
Disinfectants work to discourage bacterial growth by killing bacteria or retarding growth processes. There are other ways to affect bacterial growth too, like putting your food in the refrigerator or cooking food to a high temperature. Try testing the growth of bacteria in different temperatures.
What kind of disinfectant spray stops bacteria?
What kind of disinfectant spray stops bacteria? Bleach is one of the best “disinfectants” but the problem is that it does also remove the color from materials if the bleach is sprayed or sprinkled on the surface.
What Kills germs the best?
You can also prevent infections through:
- Hand-washing. Often overlooked, hand-washing is one of the easiest and most effective ways to protect yourself from germs and most infections. ...
- Vaccines. Vaccination is your best line of defense for certain diseases. ...
- Medicines. Some medicines offer short-term protection from certain germs. ...
Does bleach kill germs?
Bleach kills germs, including viruses and bacteria
What is the difference between sanitizers and disinfectants?
Sanitizing lowers the number of germs on surfaces or objects—either by killing them or removing them—to a safe level, according to public health standards or requirements. Disinfecting kills germs...
How do disinfectants destroy microbes?
Disinfectants work by destroying the protective membrane or protein envelope that covers a microbe. Once this is destroyed, the internal structure and contents leak out, thereby killing the microbe or rendering it non-viable to reproduce, as in the case of viruses.
Are disinfectants used to kill bacteria?
Antiseptics and disinfectants are both widely used to control infections. They kill microorganisms such as bacteria, viruses, and fungi using chemicals called biocides. Disinfectants are used to kill germs on nonliving surfaces. Antiseptics kill microorganisms on your skin.
How do chemicals kill bacteria?
They act by “denaturing” or altering the molecular structure of bacterial proteins, destroying the cell. Alcohols kill vegetative forms of bacteria but have no action on spores or viruses.
What is the mechanism of action of disinfectant?
The author describes the actions of disinfectants on the external membrane, cytoplasmic membrane and energy metabolism of cells; these actions include rupture of the membrane, loss of permeability and coagulation of the cytoplasm.
Does disinfectant destroy all harmful bacteria?
But to effectively kill harmful microorganisms such as COVID-19, you'll also need to disinfect common surfaces. Disinfecting kills most harmful bacteria, viruses, and fungi.
What is the purpose of a disinfectant?
Disinfecting uses chemicals (disinfectants) to kill germs on surfaces and objects. Some common disinfectants are bleach and alcohol solutions. You usually need to leave the disinfectant on the surfaces and objects for a certain period of time to kill the germs.
How can bacteria be killed?
In fact, at room temperature, bacteria growth can double every 20 minutes. It is a myth that bacteria are killed at temperatures below 40 degrees. In fact, bacteria growth is slowed, but not stopped. The only way to kill bacteria by temperature is by cooking food at temperatures of 165 degrees or more.
Does disinfectant reduce bacteria to a safe level?
Disinfecting kills germs on surfaces or objects. Disinfecting works by using chemicals to kill germs on surfaces or objects. This process does not necessarily clean dirty surfaces or remove germs, but by killing germs on a surface after cleaning, it can further lower the risk of spreading infection.
What is the process of killing harmful bacteria in a surface?
Disinfection is the treatment of surfaces/equipment using physical or chemical means such that the amount of microorganisms present is reduced to an acceptable level.
What's the difference between antibacterial and disinfectant?
Sanitizing kills bacteria on surfaces using chemicals. It is not intended to kill viruses. Yes, EPA registers products that sanitize. Disinfecting kills viruses and bacteria on surfaces using chemicals.
What is disinfectant in microbiology?
A disinfectant can be defined as an antimicrobial agent that can be applied on the surface of some objects in order to destroy the microorganisms residing on it.
What are the mode of action of disinfectants and antiseptics?
