
Synthesis of acetanilide from aniline
- BACKGROUND. Acetanilide is synthesized from aniline by acetylating it with acetic anhydride in presence of glacial acetic acid.
- REQUIREMENTS
- PROCEDURE. Add 20 ml of a mixture of acetic anhydride and glacial acetic acid (equal volumes) to 10 ml (10.3 g) of aniline in a conical flask of 250 ml. ...
- CONCLUSION. ...
- REFERENCES. ...
How is acetanilide synthesized from aniline?
Acetanilide is synthesized from aniline by acetylating it with acetic anhydride in presence of glacial acetic acid. Aniline or phenylamine is a primary amine and basic in nature. Acetic anhydride, an anhydride of acetic acid, acts here as a source of acyl group.
How do you make acetanilide Cal?
Acetanilide cal also be prepared by acetylating aniline with acetic anhydride in the presence of concentrated hydrochloric acid. Dissolve aniline in hydrochloric acid and add acetic anhydride stir well. Pour the mixture to sodium acetate in water.
How do you prepare sodium acetate from aniline?
Transfer 10 ml aniline in to a 500 ml beaker, add 9 ml concentrated hydrochloric acid and 25 ml distilled water into it. Stir the contents of the beaker thoroughly with a glass rod till the whole of aniline undergoes dissolution. Dissolve 16.5 g sodium acetate in 50 ml distilled water in a separate beaker.
What is the other name of acetanilide?
N-phenylacetamide, acetanilide and acetanil are other names of this compound. It was used in the past to treat fever and headache and was known as Antifebrin by its brand name. To prepare the organic compound acetanilide from aniline, glacial acetic acid /acetic anhydride and zinc dust.
See more
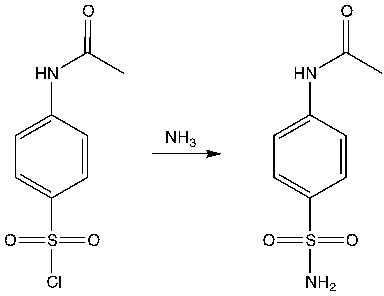
Why glacial acetic acid is used in preparation of acetanilide from aniline?
Glacial acetic acid is used because it is an excellent polar protic solvent capable of dissolving acetanilide. For the process, acetanilide is first dissolved in the solvent, glacial acetic acid, by warming. It's just a solvent to dissolve anisole (both are polar).
What is the role of zinc dust in preparation of acetanilide from aniline?
What is the need to add zinc during the preparation of acetanilide? Zinc is added to prevent the oxidation of aniline during the reaction. It reduces the coloured impurities present in the solution.
How do you make aniline acetate solution?
Dissolve 500 mg of aniline in 14 mL of water. Note that aniline is immiscible in water and two layers should be observed. Add 0.45 mL of concentrated hydrochloric acid. Measure out 0.6 mL of acetic anhydride and prepare a solution of 530 mg of sodium acetate in 3 mL of water.
How do you make an acid aniline?
(i) Take 5 mL of aniline in a 100 mL round bottom flask and add acetylating mixture containing 5 mL acetic anhydride and 5 mL glacial acetic acid. Alternatively, you can use 5 mL of acetyl chloride and 5 mL of dry pyridine as the acctylating mixture.
Why Zn dust is used?
Zinc dust is used to manufacture paints and coatings that resist corrosion when applied to structures or surfaces that are prone to harsh environmental conditions.
What is the purpose of acetanilide in the preparation?
Acetanilide is used as an inhibitor of hydrogen peroxide decomposition and is used to stabilize cellulose ester varnishes. It has also found uses in the intermediation in rubber accelerator synthesis, dyes and dye intermediate synthesis, and camphor synthesis.
Why is acetanilide used instead of aniline?
Question: Acetanilide is used instead of aniline for the nitration because aniline has an unwanted side reaction.
What is acylation of aniline?
What is aniline acetylation? A primary amine and essential in nature is aniline or phenylamine. Aniline refers to the formation of Acetanilide by nucleophilic replacement reaction with acetic anhydride and the reaction is called acetylation.
What Is type of aniline?
These include toluidines, xylidines, chloroanilines, aminobenzoic acids, nitroanilines, and many others. They often are prepared by nitration of the substituted aromatic compounds followed by reduction.
