
Full Answer
What is the atomic theory?
Atomic theory is the scientific theory that matter is composed of particles called atoms. Atomic theory traces its origins to an ancient philosophical tradition known as atomism. According to this idea, if one were to take a lump of matter and cut it into ever smaller pieces, one would reach a point where the pieces could not be further cut ...
What is the current theoretical model of the atom?
The current theoretical model of the atom involves a dense nucleus surrounded by a probabilistic "cloud" of electrons Atomic theory is the scientific theory that matter is composed of particles called atoms. Atomic theory traces its origins to an ancient philosophical tradition known as atomism.
What is the modern view of the atom?
For a history of the study of how atoms combine to form molecules, see history of molecular theory. For the modern view of the atom which developed from atomic theory, see atomic physics. The current theoretical model of the atom involves a dense nucleus surrounded by a probabilistic "cloud" of electrons
Is Dalton's atomic theory still used today?
Despite these caveats, Dalton's atomic theory is still mostly true, and it forms the framework of modern chemistry. Scientists have even developed the technology to see the world on an atomic level! Dalton's atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties.

What atomic theory is accepted today?
Dalton's atomic theory was accepted by many scientists almost immediately. Most of it is still accepted today. However, scientists now know that atoms are not the smallest particles of matter. Atoms consist of several types of smaller particles, including protons, neutrons, and electrons.
Is the atomic theory still valid today?
Dalton's atomic theory. Democritus first suggested the existence of the atom but it took almost two millennia before the atom was placed on a solid foothold as a fundamental chemical object by John Dalton (1766-1844). Although two centuries old, Dalton's atomic theory remains valid in modern chemical thought.
How is your accepted atomic theory today different from Dalton's theory?
According to Dalton's atomic theory, atoms are indivisible particles. Modern atomic theory says that atoms are composed of subatomic particles; protons, electrons, and neutrons. Dalton's theory does not explain details about isotopes. It states that all atoms of the same element are identical.
Is Dalton's atomic theory still fully accepted?
Despite these caveats, Dalton's atomic theory is still mostly true, and it forms the framework of modern chemistry. Scientists have even developed the technology to see the world on an atomic level!
How has the atomic theory changed over time?
This atomic model has changed over time. Scientists used the model to make predictions. Sometimes the results of their experiments were a surprise and they did not fit with the existing model. Scientists changed the model so that it could explain the new evidence.
What is the modern view of atom?
Our modern view of the atom is that it consists of a positively charged nucleus surrounded by electrons. The nucleus contains two types of subatomic particles: protons, which are positively charged; and neutrons, which are not charged.
Which part of Dalton's atomic theory is not accepted today?
Drawbacks of Dalton's Atomic Theory The indivisibility of an atom was proved wrong: an atom can be further subdivided into protons, neutrons and electrons. However an atom is the smallest particle that takes part in chemical reactions. According to Dalton, the atoms of same element are similar in all respects.
How is Dalton's atomic theory modified in the light of modern knowledge?
On the basis of new experimental facts, Dalton's atomic theory has been modified as: 1. Atom is no longer indivisible. Atom is composed of still smaller particles which are called electrons, protons and neutrons.
What happens if you destroy an atom?
1:582:29Why atoms can neither be created nor destroyed? - YouTubeYouTubeStart of suggested clipEnd of suggested clipInto energy in nuclear fusion reaction. But mass or atom cannot be destroyed therefore remember thatMoreInto energy in nuclear fusion reaction. But mass or atom cannot be destroyed therefore remember that we can create atoms and nuclear fission reaction are in nuclear fusion reaction.
Why can atoms not be destroyed?
2. Atoms cannot be created nor destroyed, and they are indestructible; they cannot be broken into smaller parts. This was based on the Law of Conservation of Mass. It was later learned that atoms can break into smaller parts.
What are limitations of Dalton's atomic theory?
