How does an atom become a positively charged ion quizlet?
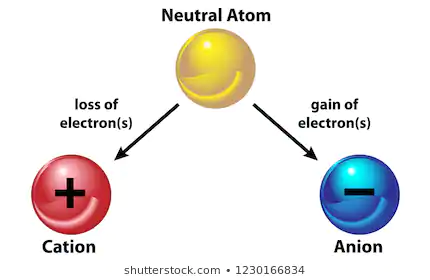
If an atom loses electrons, it loses negative charges, so it becomes a positively charged ion.
What causes an atom to gain a positive charge?
When one or more electrons is knocked off of an atom, it becomes positively charged. It is now an positive ion. A negative ion can be made by adding an electron to an atom.
What determines whether an atom becomes a positive ion or a negative ion?
If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons,it is a positive ion.
How does an atom become charged?
An entire atom becomes electrically charged when the number of electrons or protons stops being equal. The "extra" electron or proton isn't balanced by something inside the atom anymore and begins to be attracted to the protons or electrons in other atoms.
How does a neutral object become positively charged?
How does an object get positively charged? A neutral object can become positively charged when electrons get transferred to another object (see Figure 2). Figure 2. Positively charging a neutral object by transferring electrons to a positively charged object.
Which characteristic would cause an object to be positively charged?
If an object is charged, then it can be either positively or negatively charged. A positively charged object possesses more protons than electrons and a negatively charged object possesses more electrons than protons.
What does it mean when an object acquires a positive charge?
losing electronsExplanation: When an object acquires a positive charge, it is losing electrons to its surroundings. If it is losing electrons, it is losing mass (although ever so slightly).
What is the positive charge in an atom?
The positive charges equal the negative charges, so the atom has no overall charge; it is electrically neutral. Most of an atom's mass is in its nucleus; the mass of an electron is only 1/1836 the mass of the lightest nucleus, that of hydrogen....Atomic Structure.ParticleChargeMass (amu)Protons+11.00782Neutrons01.008671 more row•Aug 15, 2020