What type of reaction is the synthesis of acetanilide?
nucleophilic substitution reactionAniline reacts with acetic anhydride to form Acetanilide by nucleophilic substitution reaction and the reaction is called acetylation. In this reaction, aniline acts as the nuclepohile and acyl (CH3CO-) group from acetic anhydride acts as the electrophile.
How is aniline prepared in laboratory?
Aniline is prepared in laboratory by reducing nitrobenzene with tin (Sn) and conc. HCl. 10 ml nitrobenzene and 20 gm of granulated tin are placed in the 250 ml round bottom flask fitted with a reflux condenser.
Why is aniline a weak base?
Aniline and cyclohexylamine both hane NH2 group and six carbon ring but the difference in basicity lies in a manner that aniline is a weak base then cyclohexylamine because aniline is an aromatic ring, because of the electron-withdrawing effect of the phenyl group aniline is a weak base.
What is the role of acetic anhydride in Acetanilide from aniline reaction?
Aniline or phenylamine is a primary amine and basic in nature. Acetic anhydride, an anhydride of acetic acid, acts as a source of acyl group here . Aniline reacts with acetic anhydride to form Acetanilide by nucleophilic substitution reaction and the reaction is called acetylation.
How Acetanilide is better than aniline in aromatic electrophilic substitution reactions?
Acetanilide is less reactive towards the electrophilic substitution than Aniline, as it donates electrons to the benzene ring and to the positively polarized carbonyl group, whereas Aniline donates electrons only to the benzene ring.
Which of the following is an example for acetylating agent?
One of the most common examples of an acetylation reaction is the acetylation of salicylic acid with acetic anhydride to afford acetic acid and acetylsalicylic acid as the products.
How is chlorobenzene prepared from aniline?
It is an example of a radical-nucleophilic aromatic substitution”. In the first step aniline is going to convert to benzene diazonium chloride using sodium Nitrate and aqueous hydrochloric acid. In the second step benzene diazonium chloride reacts with cuprous chloride and forms chloro benzene.
How to make acetanilide?
The mixture is poured while hot into 100 mL of cold water. contained in a beaker with constant stirring. Filter the cooled mixture using a Buschner funnel. The slightly colored substance is dissolved in about 150 mL of boiling water, heated with 1 gram of animal charcoal (or activated charcoal) and filtered through a hot water funnel. Allow the filterate to cool. Now the precipitated acetanilide is filtered, washed with a small quantity of water and dried. The yield and the melting point determined.
How much aniline is in a flask?
About 5 ml of aniline, 6 ml glacial acetic acid and 2 grams fused sodium acetate are taken in a round bottom flask. Make sure the mouth of the flask, is fitted with a long air condenser and the mixture is gently re-fluxed, by heating the contents of the round bottom flask on a wire gauze. Continue the heating for 2 to 3 hours.
Is aniline a reflux?
Aniline is refluxed with glacial acetic acid in the presence of fused sodium acetate when acetanilide is obtained. This is the method used in the preparation of acetanilide from aniline. Write reaction here.
How is acetanilide prepared?
It is prepared by acetylation of aniline. Aim – To prepare acetanilide from aniline, glacial acetic acid (acetic anhydride) and zinc dust. Theory – On heating, aniline reacts with acetic anhydride in presence of zinc dust and glacial acetic acid and yields acetanilide. Thus, formed acetanilide is recrystallized to get pure crystals.
How to dissolve acetanilide in water?
Dissolve crude acetanilide in 50 ml distilled water using a beaker and stirrer.
How much aniline to add to a flask?
Add 20 ml aniline and 40 ml mixture of acetic anhydride and glacial acetic acid in a round bottom flask of 100 ml.
How long to heat acetanilide?
Keep heating the mixture for 30-40 min. Keep stirring the mixture. Now pour the reaction mixture from the round bottom flask into a beaker containing ice cold water. Keep stirring the mixture. Once all acetanilide is precipitated, filter the reaction mixture.
Why is zinc dust used in acetanilide?
Thus, formed acetanilide is recrystallized to get pure crystals. Purpose of using zinc dust is to prevent oxidation of aniline during the reaction. The chemical reaction involved in preparation of acetanilide is given below –. Acetanilide is also known as N-phenylacetamide, N-phenylethanamide and methanol.
What is acetanilide used for?