The main drawbacks of Dalton's Atomic Theory are: (i) It could explain the laws of chemical combination by mass but failed to explain the law of gaseous volumes. (ii) It could not explain why atoms of different elements have different masses, sizes, valencies, etc.
Is atomic theory proven?
Dalton's theory has not proven to be correct under all circumstances. The first rule was proven incorrect when scientists divided atoms in a process called nuclear fission. The second rule was proven incorrect by the discovery that not all atoms of the same element have the same mass; there are different isotopes.
How has atomic theory helped society?
Atomic theory established that all matter is made of tiny particles, a discovery that led to amazing scientific breakthroughs in areas from modern chemistry to nuclear energy.
How important is an atom in our life?
Atoms are extremely important structures that make up all of the materials on earth. Atoms are in our bodies and they bond together to form molecules, which make up matter. What is Matter? Matter is any substance that takes up space, meaning it has mass and volume.
Why Democritus is wrong?
2,500 years ago, Democritus suggested that all matter in the universe was made up of tiny, indivisible, solid objects he called "atomos." However, other Greek philosophers disliked Democritus' "atomos" theory because they felt it was illogical.
What was the first complete attempt to describe all matter in terms of atoms and their properties?
Dalton's atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties.
Which part of the theory states that all matter is made of atoms, which are indivisible?
The first part of his theory states that all matter is made of atoms, which are indivisible.
What have we learned since Dalton proposed his theory?
The short answer: a lot! For instance, we now know that atoms are not indivisible —as stated in part one—because they are made up of protons, neutrons, and electrons. The modern picture of an atom is very different from Dalton's "solid, massy" particle. In fact, experiments by Ernest Rutherford, Hans Geiger, and Ernest Marsden showed that atoms are mostly made up of empty space.
What did Dalton say about gold?
Dalton proposed that every single atom of an element, such as gold, is the same as every other atom of that element. He also noted that the atoms of one element differ from the atoms of all other elements. Today, we still know this to be mostly true. A sodium atom is different from a carbon atom. Elements may share some similar boiling points, melting points, and electronegativities, but no two elements have the same exact set of properties.
What did Dalton think of the law of conservation of mass?
He proposed that all matter is made of tiny indivisible particles called atoms, which he imagined as "solid, massy, hard, impenetrable, movable particle (s)".
Why did Dalton's theory have to be modified?
Part two of Dalton's theory had to be modified after mass spectrometry experiments demonstrated that atoms of the same element can have different masses because the number of neutrons can vary for different isotopes of the same element. For more on isotopes, you can watch this video on atomic number, mass number, and isotopes.
What is molecular modeling kit?
A basic molecular modeling kit, including spherical atoms of different size and color that can be connected by sticks to represent chemical bonds. Image credit: "Photo of modeling kit" by Sonia on Wikimedia Commons, CC-BY 3.0
What is the chemical atomic theory?
For this reason, the chemical atomic theory of the 19th century supposed that each identified chemical element has its specific atoms. And that each chemical compound has its molecules (fixed combinations of atoms). What particles act as unchanged and undivided units depends upon what kind of process is involved.
What is the origin of atom theory?
Atomic theory traces its origins to an ancient philosophical tradition known as atomism. According to this idea, if one were to take a lump of matter and cut it into ever smaller pieces, one would eventually reach a point where the pieces could not be further cut into anything smaller. Ancient Greek philosophers called these hypothetical ultimate particles of matter atoms, a word which meant “uncut.”
What is an atom?
An atom is now defined as the basic particle that composes a chemical element. Around the turn of the 20th century, physicists discovered that the particles that chemists called “atoms” are, in fact, agglomerations of even smaller particles (subatomic particles), but scientists kept the name out of the convention. The term elementary particle is now used to refer to actually indivisible particles.
What is the theory of chemistry?
Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms.
How do molecules lose their identity?