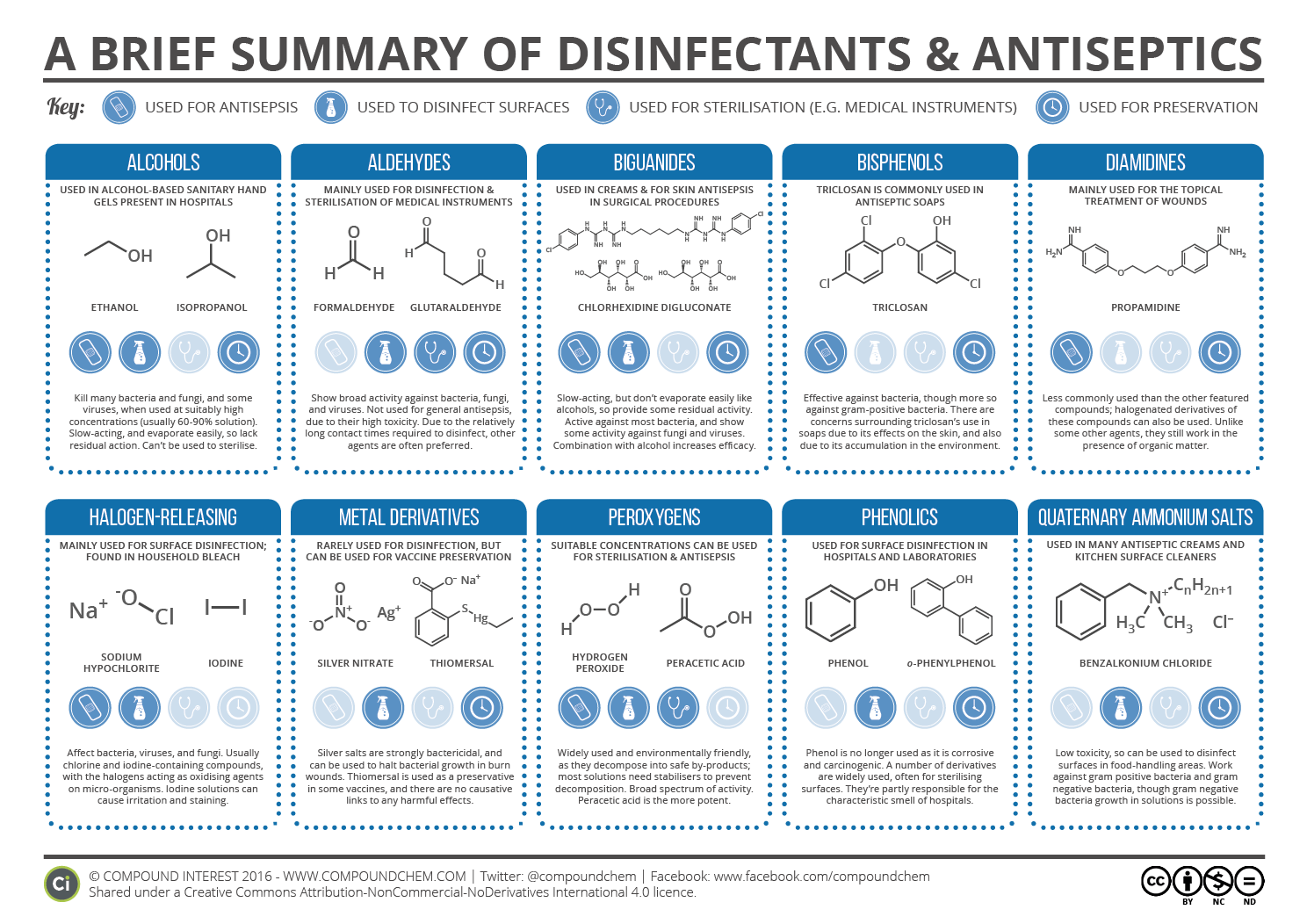
TABLE 2TargetAntiseptic or disinfectantMechanism of actionDiaminesInduction of leakage of amino acidsPHMB, alexidinePhase separation and domain formation of membrane lipidsPhenolsLeakage; some cause uncouplingCross-linking of macromoleculesFormaldehydeCross-linking of proteins, RNA, and DNA11 more rows
What substance kills bacteria?
