How do we perceive light?
The way we are able to perceive light is due to the photons that are flying through the air. They originate from the light sources that are very likely to be around you right now and then are reflected off the objects in the room.
How do electrons move from one orbit to another?
An electron moves from one orbit to another either by being energized or by releasing that energy. It has an orbit knows as its natural orbit that it prefers to be in, but it is fairly easy for them to be energized. Adding electrons through a en electric volt is just one way, and this is how light bulbs and LED lights work. When an electron is energized it jumps to a higher orbit, where it will then have the chance to energize other electrons in that orbit and force them to another orbit and so on.
What are the particles in an atom?
First, let's talk about the composition of an atom. These tiny particles are made out of a nucleus of protons and neutrons in their center. Around them are smaller ions called electrons that have a negative charge. These electrons are circling the nucleus in predetermined arcs that are still being studied very closely today. The arcs get bigger, of course, as the electrons move farther away from the nucleus. The electrons in an atom are constantly in motion and that not only means that they are circling the nucleus regularly, but it also implies that they are moving to and from different orbits all the time. That is the basis between forming a photon.
What does it mean when electrons are constantly moving?
The electrons in an atom are constantly in motion and that not only means that they are circling the nucleus regularly, but it also implies that they are moving to and from different orbits all the time. That is the basis between forming a photon.
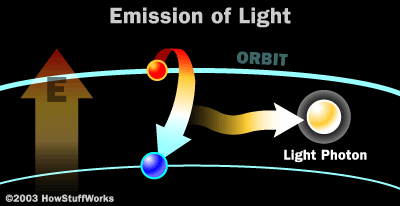
Why do electrons not stay in an orbit?
Electrons do not remain in an unnatural orbit for long, though, because they prefer to be in their own orbit. In order to get back they produce a packet of energy, which is a photon. De pending on the amount of energy released, the photon will be of different frequencies and therefore colors. Sodium atoms, for example, give off yellow photons and therefore yellow lights. Energizing atoms in a ruby crystal, however, creates a red light of a different frequency. This is how a laser is made.
How do photons work?
There are many different ways to produce photons, but all of them use the same mechanism inside an atom to do it. This mechanism involves the energizing of electro ns orbiting each atom's nucleus . How Nuclear Radiation Works describes protons, neutrons and electrons in some detail. For example, hydrogen atoms have one electron orbiting the nucleus.
What type of light emits a current through a gas?
You can see this phenomenon quite clearly in gas-discharge lamps. Fluorescent lamps, neon signs and sodium-vapor lamps are common examples of this kind of electric lighting, which passes an electric current through a gas to make the gas emit light.
How to tell if a sodium vapor light is a vapor?
You can tell a sodium vapor light because it's really yellow when you look at it. A sodium vapor light energizes sodium atoms to generate photons. A sodium atom has 11 electrons, and because of the way they're stacked in orbitals one of those electrons is most likely to accept and emit energy.
What happens to electrons when they fall from high energy to normal energy?
During the fall from high energy to normal energy, the electron emits a photon -- a packet of energy -- with very specific characteristics . The photon has a frequency, or color, that exactly matches the distance the electron falls. You can see this phenomenon quite clearly in gas-discharge lamps.
How to tell if a gas discharge light is sodium vapor?
For example, along highways and in parking lots, you often see sodium vapor lights. You can tell a sodium vapor light because it's really yellow when you look at it.
Can electrons move to higher orbitals?
There's a huge amount of theory around electron orbitals, but to understand light there is just one key fact to understand: An electron has a natural orbit that it occupies, but if you energize an atom, you can move its electrons to higher orbitals.
Who came up with the photon?
He later described his breakthrough as "an act of desperation"—an unsubstantiated trick to make the math work out. Albert Einstein , too, resisted implications of the photon theory that he helped to develop.
What is a photon in Kuo's response?
