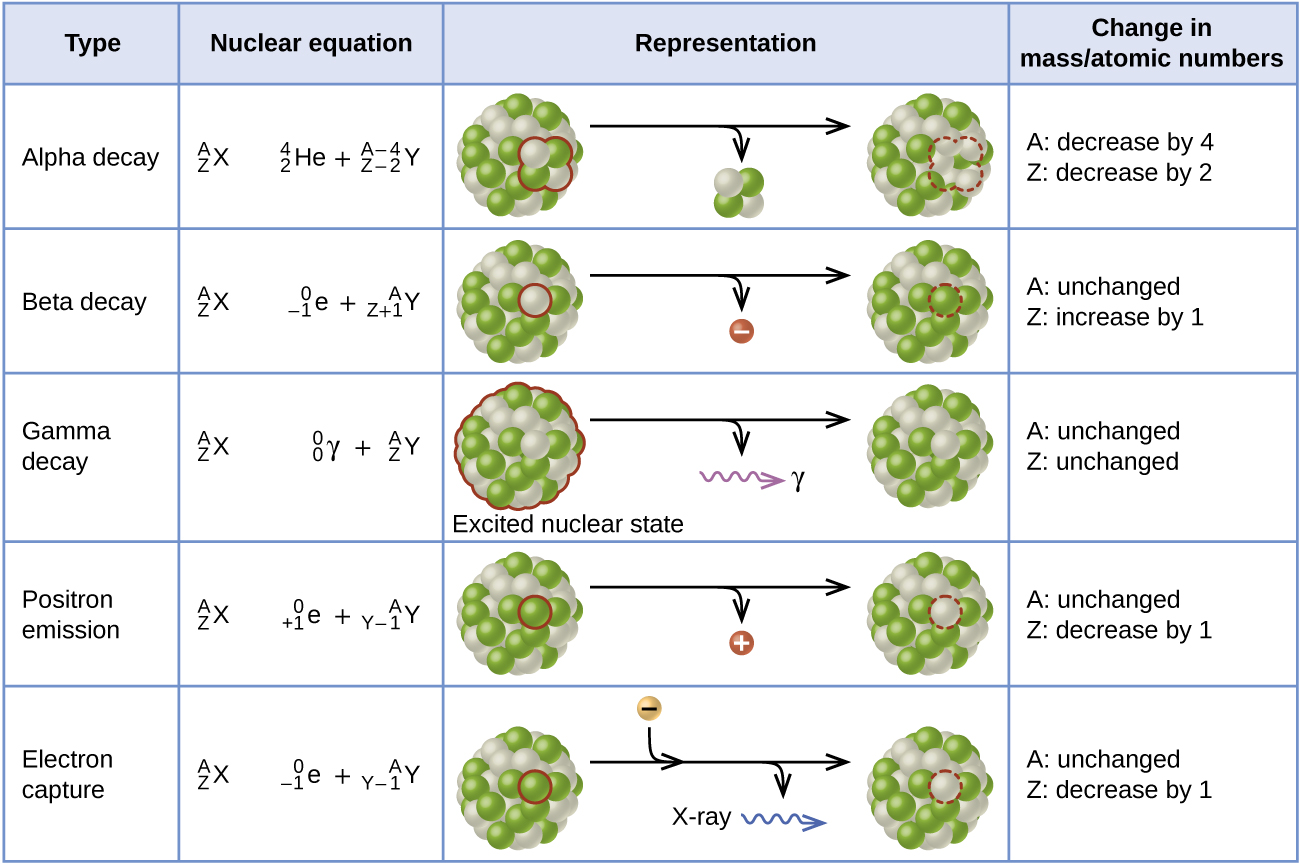
- Elements in the same row are in the same period. ...
- Elements in the same column are in the same group. ...
- Here's a close-up look at the carbon square from the Periodic Table.
What is the easiest way to learn the periodic table?
- Try and picture something for each element. For example, for hydrogen picture a star, or for uranium picture a power plant. ...
- Memorize in chunks or five, then each day add 5 elements. Do that for 23 days more or less and you have memorized 115 elements, basically the periodic table.
- The third method isn’t really a method. ...
Why do you need to learn the periodic table?
The periodic table has gone through many changes since Dmitri Mendeleev drew up its original design in 1869, yet both the first table and the modern periodic table are important for the same reason: The periodic table organizes elements according to similar properties so you can tell the characteristics of an element just by looking at its location on the table.