Hydrogen peroxide is active against a wide range of microorganisms, including bacteria, yeasts, fungi, viruses, and spores 78, 654. A 0.5% accelerated hydrogen peroxide demonstrated bactericidal and virucidal activity in 1 minute and mycobactericidal and fungicidal activity in 5 minutes 656.
What a disinfectant meaning?
Listen to pronunciation. (DIS-in-fek-tunt) Any substance or process that is used primarily on non-living objects to kill germs, such as viruses, bacteria, and other microorganisms that can cause infection and disease. Most disinfectants are harsh chemicals but sometimes heat or radiation may be used.
Does disinfectant reduce bacteria to a safe level?
Disinfecting kills germs on surfaces or objects. Disinfecting works by using chemicals to kill germs on surfaces or objects. This process does not necessarily clean dirty surfaces or remove germs, but by killing germs on a surface after cleaning, it can further lower the risk of spreading infection.
What is disinfection?
Disinfection describes a process that eliminates many or all pathogenic microorganisms, except bacterial spores, on inanimate objects (Tables 1 and 2). In health-care settings, objects usually are disinfected by liquid chemicals or wet pasteurization.
How do disinfectants work?
Disinfectants work by destroying the protective membrane or protein envelope that covers a microbe. Once this is destroyed, the internal structure and contents leak out, thereby killing the microbe or rendering it non-viable to reproduce, as in the case of viruses. Viruses technically cannot be killed because they’re not alive in the first place.
How do disinfectants break phospholipid bilayers?
Disinfectants break the molecular bonds of the phospholipid bilayer either by oxidation reaction or through binding actions that make the membrane more permeable, such as in the case of ammonium compounds. Depending on the type and concentration of the disinfectant, as well as the type or strain of microbe, the microbe may either be killed or have its metabolic functions and growth inhibited, putting it in stasis.
What is the difference between sterilisation and disinfection?
Sterilisation, on the other hand, is a more extreme procedure of destroying all potentially harmful microbes on a surface or object.
What is the best solvent for hand sanitizer?
Alcohols like ethyl alcohol and isopropyl alcohols are commonly used in hand sanitisers. They destroy microbes by dissolving the lipids on the viral or bacterial protective membranes. Other types of solvents like acetone work similarly, but aren’t as safe as ethyl and isopropyl alcohols.
Why do you need to disinfect your bathroom?
Even if you apply disinfectants, microbes can make a comeback . This is one of the reasons you should regularly disinfect your bathroom.
What happens when a membrane protein is denatured?
Denature proteins: Membrane proteins can bind with the molecules of disinfectants, causing them to be disorganised and weak. Some proteins may become denatured
What is the bilayer of a bacteria cell?
Bacterial cells are protected by a bi-layer of phospholipids. The hydrophilic heads of the cell membrane face outwards and inwards. The hydrophobic tails are sandwiched in between the outer membrane and the inner membrane.
How do we kill bacteria?
We kill bacteria by causing damage to proteins within the outer layers of the bacterial cell and the whole cell consequently simply splits open and dies. Learn more below.
What areas in your home need disinfecting?
Your kitchen is a crucial place where hygiene should come first. It’s where you prepare food, and some food-borne illnesses can be found including food poisoning. E. Coli bacteria originate from raw meat, listeria from cheeses, and salmonella from chicken and raw eggs. Help stop the spread of bacteria and viruses by disinfecting work surfaces after each food preparation with Dettol Trigger Kitchen* spray, and consistent use of an antibacterial soap like Dettol Hand Wash* after touching any raw meat or eggs. Refrigerators and cupboard handles should also be cleaned on a weekly basis.
Do disinfectants need to be treated?
Always remember, disinfectants are chemical substances and need to be treated with care – always read the product labels carefully before using.
Is it good to disinfect your home?
