other foods.
How to specify an isotope?
In fact, a common way of specifying an isotope is to use the notation "element name-mass number," as we've already seen with carbon-. Importantly, we can use an isotope's mass number to calculate the number of neutrons in its nucleus.
What is isotope notation?
Isotopic notation shows the atomic number, mass number and charge of an isotope in a single symbol. For example, consider the isotopic notation for neutral hydrogen- and the magnesium- cation: Isotope notation for helium-4 and magnesium-24.
What does the mass spectrum show?
The mass spectrum of a sample shows the relative abundances of the ions on the y-axis and their ratios on the x-axis. If for all ions, then the x-axis can instead be expressed in units of atomic mass ( ).
How is a mass spectrometer injected?
A diagram of a mass spectrometer. A sample is injected into the machine, vaporized by a heater, and then ionized by a stream of high-energy electrons. The resulting ions are accelerated through parallel electric plates and then deflected in a magnetic field before they reach a detector. Image credit: " Atomic Structure and Symbolism: Figure 5 " by OpenStax Chemistry, CC BY 4.0.
Why use unified mass units?
Using unified atomic mass units makes the masses of these particles much easier to understand and compare. For example, we can see from the numbers above that protons and neutrons are much more massive than electrons (nearly 2000 times more, to be precise!). This tells us that the bulk of an atom's mass is located in its nucleus.
Why do we use mass spectrometry?
These days, we already know the average atomic masses of most of the elements on the periodic table, so it's not often necessary to analyze individual elements using mass spectrometry—except to teach students! Most of the time, working chemists use mass spectrometry in the lab to help them determine the chemical formulas or structures of unknown molecules and compounds. Mass spectrometry also finds valuable application in other fields, including medicine, forensics, space exploration and more. Whether it's being used to analyze the atmosphere of an unexplored planet or characterize a newly-created molecule, mass spectrometry is instrumental to the advancement of scientific knowledge and understanding.
Where is the bulk of an atom located?
This tells us that the bulk of an atom's mass is located in its nucleus. In fact, it turns out that the mass of an electron is so small relative to the masses of protons and neutrons that electrons are considered to have a negligible effect on the overall mass of an atom.
Why is the study of isotopes important?
The study of environmental isotopes plays an important role in understanding past climate change. It is this understanding of changes in the past that holds the key to predicting future changes --changes that may not only influence global temperatures, but also energy needs, availability of drinking water, and adequate food supplies.
Where are isotopes preserved?
This is because these isotopes, both stable and radioactive, are preserved in various natural archives such as sediments in lakes and oceans, ice in glaciers or polar ice caps, water in precipitation and oceans, and even trees.
Why is carbon dioxide important?
Carbon dioxide is also an important greenhouse gas, making the study of its movement over time all the more important to understanding its role in climate change. Isotopes of hydrogen and oxygen can also be used to date and study the movements of water, since water is made up of these two elements.
How do we get information about climate?
One of the greatest sources of information on past climate comes from studies of polar ice caps and mountain glaciers. In such studies, cores are drilled through the ice down to bedrock. The isotope composition from this ice core can then be determined to estimate such things as temperature, precipitation rate, wind speed, and greenhouse gas concentration over extremely long periods, since the isotopes are frozen in the ice in a virtually timeless record.
How to determine the age of carbon-14?
One of the most common uses of isotopes, particularly carbon-14 ( 14 C), is to determine age by measuring the decay products of 14 C over time and, based on the known decay rate for 14 C , estimating when the 14 C would have first been absorbed. This technique, known as radiocarbon dating, is widely used to date fossils and sediments, but can also be used in atmospheric studies, since carbon dioxide is present in large quantities in the earth′s atmosphere. Tracing of 14 C and elated stable isotopes such as carbon-13 ( 13 C) and oxygen-18 ( 18 O) in atmospheric carbon dioxide can be used to study interactions between oceanic, atmospheric, and terrestial carbon reservoirs over time. Carbon dioxide is also an important greenhouse gas, making the study of its movement over time all the more important to understanding its role in climate change.
What is the purpose of tracing 14 C and elated stable isotopes?
Tracing of 14 C and elated stable isotopes such as carbon-13 ( 13 C) and oxygen-18 ( 18 O) in atmospheric carbon dioxide can be used to study interactions between oceanic, atmospheric, and terrestial carbon reservoirs over time.
What is radiocarbon dating?
This technique, known as radiocarbon dating, is widely used to date fossils and sediments, but can also be used in atmospheric studies , since carbon dioxide is present in large quantities in the earth′s atmosphere. Tracing of 14 C and elated stable isotopes such as carbon-13 ( 13 C) and oxygen-18 ...
Why do fossils only provide relative ages?
Originally fossils only provided us with relative ages because, although early paleontologists understood biological succession, they did not know the absolute ages of the different organisms. It was only in the early part of the 20th century, when isotopic dating methods were first applied, that it became possible to discover the absolute ages ...
Can sedimentary rocks be isotopically dated?
That could be useful information, but it would not provide an accurate date for the rock in question. It might be possible to directly date some chemical sedimentary rocks isotopically, but there are no useful isotopes that can be used on old chemical sedimentary rocks.