Some phenomena, such as evaporation, are explained by a process in which the molecules remain unchanged and identical. In chemical reactions the molecules lose their identity. Their structures are broken up, and the composing atoms, while retaining their own identity, are rearranged into new molecules. With nuclear reactions, a new level is reached.
Where did the idea of matter come from?
The idea that matter is made up of discrete units is a very old idea, appearing in many ancient cultures such as Greece and India. The word “atom,” meaning “uncuttable,” was coined by the Pre-Socratic Greek philosophers Leucippus and his pupil Democritus (c.460–c.370 BC).
Is matter indivisible?
The atoms themselves are no longer considered indivisible: more elementary particles than the atoms appear in the explanations of nuclear reactions. Atomic theory is the scientific theory that matter is composed of particles called atoms.
What is the modern view of the atom?
For the modern view of the atom which developed from atomic theory, see atomic physics. Atomic theory is the scientific theory that matter is composed of particles called atoms. Atomic theory traces its origins to an ancient philosophical tradition known as atomism. According to this idea, if one were to take a lump of matter ...
Who wrote the Atomic Theory?
Charles Adolphe Wurtz (1881) The Atomic Theory, D. Appleton and Company, New York.
How did Dalton calculate the atomic weight of a molecule?
Dalton estimated the atomic weights according to the mass ratios in which they combined, with the hydrogen atom taken as unity. However, Dalton did not conceive that with some elements atoms exist in molecules—e.g. pure oxygen exists as O 2. He also mistakenly believed that the simplest compound between any two elements is always one atom of each (so he thought water was HO, not H 2 O). This, in addition to the crudity of his equipment, flawed his results. For instance, in 1803 he believed that oxygen atoms were 5.5 times heavier than hydrogen atoms, because in water he measured 5.5 grams of oxygen for every 1 gram of hydrogen and believed the formula for water was HO. Adopting better data, in 1806 he concluded that the atomic weight of oxygen must actually be 7 rather than 5.5, and he retained this weight for the rest of his life. Others at this time had already concluded that the oxygen atom must weigh 8 relative to hydrogen equals 1, if one assumes Dalton's formula for the water molecule (HO), or 16 if one assumes the modern water formula (H 2 O).
What did Dalton say about atoms?
Dalton proposed that each chemical element is composed of atoms of a single, unique type, and though they cannot be altered or destroyed by chemical means, they can combine to form more complex structures ( chemical compounds ). This marked the first truly scientific theory of the atom, since Dalton reached his conclusions by experimentation and examination of the results in an empirical fashion.
Why did Rutherford and Marsden have doubts about the Thomson model?
Ernest Rutherford and his colleagues Hans Geiger and Ernest Marsden came to have doubts about the Thomson model after they encountered difficulties when they tried to build an instrument to measure the charge-to-mass ratio of alpha particles ( these are positively-charged particles emitted by certain radioactive substances such as radium ). The alpha particles were being scattered by the air in the detection chamber, which made the measurements unreliable. Thomson had encountered a similar problem in his work on cathode rays, which he solved by creating a near-perfect vacuum in his instruments. Rutherford didn't think he'd run into this same problem because alpha particles are much heavier than electrons. According to Thomson's model of the atom, the positive charge in the atom is not concentrated enough to produce an electric field strong enough to deflect an alpha particle, and the electrons are so lightweight they should be pushed aside effortlessly by the much heavier alpha particles. Yet there was scattering, so Rutherford and his colleagues decided to investigate this scattering carefully.
How many electrons are in each neon atom?
Each orbital holds up to two electrons, which most probably exist in the zones represented by the colored bubbles. Each electron is equally present in both orbital zones, shown here by color only to highlight the different wave phase.
What was Dalton's theory of atomic weights?