Cleaning your home isn’t always enough – that’s why disinfecting your home is a crucial part of supporting your family’s protection. Read more to learn about our top tips.
Can a virus replicate itself?
The aim of a virus is to replicate itself, but it cannot do this on its own. It needs to get inside of us to infect our healthy cells. Surfaces can easily become contaminated with viral particles, especially if somebody who is infected has been in close proximity to it. These surfaces provide temporary homes for the virus to transfer, primarily to the hands of those who come into contact with it. Contaminated hands can then transfer some viruses present to our faces. This provides the virus with a route to enter our bodies.
How do disinfectants work?
Disinfectants work by adversely affecting the microbe’s cell by either disrupting its physical makeup or by blocking its energy-yielding or synthetic processes. It is important to remember that disinfectants are consumed or expend their ability to affect the microorganisms during this process. A brief history of disinfectants.
Why are disinfectants bad for you?
The very reason that these elements make food disinfectants is also their biggest drawback. They attack cells and destroy their structures. They are, however, non-selective and can also be aggressive and corrosive to human skin or other environmental surfaces.
Why are quats the most popular disinfectant?
Quats are by far the most popular choice of disinfectants today because of their low cost and selective aggression. They have broad spectrum effectiveness against a range of microorganisms and provide the highest overall value to the customer.
What is the chemical that kills microorganisms?
Disinfectants are the chemical agents that either inhibit microbial activities and growth or are lethal and kill the microorganisms. In man’s struggle to control the microbes responsible for disease and illness many organic and inorganic chemicals have been found to be toxic to microorganisms.
What are the most common chemicals used in disinfectant products?
What are the different chemical compounds used in manufacturing disinfectant products? Halogens are a family of elements, the most common being fluorine, chlorine, bromine, and iodine.
Why isn't fluorine used in chemistry?
Fluorine is not used because of the extreme toxicity associated with fluoride compounds. Phenol, originally extracted from coal tar, was known as carbolic acid and is produced synthetically today. In its raw form phenol is a white poisonous corrosive crystal.
What is the difference between bacteria and fungi?
Bacteria are microscopic single cell living organisms. Fungi are more advanced multi-cellular organisms which can be infectious. Mold and mildew are types of fungi which cause deterioration. Protozoa are small unicellular animal microorganisms. Some are parasitic and others can live free of a host.
What disinfectant is used to disinfect endoscopes?
Alcohols have been used to disinfect fiberoptic endoscopes 503, 504 but failure of this disinfectant have lead to infection 280, 505. Alcohol towelettes have been used for years to disinfect small surfaces such as rubber stoppers of multiple-dose medication vials or vaccine bottles.
How long does it take for chlorine to kill bacteria?
Low concentrations of free available chlorine (e.g., HOCl, OCl –, and elemental chlorine-Cl 2) have a biocidal effect on mycoplasma (25 ppm) and vegetative bacteria (<5 ppm) in seconds in the absence of an organic load 329, 418. Higher concentrations (1,000 ppm) of chlorine are required to kill M. tuberculosis using the Association of Official Analytical Chemists (AOAC) tuberculocidal test 73. A concentration of 100 ppm will kill ≥99.9% of B. atrophaeus spores within 5 minutes 541, 542 and destroy mycotic agents in <1 hour 329. Acidified bleach and regular bleach (5,000 ppm chlorine) can inactivate 10 6 Clostridium difficile spores in ≤10 minutes 262. One study reported that 25 different viruses were inactivated in 10 minutes with 200 ppm available chlorine 72. Several studies have demonstrated the effectiveness of diluted sodium hypochlorite and other disinfectants to inactivate HIV 61. Chlorine (500 ppm) showed inhibition of Candida after 30 seconds of exposure 54. In experiments using the AOAC Use-Dilution Method, 100 ppm of free chlorine killed 10 6 –10 7 S. aureus, Salmonella choleraesuis, and P. aeruginosa in <10 minutes 327. Because household bleach contains 5.25%–6.15% sodium hypochlorite, or 52,500–61,500 ppm available chlorine, a 1:1,000 dilution provides about 53–62 ppm available chlorine, and a 1:10 dilution of household bleach provides about 5250–6150 ppm.
