Temperature affects phase change by slowing down the movement in between the atoms, thus causing a change in kinetic energy, which in turn causes the atoms to undergo forms of combining or a type of disepersion. Explanation: Kinetic energy while being the reason phase changes are constant, Kinetic Energy can be caused by other means.
What causes a phase change?
Phase changes require either the addition of heat energy (melting, evaporation, and sublimation) or subtraction of heat energy (condensation and freezing). Changing the amount of heat energy usually causes a temperature change.
What is the formula for change in temperature?
What is the formula for temperature change? The quantitative relationship between heat transfer and temperature change contains all three factors: Q = mcΔT, where Q is the symbol for heat transfer, m is the mass of the substance, and ΔT is the change in temperature. The symbol c stands for specific heat and depends on the material and phase.
What are some examples of phase changes?
Types of phase transition
- A eutectic transformation, in which a two-component single-phase liquid is cooled and transforms into two solid phases. ...
- A metastable to equilibrium phase transformation. ...
- A peritectic transformation, in which a two-component single-phase solid is heated and transforms into a solid phase and a liquid phase.
What are the names of all the phase changes?
Phase Change Name: Sublimation. Phase: Solid to gas. Energy Change: Endothermic. Example: Dry ice. Phase Change Name: Deposition. Phase: Gas to solid. Energy Change: Exothermic. Example: Formation of frost. A good way to remember all of these is that opposite phase changes have opposite energy needs.
How are liquid and gas phases in equilibrium?
What happens when a liquid is cooled to even lower temperatures?
When does water evaporate and ice sublimate?
Why are the lower curves not hyperbolas?
What is the critical point of a gas?
What is the relationship between PV and NKT?
How does ice melt?
See 4 more
About this website

How is temperature change related to phase changes?
Substances can change phase — often because of a temperature change. At low temperatures, most substances are solid; as the temperature increases, they become liquid; at higher temperatures still, they become gaseous.
Does temperature change in phase change?
Since the energy involved in a phase changes is used to break bonds, there is no increase in the kinetic energies of the particles, and therefore no rise in temperature.
Why does temperature increase during a phase change?
During a phase transition the supplied energy is not used to increase the kinetic energy of the molecules, but to change the binding energies (increase in internal energy)! The amount of heat required to completely vaporize a liquid is called the heat of vaporization.
How is temperature and pressure related to phase changes?
Therefore at standard pressure as temperature increases, most substances change from solid to liquid to gas, and at standard temperature as pressure increases, most substances change from gas to liquid to solid.
How does temperature affect the phase of matter?
As the temperature of a solid, liquid or gas increases, the particles move more rapidly. As the temperature falls, the particles slow down. If a liquid is cooled sufficiently, it forms a solid. If a liquid is heated sufficiently, it forms a gas.
How does temperature affect phases of water?
But if we lower the temperature below 0 degrees Celsius, or 32 degrees Fahrenheit, water changes its phase into a solid called ice. Similarly, if we heat a volume of water above 100 degrees Celsius, or 212 degrees Fahrenheit, water changes its phase into a gas called water vapor.
How does pressure cause a phase change?
As the piston compresses the gas, the pressure increases. Once the boiling point has been reached, the gas will condense into a liquid. As the piston continues to compress the liquid, the pressure will increase until the melting point has been reached. The liquid will then freeze into a solid.
Does phase change depend on pressure?
The phase of a given substance depends on the pressure and temperature. Thus, plots of pressure versus temperature showing the phase in each region provide considerable insight into thermal properties of substances. Such a pT graph is called a phase diagram. Figure 1.12 shows the phase diagram for water.
Does pressure change during phase change?
The pressure is still constant.
Why does pressure cause phase change?
Increasing the atmospheric pressure will generally increase the melting point of most substances, therefore increasing the energy required to facilitate fusion. This is because of volume: Most solids have less volume than liquids, so melting would cause the solid to expand.
What happens to the temperature during a phase change? | Socratic
During a phase change, the temperature remains constant until the phase change is complete. During a phase change, the temperature remains the same until the phase change is complete. This is how you can tell if a phase change is occurring. The phase change diagram below illustrates this concept. Notice that during a phase change, the temperature is constant, which is represented by a straight ...