Kuo gives a circular response. "A photon is the click registered by a single-photon resolving detector," she says. Vaguer words than Kuo's have been used to describe the photon. It's a wave and a particle of light, or it's a quantization of the electromagnetic field.
Why does Landsman want to understand electrons?
Landsman, a theorist, is working to understand why electrons leave certain molecules faster than others . Some molecules, for example, confine the electron to a space such that the electron forms a standing wave. This condition, known as shape resonance, temporarily traps the electron, slowing down its escape. Ultimately, Landsman wants to elucidate all the factors that delay atoms and molecules from releasing the electron to zero in on how long the photon and electron encounter each other. "These experiments give us more insight as to how a photon interacts with an electron," she says.
What is the second quantum revolution?
Many experts have dubbed the current era of single-photon technology as the "second quantum revolution," a paradigm shift where scientists not only understand the counterintuitive principles of quantum mechanics—entanglement, superposition, and wave-particle duality—but can exploit them in technologies.
How does Minev describe the photon?
"I'm currently reevaluating my own understanding." Currently, he thinks the photon is a "quantum of action," where "action" refers to an abstract quantity describing the allowed behavior of his system.
What is the most familiar particle?
The photon might be the most familiar of elementary particles. Traveling at the speed of light, the particles bombard us daily from the sun, moon, and stars. For more than a century, scientists and engineers have harnessed them in aggregate to illuminate our cities and now, our screens.
Can you split one photon into two?
For example, she designed a crystal to double the frequency of an input photon, enabling conversion between red and infrared light. "You can fuse two photons together, or split one photon into two," she says. "Or even higher order processes. You can fuse three photons into one, or split one photon into three." Complementing these techniques, she uses state-of-the-art single-photon detectors, made of superconducting wires that become nonsuperconducting when they absorb a single photon. These types of detectors deliver highly accurate counts, detecting photons with up to 99 percent efficiency.
How do lasers work?
A laser is created when the electrons in atoms in special glasses, crystals, or gases absorb energy from an electrical current or another laser and become “excited.”. The excited electrons move from a lower-energy orbit to a higher-energy orbit ...
How do mirrors work in glass?
Mirrors at both ends of the glass amplifier cause the photons to travel back and forth through the glass, stimulating more electrons to drop to their lower energy states and emit photons. This process produces huge numbers of photons of the same wavelength and direction—an extremely bright and straight beam of light.
What type of lasers emit light?
Some lasers, such as ruby lasers, emit short pulses of light. Others, like helium–neon gas lasers or liquid dye lasers, emit light that is continuous. NIF, like the ruby laser, emits pulses of light lasting only billionths of a second. Laser light does not need to be visible.
How is laser light different from normal light?
Laser light is different from normal light in other ways as well. First, its light contains only one wavelength (one specific color). The particular wavelength of light is determined by the amount of energy released when the excited electron drops to a lower orbit. Second, laser light is directional. Whereas a laser generates a very tight beam, a flashlight produces light that is diffuse. Because laser light is coherent, it stays focused for vast distances, even to the moon and back.
How much power does a laser have?
Modern lasers can produce pulses that are billions of times more powerful. Scientists have demonstrated NIF’s ability to generate more than 500 trillion watts of power. Some lasers, such as ruby lasers, emit short pulses of light.
How long does a flash of light last in NIF?
In NIF, as in most large lasers, intense flashes of white light from giant flashlamps “pump” electrons in large slabs of laser glass to a higher-energy state that lasts only about one-millionth of a second.
How many watts does a laser have?
Early lasers could produce peak powers of some 10,000 watts. Modern lasers can produce pulses that are billions of times more powerful. Scientists have demonstrated NIF’s ability to generate more than 500 trillion watts of power.
How do photons absorb energy?
When a photon collides with another atom, the atom may absorb the photon's energy by boosting an electron to a higher level. For this to happen, the energy level of the photon has to match the energy difference between the two electron positions. If not, the photon can't shift electrons between orbitals.
How are visible light and X-rays produced?