What are the advantages of sodium dichloroisocyanurate?
The advantage of these compounds over the hypochlorites is that they retain chlorine longer and so exert a more prolonged bactericidal effect. Sodium dichloroisocyanurate tablets are stable, and for two reasons, the microbicidal activity of solutions prepared from sodium dichloroisocyanurate tablets might be greater than that of sodium hypochlorite solutions containing the same total available chlorine. First, with sodium dichloroisocyanurate, only 50% of the total available chlorine is free (HOCl and OCl – ), whereas the remainder is combined (monochloroisocyanurate or dichloroisocyanurate), and as free available chlorine is used up, the latter is released to restore the equilibrium. Second, solutions of sodium dichloroisocyanurate are acidic, whereas sodium hypochlorite solutions are alkaline, and the more microbicidal type of chlorine (HOCl) is believed to predominate 530-533. Chlorine dioxide-based disinfectants are prepared fresh as required by mixing the two components (base solution [citric acid with preservatives and corrosion inhibitors] and the activator solution [sodium chlorite]). In vitro suspension tests showed that solutions containing about 140 ppm chlorine dioxide achieved a reduction factor exceeding 10 6 of S. aureus in 1 minute and of Bacillus atrophaeus spores in 2.5 minutes in the presence of 3 g/L bovine albumin. The potential for damaging equipment requires consideration because long-term use can damage the outer plastic coat of the insertion tube 534. In another study, chlorine dioxide solutions at either 600 ppm or 30 ppm killed Mycobacterium avium-intracellulare within 60 seconds after contact but contamination by organic material significantly affected the microbicidal properties 535.
What is the most common disinfectant?
Hypochlorites, the most widely used of the chlorine disinfectants, are available as liquid (e.g., sodium hypochlorite) or solid (e.g., calcium hypochlorite). The most prevalent chlorine products in the United States are aqueous solutions of 5.25%–6.15% sodium hypochlorite (see glossary), usually called household bleach. They have a broad spectrum of antimicrobial activity, do not leave toxic residues, are unaffected by water hardness, are inexpensive and fast acting 328, remove dried or fixed organisms and biofilms from surfaces 465, and have a low incidence of serious toxicity 515-517. Sodium hypochlorite at the concentration used in household bleach (5.25-6.15%) can produce ocular irritation or oropharyngeal, esophageal, and gastric burns 318, 518-522. Other disadvantages of hypochlorites include corrosiveness to metals in high concentrations (>500 ppm), inactivation by organic matter, discoloring or “bleaching” of fabrics, release of toxic chlorine gas when mixed with ammonia or acid (e.g., household cleaning agents) 523-525, and relative stability 327. The microbicidal activity of chlorine is attributed largely to undissociated hypochlorous acid (HOCl). The dissociation of HOCI to the less microbicidal form (hypochlorite ion OCl ‑) depends on pH. The disinfecting efficacy of chlorine decreases with an increase in pH that parallels the conversion of undissociated HOCI to OCl ‑ 329, 526. A potential hazard is production of the carcinogen bis (chloromethyl) ether when hypochlorite solutions contact formaldehyde 527 and the production of the animal carcinogen trihalomethane when hot water is hyperchlorinated 528. After reviewing environmental fate and ecologic data, EPA has determined the currently registered uses of hypochlorites will not result in unreasonable adverse effects to the environment 529.
How does hydrogen peroxide work?
Hydrogen peroxide works by producing destructive hydroxyl free radicals that can attack membrane lipids , DNA, and other essential cell components . Catalase, produced by aerobic organisms and facultative anaerobes that possess cytochrome systems, can protect cells from metabolically produced hydrogen peroxide by degrading hydrogen peroxide to water and oxygen. This defense is overwhelmed by the concentrations used for disinfection 653, 654.