Phase Change Temperature - an overview | ScienceDirect Topics
R. Parameshwaran, S. Kalaiselvam, in Eco-Efficient Materials for Mitigating Building Cooling Needs, 2015 15.4.2 Phase change temperature and latent heat of enthalpy. In LTES applications, phase change temperature and latent heat of enthalpy are considered the two decisive parameters, which actually reveals the viability of PCMs for specific applications (either cooling or heating) in buildings.
Phase Diagrams States of Matter and Phase Changes
A phase diagram is used to show the relationship between temperature, pressure and state of matter. Before moving ahead, let us review some vocabulary and particle diagrams.
How does temperature affect phase change?
Temperature affects phase change by slowing down the movement in between the atoms, thu s causing a change in kinetic energy, which in turn causes the atoms to undergo forms of combining or a type of disepersion.
What forces atoms to move closer together?
Making a group of atoms or compounds compacts will force the atoms to move closer together thus with a lower net kinetic energy energy. Reducing temperature also works along the same lines.
Can kinetic energy be caused by pressure?
Pressure and temperature can affect many other states kinetic energy, which in turn can affect each state of matter.
What is the origin of solidification of a supercooled liquid?
The origin of solidification of a supercooled liquid of a pure substance is not in the growth of a sharp interface separating solid and liquid phases but in the finite size effect of the interface lying between stable solid and stable liquid phases as shown in Fig. 4.1.1. In other words, it can be said that the solidification of a supercooled liquid cannot progress without the formation of a mushy region in front of the solidification front. The mushy region contains both solid and liquid phases and solid could be present in it in the form of dendrites of different types. Therefore it is clear that any mathematical modelling of the solidification of a supercooled liquid should take into account solid, liquid and mushy regions, i.e., the finite-size effect of the interface. Does it mean that the classical formulation of the Stefan problem with supercooling accounted for by the Gibbs-Thomson law is not justified as there is no mushy region in the formulation? The rigorous mathematical justification of the sharp freezing front model (classical formulation) with equilibrium temperature given by ( 4.1.1) comes from the fact that the modified Gibbs-Thomson relation can be obtained if appropriate limits of the phase-field, model which considers the finite width of the interface, are taken. Some phase-field models are being discussed here with this objective.
What is supercooling in PCM?
Supercooling is the state attained by the PCM wherein even below the phase transition temperature the PCM still remains in liquid form without freezing. Further cooling of the PCM can lead to the formation of ice crystals inside the PCM, and therefore it starts to freeze completely. However, if the PCM has a high degree of supercooling, it is a direct measure for higher energy consumption during the cooling process. That is, during the charging process (nighttime), most of the cooling energy would be consumed for initiating the freezing process to take place in the PCM. It is therefore a prime task to reduce the supercooling effects in the PCM.
What is a PCM?
PCMs are mainly characterized by a suitable phase change temperature and enthalpy for applications. Further PCM requirements are listed in Section 1.1. As low-temperature PCMs for cooling of buildings water/ice, slurries using a solution of water–salt (brine) and water–glycol are utilized.
What are the two decisive parameters of a PCM?
In LTES applications, phase change temperature and latent heat of enthalpy are considered the two decisive parameters, which actually reveals the viability of PCMs for specific applications (either cooling or heating) in buildings. It was also mentioned that water as a PCM possesses better latent heat enthalpy (latent heat of fusion) than organic and salt hydrate PCMs. This is because water, when utilized as a PCM, may undergo incongruent phase transition and supercooling, which limits its application for reducing building cooling requirements. Besides, for the same cooling load requirements, the TES systems utilizing water or ice require huge storage tanks compared to the PCM-based TES systems.
What are graphite nanosheets made of?
Based on the scanning electron microscope image ( Figure 15.14a ), it can be visualized that the graphite interconnected nanosheets are comprised of mesopores and macropores, which indicate their high liquid sorption capacity. The graphite nanosheets have a surface area of ~ 100 m 2 /g, which permits higher absorbability. In Figure 15.14b, it can be seen that the CA–PA PCM gets filled in the pores of the graphite interconnected nanosheets.
What is the temperature of a NEPCM?