It is an odorless, white, solid or flaky compound. Its preparation method is used to introduce students with synthesis of organic compounds in chemistry labs. It is prepared by acetylation of aniline.
How many times can you recrystallize acetanilide?
If you want to get more pure crystals of acetanilide then you can perform the recrystallization process twice .
How is acetanilide prepared?
Acetanilide is prepared from aniline when it reacts with acetic anhydride/glacial acetic acid in the presence of zinc du st. A mixture of aniline, glacial acetic acid, acetic anhydride and zinc dust is refluxed under anhydrous condition and then poured the mixture into ice cold water to get acetic anhydride precipitate.
What is a crude sample of acetanilide?
The precipitate obtained is a crude sample of acetanilide. To get the pure crystals crystallization should be carried out.
What is acetanilide used for?
N-phenylacetamide, acetanilide and acetanil are other names of this compound. It was used in the past to treat fever and headache and was known as Antifebrin by its brand name.
What are the two acetylating agents?
Acetic anhydride and acetyl chloride are the two acetylating agents.
Which ion is obtained from the hydrolysis of the salt (sodium acetate)?
The acetate ion obtained from the hydrolysis of the salt (sodium acetate) helps to sustain the acetylation reaction in the forward direction to yield acetanilide completely.1.
Is aniline soluble in water?
The freshly redistilled aniline, is almost a colourless oily liquid which being practically insoluble in water. Therefore, before carrying out the ‘acetylation’ aniline has got to be made soluble in the aqueous medium.
Why is aniline used in a reaction?
Freshly distilled aniline should be used in order to get good results or small amount of zinc can be added in the reaction mixture. Zinc reduces the coloured impurities in the aniline and also prevents its oxidation during the reaction. Prolonged heating and use of excess of acetic anhydride should be avoided.
How long to boil a flask of water?
Place the flask on a wire-gauze placed on a tripod stand as shown in Fig. Boil the mixture for 10-15 minutes. Allow the mixture to cool. Detach the condenser and pour the liquid into about 150 ml ice-cold water contained in a beaker. During addition, stir vigorously the contents of the beaker with the help of glass-rod.
Can acetanilide be cooled?
Reaction mixture should first be cooled and then poured in ice -cold water otherwise hydrolysis of acetanilide may take place.

Theory
- Chemicals: Acetic acid/anhydride mixture – 20 ml Aniline – 10 ml Apparatus:Conical flask – 250 ml Reflux water-condenser set Buchner funnel Measuring cylinder Filter paper
Requirements
Procedure
For Recrystallization
Precautions
- On heating, aniline reacts with acetic anhydride in presence of zinc dust and glacial acetic acid and yields acetanilide. Thus, formed acetanilide is recrystallized to get pure crystals. The purpose of using zinc dust is to prevent the oxidation of aniline during the reaction. The chemical reaction involved in the preparation of acetanilide is give...
Various Acetanilide Tests
- Round bottom flask of 100 ml, wire gauze, tripod stand, burner, clamp, reflux condenser, stirrer, filter paper, beaker, pipette, electronic balance, iron stand, measuring cylinder, aniline (20 ml), acetic anhydride, and glacial acetic acid mixture (40 ml), zinc dust.
Experiment Process
- Wash all the apparatus with distilled water carefully before starting the experiment.
- Add a 20 ml aniline and 40 ml mixture of acetic anhydride and glacial acetic acid in a round bottom flask of 100 ml.
- Keep stirring the reaction mixture.
- Now add zinc dust to the flask.
Green Method
- Dissolve crude acetanilide in 50 ml distilled water using a beaker and stirrer.
- Add a bit of ethanol as well.
- Now heat this solution until it gets clear and keep stirring it as well. After this you will get a solution almost like this
Uses of Acetanilide in Daily Life
- Aniline is a carcinogenic compound so it must be handled very carefully.
- Don’t inhale the fumes of acetic anhydride.
- Always dry the crystals before taking their weight and melting point to avoid any error due to moisture.
- Keep stirring the reaction mixture during the experiment. Vigorous heating is a must when th…
- Aniline is a carcinogenic compound so it must be handled very carefully.
- Don’t inhale the fumes of acetic anhydride.
- Always dry the crystals before taking their weight and melting point to avoid any error due to moisture.
- Keep stirring the reaction mixture during the experiment. Vigorous heating is a must when the mixture is added to ice-cold water.