How long does it take for alcohol to kill tuberculosis?
In tests of the effect of ethyl alcohol against M. tuberculosis, 95% ethanol killed the tubercle bacilli in sputum or water suspension within 15 seconds 492. In 1964, Spaulding stated that alcohols were the germicide of choice for tuberculocidal activity, and they should be the standard by which all other tuberculocides are compared. For example, he compared the tuberculocidal activity of iodophor (450 ppm), a substituted phenol (3%), and isopropanol (70%/volume) using the mucin-loop test (10 6 M. tuberculosis per loop) and determined the contact times needed for complete destruction were 120–180 minutes, 45–60 minutes, and 5 minutes, respectively. The mucin-loop test is a severe test developed to produce long survival times. Thus, these figures should not be extrapolated to the exposure times needed when these germicides are used on medical or surgical material 482.
Which alcohol has the weakest bactericidal action?
The bactericidal activity of various concentrations of et hyl alcohol (ethanol) was examined against a variety of microorganisms in exposure periods ranging from 10 seconds to 1 hour 483. Pseudomonas aeruginosa was killed in 10 seconds by all concentrations of ethanol from 30% to 100% (v/v), and Serratia marcescens, E, coli and Salmonella typhosa were killed in 10 seconds by all concentrations of ethanol from 40% to 100%. The gram-positive organisms Staphylococcus aureus and Streptococcus pyogenes were slightly more resistant, being killed in 10 seconds by ethyl alcohol concentrations of 60%–95%. Isopropyl alcohol (isopropanol) was slightly more bactericidal than ethyl alcohol for E. coli and S. aureus 489.
How do disinfectants work?
Disinfectants work to discourage bacterial growth by killing bacteria or retarding growth processes. There are other ways to affect bacterial growth too, like putting your food in the refrigerator or cooking food to a high temperature. Try testing the growth of bacteria in different temperatures.
How to avoid bacteria in your mouth?
Keep your nose and mouth away from tubes, pipettes, or other tools that come in contact with bacterial cultures, in order to avoid ingesting or inhaling any bacteria.
How to use a cotton tipped nutrient agar plate?
With your free hand, open the lid of the matching nutrient agar plate and swipe the cotton-tipped applicator gently across the agar surface using a zig-zag motion. Immediately replace the lid of the agar plate and secure with a few pieces of clear tape. DO NOT set the lid down while you are streaking the agar because this can contaminate the lid and change your results!
How long to leave nutrient agar plates on?
Leave the nutrient agar plates on the cookie sheets or trays in a warm place for 2-4 days, until bacterial colonies are visible. Count the number of colonies on each nutrient agar plate and write your results in the data table in your lab notebook, along with any other observations you have.
How to transfer a sample from cutting board to agar plate?
Use the swabs to transfer a sample from the cutting board to the agar plate. Holding the wooden end of the swab, swipe the cotton end around one section of the cutting board using a circular motion. DO NOT allow the tip of the swab to contact anything else!
Is it safe to use bacteria?
However, for maximum safety, all bacterial cultures should always be treated as potential hazards. This means that proper handling, cleanup, and disposal are necessary. Below are a few important safety reminders.
Can bleach be used on clothes?
Use caution when handling the bleach, as it can ruin your clothes if spilled, and any disinfectant can be harmful if splash ed in your eyes.
What is disinfectant made of?
Disinfectant is described in the dictionary as “a chemical liquid that destroys bacteria”, however, Wikipedia’s explanation delves even deeper and explains that disinfectant is actually a chemical agent that is designed to destroy or to render micro-organisms inactive on surfaces.
What is the difference between cleaning and disinfecting?
Soap alone cannot disinfect surfaces. For it to be effective it has to be mixed with water and rubbed thoroughly until it becomes a lather, and since soap usually doesn’t contain chemicals, it might not kill all germs, although it will certainly lower the numbers and help combat the risk of spreading viruses.