It is thus suggested that the NEPCM, which exhibits phase transition temperature (freezing: − 3.43 °C; melting: 4.04 °C) and latent heat of enthalpy (freezing: 91.27 J/g; melting: 98.71 J/g), can be a suitable candidate for cold TES applications in buildings. The nanocapsules that are subjected to more than 40 rounds of the thermal cycles (freezing-melting) show no delamination and are complete and stable.
How many PCMs are there?
There are about 500 PCMs known, which differ from one another in their phase-change temperature ranges and their latent heat storage capacities. Common PCMs are paraffins, salt hydrates, organic materials, and eutectics.
Why does tea spread out?
The tea spreads out from the teabag when we immerse it in hot water in the same way the molecules that are restricted to a particular area during the phase will try every possible way to spread themselves. This is known as escaping tendency to understand the fundamentals of equilibrium and transformations for chemical reactions, and one needs to be familiar with these phase changes.
What is the equilibrium value of a substance?
The equilibrium value depends on the substance that you are using and the temperature around it. The other name of this property is equilibrium vapor pressure.
What happens when a matter changes its shape?
There’s a transfer of heat and energy when a matter changes its shape. If it goes from solid to liquid, the heat and energy are being released. Adding and removing the heat from a given matter can lead to the phase change of a given matter.
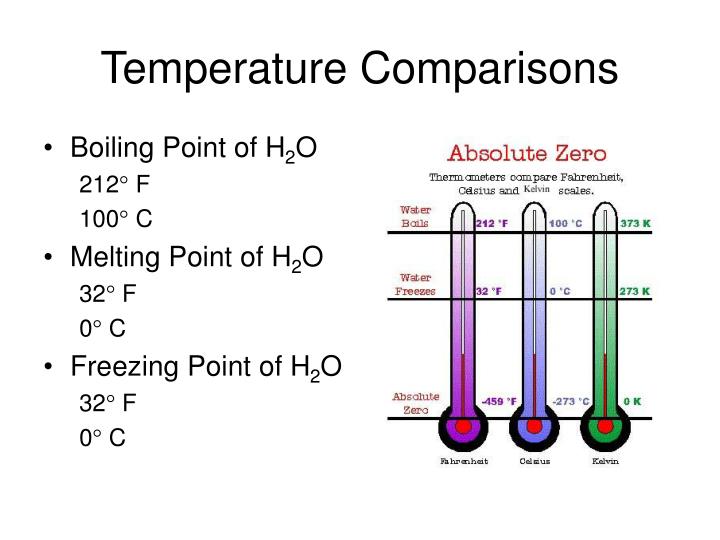
What is the boiling point of water?
The boiling point of water is 100 degrees Celcius, and the freezing point of water is 0 degrees celsius.
When a matter changes from liquid to solid, when you put water in the freezer, and it converts to answer?
When a matter changes from liquid to solid, when you put water in the freezer, and it converts into ice.
When a matter changes its state from solid to liquid, what happens?
When a matter changes its state from solid to liquid, for example, ice cubes, when taken out from the freezer, start to melt.
Can gas particles be held together?
Gas particles are not held together in any way possible, as soon as you give them a little bit of space they expand in that region. Thus, they move around a lot and can quickly fill an entire area in no time.
What is the energy required to melt all the ice?
The energy required to melt all the ice is simply the integral of the heating rate over time. That goes on the left side, because it's a source. The change that we're observing is the melting of the ice into liquid water. For this first part, there is no temperature change, just a phase change.
How to solve energy problems?
To solve energy problems you can generally follow these steps: Identify the energy source and write it on the left-hand side of the equation. Identify all the changes in temperature and in phase and put them on the right-hand side. You should know all of the variables in the equation except one.
How many times can you take the quiz 3-3?
When you feel you are ready, take Quiz 3-3. You will be allowed to take this quiz only once. Good luck!
How much energy is needed to make ice into liquid water?
The energy required is about three million joules.
How does energy come from cloud drop?
It can come from some external source, such as the sun, from chemical reactions, or from the air, which loses some energy and thus cools. Thus, temperature changes and phase changes are related, although we can think of phase changes as occurring at a constant temperature. The energy associated with phase changes drives much of our weather, especially our severe weather, such as hurricanes and deep convection. We can quantify the temperature changes that result from phase changes if we have a little information on the mass of the air and the mass and phases of the water.
