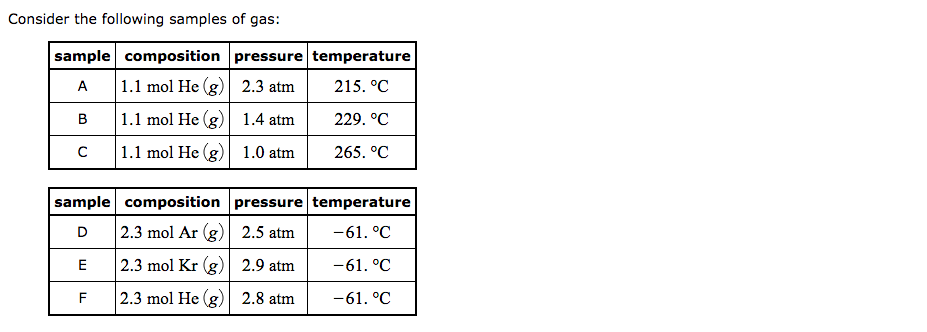
How do you find the volume of a gas sample at STP? If you have the mass of the gas , you can divide the mass by the molecular weight of the gas molecules to get the number of moles. Then multiply this by 22.4 Liters / mole to get the volume .
What is the volume of a gas at STP?
Mar 11, 2020 · How do you find the volume of a gas sample at STP? If you have the mass of the gas , you can divide the mass by the molecular weight of the gas molecules to get the number of moles. Then multiply this by 22.4 Liters / mole to get the volume .
How do you calculate volume of a mixture at STP?
Considering the gas in the question to be an ideal gas, you can find out the volume of a gas at STP following Ideal Gas Equation. Ideal gas equation is. PV = nRT. Where; P = pressure of the gas, V = volume of the gas, R = gas constant = 0.0821 L atm / (Kmol) n = no. of moles of gas present.
How do you find the volume of a gas?
Additionally, how do you find the volume of a dry gas? To obtain the gas pressure, subtract the vapor pressure of water from the total pressure. EXAMPLE: A 325 mL sample of gas is collected over water at 26oC and 742 mm Hg pressure. Calculate the volume of the dry gas at STP. The vapor pressure of water is 25 mm Hg at 25oC.
What is the volume of nitrogen gas in cm3 at STP?
Nov 30, 2020 · Solution: Given the number of moles of carbon dioxide, CO 2 = 0.4 mol. Therefore, the volume of CO 2 = number of moles of CO 2 x molar volume at STP. = 0.4 x 22.4 dm 3 = 8.96 dm 3. 2. Find the number of moles of ammonia gas contained in a sample of 60 cm3 of the gas at room conditions.
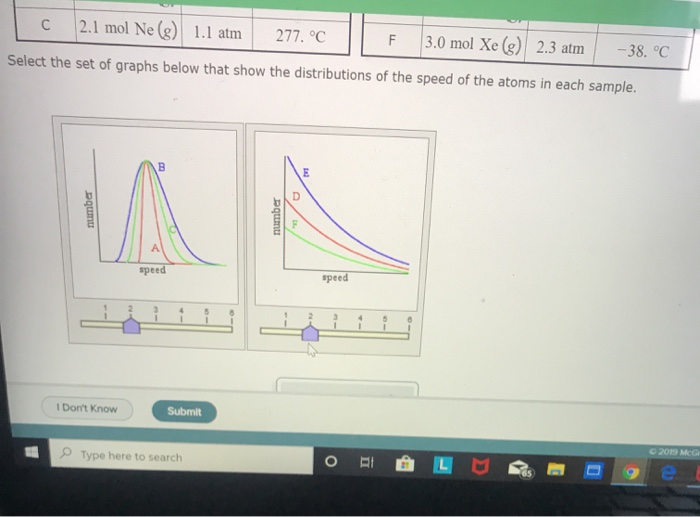
How to find the volume of a gas?
To find the exact volume of your gas, it depends on what information you have: 1 If you have the amount of gas in moles, simply multiply this amount by 22.4 Liters / mole to get the volume of the gas. For example, if you have 2 moles of N2 gas, the gas takes up 44.8 Liters. 2 If you have the mass of the gas, you can divide the mass by the molecular weight of the gas molecules to get the num
What determines the volume of a gas?
The volume of a gas depends on a number of factors. The temperature of the gas, the pressure of the gas and the amount of gas (number of moles).
How many liters does a mole of N2 take up?
For example, if you have 2 moles of N2 gas, the gas takes up 44.8 Liters. If you have the mass of the gas, you can divide the mass by the molecular weight of the gas molecules to get the num. Continue Reading. At STP, a mole of gas takes up 22.4 Liters.
How many dm3 is a mole of CO2?
1 mole of CO2 (or any gas) occupies 22.4 dm^3 at s.t.p, where 22.4 dm^3 is called molar volume of a gas at s.t.p.
What is the molar volume of an ideal gas?
Under these conditions, the molar volume of an ideal gas is: 22.711 L/mol.
Is temperature a valid assumption for gas?
This will allow you to take measurements under your current conditions and convert to Standard conditions - assuming ideal behaviour of the gas. For small changes in temperature and pressure, this is a valid assumption for most gases.
Is temperature measured in K?
Therefore temperature MUST be measured in kelvin, K but pressure and volume can be any convenient unit providing the same units are applied to both sides of the equation.
What is the standard temperature of a gas at STP?
Note: STP refers to standard temperature of 0°C and pressure of 1 atmosphere. Room conditions refer to the temperature of 25°C and the pressure of 1 atmosphere. This means that one mole of any gas occupies the same volume at STP, which is 22.4 dm 3. Under room conditions, one mole of any gas occupies 24 dm 3. ...
What is the molar volume of a gas?
Therefore, chemists introduced the concept of molar volume. Molar volume of a gas is defined as the volume of one mole of the gas. Thus, the molar volume is also the volume occupied by 6.02 x 10 23 particles of gas.
How to find moles of a gas?
It is rather tricky to find the number of moles of a gas by weighing its mass. Chemists determine the number of moles of any gas by measuring its volume. However, this cannot be done for solids and liquids. It is found that under the same temperature and pressure, equal volumes of all gases contain the same number of particles.