Hillcross Hygiene
If you require disinfectant services in and around Kent, contact Hillcross Hygiene for a free quote.
How many mechanisms of action do disinfectants have?
In general, disinfectants have three mechanisms of action or ways that they affect or kill an organism: ...
Why are surfactants important in disinfectant cleaning?
In particular, surfactants are often important ingredients to disinfectant cleaning solutions because they achieve uniform wetting of surfaces and frequently help with cleaning.
What is chlorine used for?
Chlorine is a very common disinfectant used in a wide variety of cleaning solutions and applications — even in drinking water — because, even in very small amounts, it exhibits fast bactericidal action. Chlorine works by oxidizing proteins, lipids and carbohydrates. Hypochlorous acid, which is a weak acid that forms when chlorine is dissolved in water, has the most effect on the bacterial cell, targeting some key metabolic enzymes and destroying the organism. Chlorine compounds have also been shown to affect surface antigen in enveloped viruses and deoxyribonucleic acid (DNA) as well as structural alterations in non-enveloped viruses.
How does peroxygen kill spores?
Peroxygen compounds also kill spores by removing proteins from the spore coat, exposing its core to the lethal disinfectant.
What is the most widely used disinfectant?
Quaternary ammonium compounds (quats) are some of the most widely used disinfectants today because of their broad spectrum effectiveness. Quaternary ammonium compounds work by denaturing the proteins of the bacterial or fungal cell, affecting the metabolic reactions of the cell and causing vital substances to leak out of the cell, causing death.
What is the mechanism of action of alcohol?
Alcohol. Mechanism of action: Cross-linking, coagulating, clumping. Like many disinfectants, alcohols are generally considered to be non-specific antimicrobials because of their many toxic effects. Alcohols cause cell proteins to clump and lose their function.
Can phenols be used as sporicides?
Though phenols can act at the germination — beginning of growth — stage of bacterial spore development, this effect is reversible, making them unsuitable as sporicides.
What are antiseptics and disinfectants used for?
Antiseptics and disinfectants are used extensively in hospitals and other health care settings for a variety of topical and hard-surface applications. In particular, they are an essential part of infection control practices and aid in the prevention of nosocomial infections (277, 454). Mounting concerns over the potential for microbial contamination and infection risks in the food and general consumer markets have also led to increased use of antiseptics and disinfectants by the general public. A wide variety of active chemical agents (or “biocides”) are found in these products, many of which have been used for hundreds of years for antisepsis, disinfection, and preservation (39). Despite this, less is known about the mode of action of these active agents than about antibiotics. In general, biocides have a broader spectrum of activity than antibiotics, and, while antibiotics tend to have specific intracellular targets, biocides may have multiple targets. The widespread use of antiseptic and disinfectant products has prompted some speculation on the development of microbial resistance, in particular cross-resistance to antibiotics. This review considers what is known about the mode of action of, and mechanisms of microbial resistance to, antiseptics and disinfectants and attempts, wherever possible, to relate current knowledge to the clinical environment.
What is the process of sterilizing a product?
Sterilization refers to a physical or chemical process that completely destroys or removes all microbial life, including spores. Preservation is the prevention of multiplication of microorganisms in formulated products, including pharmaceuticals and foods. A number of biocides are also used for cleaning purposes; cleaning in these cases refers to the physical removal of foreign material from a surface (40).
What are biocides used for?
A summary of the various types of biocides used in antiseptics and disinfectants, their chemical structures , and their clinical uses is shown in Table Table1.1. It is important to note that many of these biocides may be used singly or in combination in a variety of products which vary considerably in activity against microorganisms. Antimicrobial activity can be influenced by many factors such as formulation effects, presence of an organic load, synergy, temperature, dilution, and test method. These issues are beyond the scope of this review and are discussed elsewhere (123, 425, 444, 446, 451).
Why are antiprotozoal cells not widely investigated?