Visible light photons and X-ray photons are both produced by the movement of electrons in atoms. Electrons occupy different energy levels, or orbitals, around an atom's nucleus. When an electron drops to a lower orbital, it needs to release some energy -- it releases the extra energy in the form of a photon.
Why do radio waves pass through things?
Radio waves don't have enough energy to move electrons between orbitals in larger atoms, so they pass through most stuff. X-ray photons also pass through most things, but for the opposite reason: They have too much energy.
How does fluoroscopy work?
In fluoroscopy, the X-rays pass through the body onto a fluorescent screen, creating a moving X-ray image. Doctors may use fluoroscopy to trace the passage of contrast media through the body. Doctors can also record the moving X-ray images on film or video.
What did Roentgen see on the X-ray screen?
Finally, he put his hand in front of the tube, and saw the silhouette of his bones projected onto the fluorescent screen. Immediately after discovering X-rays themselves, he had discovered their most beneficial application.
Which atoms absorb the most X-rays?
The soft tissue in your body is composed of smaller atoms, and so does not absorb X-ray photons particularly well. The calcium atoms that make up your bones are much larger, so they are better at absorbing X-ray photons .
What is X-ray technology?
X-ray technology lets doctors see straight through human tissue to examine broken bones, cavities and swallowed objects with extraordinary ease. Modified X-ray procedures can be used to examine softer tissue, such as the lungs, blood vessels or the intestines.

Photon Discovery
Are Photons particles?
- Strictly speaking, photons are neither particles nor waves; they are a combination of both. In some situations, their particle-like nature comes out more, and in others, their wave-like nature is more apparent. For example, a detector can register the arrival of a single photon, which appears as a point-like particle. The process known as Compton scattering involves a photon striking an elect…
Do Photons Have Mass and Momentum?
- Photons have zero mass, which allows them to travel at the fastest possible speed in the universe, the speed of light. However, they do have energy and momentum. The energy of a photon is given by Planck's constant times the frequency of the light, and the momentum of a photon is given by Planck's constant times the frequency of the light times the...
Do Photons Experience time?
- Our understanding of the rate of the passage of time comes from Einstein's theory of special relativity, which states that objects traveling closer and closer to the speed of light will experience slower and slower rates of the passage of time. In other words, moving clocks run slowly, according to John D. Horton of the University of Pittsburgh(opens in new tab). However, the mat…
Are Photons Affected by Gravity?
- Because photons have both energy and momentum, they are influenced by gravity. Under Einstein's theory of general relativity, which is our modern understanding of gravity, anything with any form of energy (including mass, momentum and torsion) is influenced by gravity. Specifically, massless particles, such as photons, follow "geodesics," which are paths of minimum distance f…
Additional Resources
- You can dig deeper into the relationship between light and time in this YouTube video(opens in new tab)hosted by the author of this article, astrophysicist Paul M. Sutter.
- For a fun exploration of the nature of quantum mechanics (which, of course, also discusses photons), check out "How to Teach Quantum Physics to Your Dog"(opens in new tab)(Scribner, 2010) by physic...
- You can dig deeper into the relationship between light and time in this YouTube video(opens in new tab)hosted by the author of this article, astrophysicist Paul M. Sutter.
- For a fun exploration of the nature of quantum mechanics (which, of course, also discusses photons), check out "How to Teach Quantum Physics to Your Dog"(opens in new tab)(Scribner, 2010) by physic...
- The Physics Asylum also hosts a great video explainer on the nature of the photon, which you can watch here(opens in new tab).
Bibliography
- Afework, B., Boechler, E., Campbell, A., Hanania, J., Heffernan, B., Jenden, J., Street, K., & Donev, J. (2021, October 22). Photon. Energy Education. https://energyeducation.ca/encyclopedia/Photon (opens in new tab) American Physical Society. (2005, January). This month in physics history: Einstein and the photoelectric effect. APS News. https://www.aps.org/publications/apsnews/20…