What are facts about the periodic table?
Fun facts about the Periodic Table
- Carbon is unique in that it is known to form up to 10 million different compounds. ...
- Francium is the rarest element on earth. ...
- The only letter not in the periodic table is the letter J.
- The country Argentina is named after the element silver (symbol Ag) which is argentum in Latin.
How do you read the periodic table protons neutrons electrons?
Vocabulary:
- Protons: Positively charged subatomic particles located in the nucleus of an atom.
- Neutrons: Neutrally charged subatomic particles located in the nucleus of an atom.
- Electrons: Negatively charged subatomic particles located in orbitals surrounding the nucleus.
How do you read the periodic table for dummies?
0:073:07The Periodic Table Explained - YouTubeYouTubeStart of suggested clipEnd of suggested clipFirst the letters are symbols. Each box represents an element each element is made up of the sameMoreFirst the letters are symbols. Each box represents an element each element is made up of the same kind of atom with a specific number of protons in its nucleus.
How is the periodic table organized and how do we read it?
The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). Elements in the same group have the same number of valence electrons. Meanwhile, elements in the same period have the same number of occupied electron shells.
How do you read the numbers of elements?
Element Boxes The atomic number, the number at the top of each element box, is the number of protons in the nucleus and the number of electrons in the atom. The elemental symbol is an abbreviation of the element name. Some periodic tables do not include the element names, only the symbols.
What is an easy way to know the periodic table?
Memorization StrategiesBreak down the table into sections. ... Spread out the memorization process. ... Learn the elements in a song. ... Make nonsense words made from element symbols. ... Use color to learn element groups. ... Use a mnemonic device to help remember the order of the elements.
What do the numbers on a periodic table mean?
In the modern periodic table, the elements are listed in order of increasing atomic number. The atomic number is the number of protons in the nucleus of an atom. The number of protons define the identity of an element (i.e., an element with 6 protons is a carbon atom, no matter how many neutrons may be present).
What do the rows on the periodic table represent?
The table has seven rows and 18 columns. Each row represents one period; the period number of an element indicates how many of its energy levels house electrons. Sodium, for instance, sits in the third period, which means a sodium atom typically has electrons in the first three energy levels.
How do you read a table?
A table can be read from left to right or from top to bottom. If you read a table across the row, you read the information from left to right. In the Cats and Dogs Table, the number of black animals is 2 + 2 = 4. You'll see that those are the numbers in the row directly to the right of the word 'Black.
How is the periodic table arranged?
The periodic table is arranged by atomic weight and valence electrons. These variables allowed Mendeleev to place each element in a certain row (called a period) and column (called a group). The table comprises seven rows and 18 columns.
What does the bottom number on the periodic table mean?
The bottom number is the proton number, it is simply the number of protons in an atom of the element.
How can I memorize Chemistry?
Memorizing Chemistry Using Repetition As you become more familiar with a word/structure/sequence, it will become easier to remember it. This is the memorization method most of us use. We copy notes, use flashcard to recall information in a new order, and draw out structures over and over again from memory.
Do you have to memorize the periodic table for chemistry?
Students don't need to memorise the periodic table. The periodic table is the chemist's alphabet, and we need to be very familiar with it. By putting the elements together in various combinations, as we do when we spell words, we can build our dictionary, containing all the substances in the universe.
Do I need to memorize the periodic table?
Memorizing the periodic table is important because it is organized and gives a lot of information about elements. Also, it makes students understand how elements relate to one another. It is used to predict the properties of every element.
How do you read protons neutrons and electrons on the periodic table?
The atomic number (number at the top) is the amount of protons and the amount of electrons. So if an element has an atomic number of 5, you know that it has 5 protons and 5 electrons. The atomic mass (number at the bottom) is the amount of protons and neutrons added together.
Why is the periodic table organized the way it is?
Answer and Explanation: The periodic table of elements is arranged the way it is by showcasing features that elements have in common with each other. From left to right, the elements increase in atomic number and mass.
What are the 7 families of the periodic table?
VocabularyGroup (family): A vertical column in the periodic table.Alkali metals: Group 1A of the periodic table.Alkaline earth metals: Group 2A of the periodic table.Halogens: Group 7A of the periodic table.Noble gases: Group 8A of the periodic table.Transition elements: Groups 3 to 12 of the periodic table.