What is phase change?
So to figure out the energy that must be added or removed to cause a phase change, we only need to know what the phase change is (melting/freezing, sublimating/depositing, evaporating/condensing) and the mass of water that is changing phase.
Which side of the equation should the variable of interest be on?
You should know all of the variables in the equation except one. Rewrite the equation so that the variable of interest is on the left-hand side and all the rest are on the right-hand side.
Why Do Phase Changes Occur?
Phase changes typically occur when the temperature or pressure of a system is altered. When temperature or pressure increases, molecules interact more with each other. When pressure increases or temperature decreases, it's easier for atoms and molecules to settle into a more rigid structure. When pressure is released, it's easier for particles to move away from each other.
How do liquids form?
Liquids form by condensation of gases and melting of solids. Gases: Gases can ionize into plasma, condense into liquids, or undergo deposition into solids. Gases form from the sublimation of solids, vaporization of liquids, and recombination of plasma. Plasma: Plasma can recombine to form a gas.
How do states of matter change?
Another way to list phase changes is by states of matter: Solids: Solids can melt into liquids or sublime into gases. Solids form by deposition from gases or freezing of liquids. Liquids: Liquids can vaporize into gases or freeze into solids.
What is sublimation in chemistry?
Sublimation is the transition from a solid phase to a gas phase without passing through an intermediate liquid phase. Another example is when ice directly transitions into water vapor on a cold, windy winter day.
What is the white vapor that is observed in sublimation?
For example, if you view the sublimation of dry ice into carbon dioxide gas, the white vapor that is observed is mostly water that is condensing from water vapor in the air into fog droplets.
What is the opposite of evaporation?
This photo displays the process of condensation of water vapor into dew drops. Condensation, the opposite of evaporation, is the change in the state of matter from the gas phase to the liquid phase.
What is silver deposition?
Deposition is the settling of particles or sediment onto a surface. The particles may originate from a vapor, solution, suspension, or mixture. Deposition also refers to the phase change from gas to solid.
How are liquid and gas phases in equilibrium?
Liquid and gas phases are in equilibrium at the boiling temperature. (See Figure 4.) If a substance is in a closed container at the boiling point, then the liquid is boiling and the gas is condensing at the same rate without net change in their relative amount. Molecules in the liquid escape as a gas at the same rate at which gas molecules stick to the liquid, or form droplets and become part of the liquid phase. The combination of temperature and pressure has to be “just right”; if the temperature and pressure are increased, equilibrium is maintained by the same increase of boiling and condensation rates.
What happens when a liquid is cooled to even lower temperatures?
The substance changes from a gas to a liquid. When a liquid is cooled to even lower temperatures, it becomes a solid. The volume never reaches zero because of the finite volume of the molecules. Figure 1.
When does water evaporate and ice sublimate?
Thus water evaporates and ice sublimates when their vapor pressures exceed the partial pressure of water vapor in the surrounding mixture of gases. If their vapor pressures are less than the partial pressure of water vapor in the surrounding gas, liquid droplets or ice crystals (frost) form.
Why are the lower curves not hyperbolas?
The lower curves are not hyperbolas, because the gas is no longer an ideal gas. (b) An expanded portion of the diagram for low temperatures, where the phase can change from a gas to a liquid. The term “vapor” refers to the gas phase when it exists at a temperature below the boiling temperature. Table 1.
What is the critical point of a gas?
There is a critical point —that is, a critical temperature —above which liquid cannot exist. At sufficiently high pressure above the critical point, the gas will have the density of a liquid but will not condense. Carbon dioxide, for example, cannot be liquefied at a temperature above 31.0ºC.
What is the relationship between PV and NKT?
That is, PV = NkT (ideal gas). Now, assuming the number of molecules and the temperature are fixed, PV = constant (ideal gas, constant temperature). For example, the volume of the gas will decrease as the pressure increases. If you plot the relationship PV = constant on a PV diagram, you find a hyperbola.
How does ice melt?
Ice melts from pressure in the hands of a snowball maker. From the phase diagram, we can also say that the melting temperature of ice rises with increased pressure. When a car is driven over snow, the increased pressure from the tires melts the snowflakes; afterwards the water refreezes and forms an ice layer.