One reason for this is the difficulty in culturing some protozoa (e.g., Cryptosporidium) under laboratory conditions. However, the different life stages (trophozoites and cysts) do provide a fascinating example of the problem of how changes in cytology and physiology can modify responses to antiseptics and disinfectants. Khunkitti et al. (251–255) have explored this aspect by using indices of viability, leakage, uptake, and electron microscopy as experimental tools.
What is the effect of alcohol on the cell membrane?
Little is known about the specific mode of action of alcohols, but based on the increased efficacy in the presence of water, it is generally believed that they cause membrane damage and rapid denaturation of proteins, with subsequent interference with metabolism and cell lysis (278, 337). This is supported by specific reports of denaturation of Escherichia colidehydrogenases (499) and an increased lag phase in Enterobacter aerogenes, speculated to be due to inhibition of metabolism required for rapid cell division (101).
What are the targets of viruses?
Viral targets are predominantly the viral envelope (if present), derived from the host cell cytoplasmic or nuclear membrane; the capsid, which is responsible for the shape of virus particles and for the protection of viral nucleic acid; and the viral genome. Release of an intact viral nucleic acid into the environment following capsid destruction is of potential concern since some nucleic acids are infective when liberated from the capsid (317), an aspect that must be considered in viral disinfection. Important considerations in viral inactivation are dealt with by Klein and Deforest (259) and Prince et al. (384), while an earlier paper by Grossgebauer is highly recommended (189).
Is alcohol a good antimicrobial?
Although several alcohols have been shown to be effective antimicrobials, ethyl alcohol (ethanol, alcohol), isopropyl alcohol (isopropanol, propan-2-ol) and n-propanol (in particular in Europe) are the most widely used (337). Alcohols exhibit rapid broad-spectrum antimicrobial activity against vegetative bacteria (including mycobacteria), viruses, and fungi but are not sporicidal. They are, however, known to inhibit sporulation and spore germination (545), but this effect is reversible (513). Because of the lack of sporicidal activity, alcohols are not recommended for sterilization but are widely used for both hard-surface disinfection and skin antisepsis. Lower concentrations may also be used as preservatives and to potentiate the activity of other biocides. Many alcohol products include low levels of other biocides (in particular chlorhexidine), which remain on the skin following evaporation of the alcohol, or excipients (including emollients), which decrease the evaporation time of the alcohol and can significantly increase product efficacy (68). In general, isopropyl alcohol is considered slightly more efficacious against bacteria (95) and ethyl alcohol is more potent against viruses (259); however, this is dependent on the concentrations of both the active agent and the test microorganism. For example, isopropyl alcohol has greater lipophilic properties than ethyl alcohol and is less active against hydrophilic viruses (e.g., poliovirus) (259). Generally, the antimicrobial activity of alcohols is significantly lower at concentrations below 50% and is optimal in the 60 to 90% range.
How do disinfectants work?
Disinfectants are chemical products that kill germs after a surface is cleaned. If these products claim to kill any pathogen, they must be registered with the EPA. Bleach and alcohol are common disinfectants.
What is the chemical that is used to disinfect water?
Vital Oxide is also an environmentally-friendly option that uses a chemical compound called chlorine dioxide. Chlorine dioxide is gentler than bleach and has been used to disinfect water.
How do cleaners work?
The primary goal of cleaners is to get rid of dirt and grime, and also physically remove bacteria, viruses, mold, and fungi.
What disinfectant is used for sars?
The EPA has a list of registered disinfectants for use against SARS-CoV-2, the virus that causes COVID-19. If you're concerned about the chemicals in disinfectants, there are other options available that have a lower-toxicity rating with the EPA.
How long does it take to clean a surface after disinfecting?
A surface is only clean until it comes into contact with germs again, which could be minutes or hours.
Why is it important to clean your house?
Cleaning can also remove food and water that allows germs to survive and reproduce. This can lower the number of germs and reduce the risk of spreading infection.
How to clean a building?
If you want to create a cleaning plan for your building, remember the following three steps: 1 Clean 2 Disinfect 3 Protect