How do you read valence electrons on the periodic table?
How do you find the valence electrons? For neutral atoms, the number of valence electrons is equal to the atom's main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons.
Why are valence electrons important?
The reason for this is that the valence electrons, which are the electrons in the outermost shell, are the ones taking part in chemical reactions. These electrons are either donating, accepting, or sharing. Moreover, the more filled the valence shell is, the more stable the element.
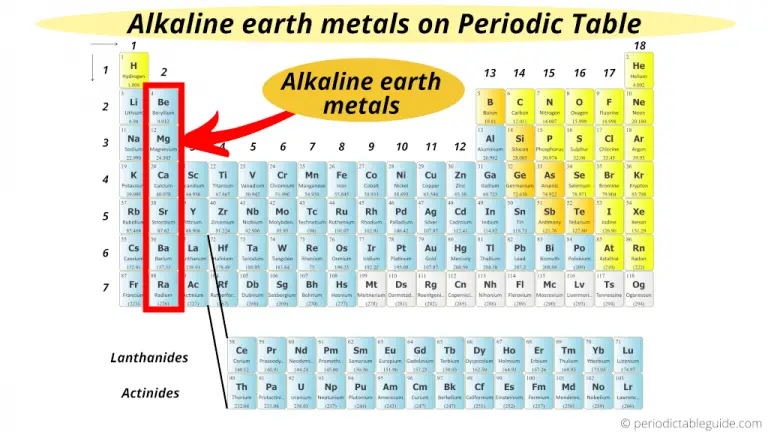
What group are alkali metals in?
The Alkali Metals (Group 1) The alkali metals consist of all of the elements in group one with the exception of hydrogen. These elements are extremely reactive and for this reason, are usually found in compounds. In addition, they are water-sensitive (they react violently with water), so they must be stored in oil.
What are noble gases?
The noble gases, also called aerogens, are inert gases. Some examples include argon, krypton, and neon. They can be found in group eighteen on the periodic table. Likewise, this means they have a complete valence shell. For this reason, they are stable and relatively unreactive.
What is an element in the periodic table?
Vocabulary. Elements: A pure substance composed of a single atom. Groups: The vertical column of the periodic table that signifies the number of valence electrons in an element. Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element. Families: Elements that have the same number ...
What is the name of the group of elements that are found in the three states of matter at standard temperature?
The name halogen means “salt formers” in greek. This is evident in nature as halogens interact with metals to form various salts. On another note, the halogens are a unique group of elements. They are the only periodic family that contains elements in the three states of matter at standard temperature. There are 6 halogens and they are located in group 17. These elements include fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). They are highly reactive, highly electronegative, and highly toxic non-metals.
Which metals are the second most reactive?
The alkaline earth metals are the second most reactive family on the periodic table (following behind the alkali metals). Moreover, they are strong reducing agents which means they donate electrons in chemical reactions. They are also good thermal and electrical conductors.
How are elements organized in the periodic table?
The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). Elements in the same group have the same number of valence electrons. Meanwhile, elements in the same period have the same number of occupied electron shells. In 1869 Russian chemist Dmitri Mendeleev noticed there existed an innate pattern of organization for the chemical elements. From this deduction, he formed the periodic table. It is important to note how the location of elements on this table tells us about their properties. A quick way to understand an element’s chemical and physical properties is to know the periodic trends. These trends tell you where the highest and lowest types of properties are concentrated on the periodic table. For a more in-depth explanation of periodic trends, click here.
What is the periodic table?
Periodic Table An arrangement of the elements by their properties and their atomic number. Element A substance that cannot be divided into simpler substances by chemical means. Symbol A one- or two-letter abbreviation used to represent an element. The first letter is capitalized. Any second letter is always lower case.
What is the atomic mass of magnesium?
It has an atomic number of 12 and an atomic mass of 24.305 amu.
Where is the atomic number of an element?
The atomic number of the element is represented by the number in the upper right-hand corner of the entry. This number represents the number of protons in the nucleus of the atom which determines the element. For this example, the atomic number is 12.
What is an element?
Element A substance that cannot be divided into simpler substances by chemical means.
Do you ever wonder what makes up the world around you?
Everything you can think of is made up of one or more elements — the chemical building blocks of the universe.
Elements
Just like when you use Google Maps to find the address of a site you want to visit, you can use a square on the periodic table to learn basic information about an element.
Groups
Groups appear as columns numbered from 1-18, from left to right at the top of the table.
Periods
Periods appear as rows numbered from 1-7, from top to bottom on the left side of the table.
Take Action
You don't need to know every element, but getting some practice with the periodic table will help you become more familiar with the elements you need to know in class.
What is precision timekeeping?
Precision timekeeping may not be something you associate with the periodic table, but for NIST researchers who build cutting-edge atomic clocks, it could be the first thing that comes to mind . Since 1967, the second has been defined by atomic clocks using cesium atoms.
Why are atomic clocks useful?
Precise time measurements are useful for time-stamping financial transactions, synchronizing communications and data, and navigating using the Global Positioning System (GPS). More recently, NIST researchers are making clocks with other atoms such as strontium, ytterbium, mercury and aluminum. The researchers change the quantum states of these atoms using optical radiation, with frequencies of hundreds of trillions of cycles per second (much higher than the microwave radiation used in cesium clocks). These “optical clocks” enable the second to be split into even smaller intervals that could be useful for things such as detecting underground geologic deposits and even dark matter.
What is the blue light on the periodic table?
A blue laser beam excites a cube-shaped cloud of strontium atoms located behind the round window in the middle of the table. Strontium atoms fluorescence strongly when excited with blue light. If you love numbers, well, of course, the periodic table is filled with them. Each atom on the table has a bevy of quantities.
What atoms are used in clocks?
More recently, NIST researchers are making clocks with other atoms such as strontium, ytterbium, mercury and aluminum. The researchers change the quantum states of these atoms using optical radiation, with frequencies of hundreds of trillions of cycles per second (much higher than the microwave radiation used in cesium clocks).
What element did Mendeleev predict?
The genius of Mendeleev was that he left spaces for elements yet to be discovered, and in so doing he predicted their existence, such as gallium in 1875 and germanium in 1886. As you may have guessed, the latter was named after Germany (the home country of discoverer Clemens Winkler). As for the former, Paul Emile Lecoq de Boisbaudran named the element “gallia,” after Gaul, the Iron Age region that includes present-day France.
How many protons does hydrogen have?
Hydrogen has one proton, so its atomic number is 1, and uranium has 92, so its atomic number is …. 92. If there are an equal number of electrons and protons, the atom is electrically neutral. If there are fewer or more electrons than protons, the atom is electrically charged and known as an ion. The official NIST periodic table.
What is the best material to use to make solar cells?
Indium nitride can absorb light over a narrower range of wavelengths, making it a great candidate for solar cells, as it absorbs the specific wavelengths of light most common from the sun. And, of course, there’s materials engineering, too.

CORE Concepts
Related Articles
Vocabulary
- Elements: A pure substance composed of a single atom.
- Groups: The vertical column of the periodic table that signifies the number of valence electrons in an element.
- Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element.
- Elements: A pure substance composed of a single atom.
- Groups: The vertical column of the periodic table that signifies the number of valence electrons in an element.
- Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element.
- Families: Elements that have the same number of valence electrons and therefore similar properties.
The Periodic Table and The Periodic Trends
- The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). Elements in the same group have the same number of valence electrons. Meanwhile, elements in the same period have the same number of occupied electron shells. In 1869 Russian chemist Dmitri Mendeleev noticed...
Periods on The Periodic Table
- So what is a period on the periodic table? Periods are the horizontal rows of the periodic table. There are seven periods total and each element in a period has the same number of atomic orbitals. The top period, which contains hydrogen and helium, has only two orbitals. As you go down the rows, the number of orbitals increases. Below is a table to help visuals the periodic nu…
Groups of The Periodic Table
- As previously mentioned, the vertical columns on the periodic table are called “groups”. There is eighteen groups on the periodic table in total, and each periodic table group contains elements with the same number of valence electrons. The number of valence electrons present dictates the properties of an element. The reason for this is that the valence electrons, which are the electro…
Families of The Periodic Table
- On the periodic table, there are familieswhich are groups of elements with similar properties. These families are alkali metals, alkaline earth metals, transition metals, post-transition metals, metalloids, halogens, noble metals, and noble gases. Many of these families belong to a single group on the periodic table. However, not all of the families overlap with periodic table groups. F…