Dalton did not indicate how he obtained the relative weights, but he initially hypothesized that variation in solubility was due to differences in mass and complexity of the gas particles – an idea that he abandoned by the time the paper was finally published in 1805. Over the years, several historians have attributed the development of Dalton’s atomic theory to his study of gaseous solubility, but a recent study of his laboratory notebook entries concludes he developed the chemical atomic theory in 1803 to reconcile Cavendish’s and Lavoisier’s analytical data on the composition of nitric acid, not to explain the solubility of gases in water.
What is the atomic theory?
Atomic theory states that all matter consist of dicreet units known as atoms. The path to atomic theory was complex and full of many surprising findings. The atomic theory is today quite clear and proven. Periodic Table
What is the purpose of the Atomic Theory website?
Main purpose of this website is to help the public to learn some interesting and important information about radiation and dosimeters.
How many elements are there in the universe?
To date, 105 different elements have been confirmed to exist. Of the 105 confirmed elements, 90 exist in nature and 15 are man-made.
Where did the word "atom" come from?
The word “atom” was coined by the ancient Greek philosophers Leucippus, Epicurus and Democritus. They supposed that the properties of materials were determined by the different shapes and forms of their atoms. This word comes from the Ancient Greek adjective atomos, meaning “ indivisible “.
When did the modem idea start?
Modem ideas about the structure of matter originated in the 17th century. In 1661 the English chemist Robert Boyle laid down the modern criterion of an element published the modern criterion for an element:
What are the two parts of matter?
The atoms consist of two parts. An atomic nucleus and an electron cloud. The electrons are spinning around the atomic nucleus. The nucleus itself is generally made of protons and neutrons.
What is the lowest level orbital?
The lowest level orbitals are nice simple spheres. At higher levels they take on interesting shapes which determine the geometry and strength of chemical bonds between atoms. Except for the Hydrogen Atom, these have no analytical solution. We can make very good numerical approximations.
Do electrons behave like planets?
But we know that electrons don't really behave like planets orbiting a central star . We can only describe such particles by saying where they will probably be most of the time. These probabilities can be visualized as clouds of electron density which are often referred to as orbitals. The lowest level orbitals are nice simple spheres. At higher levels they take on interesting shapes which determine the geometry and strength of chemical bonds between atoms.
What does Rutherford's discovery indicate about the positive charge in an atom?
This indicates that the positive charge in an atom is not evenly distributed. Since only a small amount of alpha particles were being reflected, the positively charged particles’ volume is minimal compared to the total volume of an atom, which cannot be explained using the previous Thompson’s atomic model.
Who proposed the atomic model of the solid sphere?
John Dalton proposed the solid sphere atomic model based on his prior findings of the conservation of mass and constant composition law during chemical reactions in 1812. As the name suggests, atoms were described as tiny solid spheres that cannot be subdivided, destroyed, or altered. Dalton suggests that atoms of the same element have identical properties and mass and cannot be changed.
What does JJ Thompson mean by atoms?
Thompson knew that atoms have an overall neutral charge, which means there needs to be another type of positively charged particles to balance the charge. JJ Thompson proposed that atoms are unified spheres of positive charges with negatively charged electrons embedded inside them, just like a plum pudding, where electrons are plums inside the pudding. (“2.2 Evolution of Atomic Theory — Chemistry | OpenStax”). The image below illustrates this model.
Which scientist proposed that electrons can only circulate in fixed orbits?
Niels Bohr implemented quantum theory to Rutherford’s planetary model. In Bohr’s atomic model, he proposed that electrons can only circulate the nucleus in certain fixed orbits called stationary orbits where no energy is being radiated from the electron. A diagram of Bohr’s atomic model is shown below.
What happens when electrons are put together with mercury?
According to a lab research paper (ENERGY STATES OF ATOMS — THE FRANCK-HERTZ EXPERIMENT), in this experiment, scientists James Franck and Gustav Hertz found that when energetic electrons were put together with Mercury, Mercury can only absorb the inelastic collision’s energy at specific discrete values. This experiment supports the theory of fixed orbits with quantized energy levels for electrons in an atom.
How do electrons gain energy?
In Bohr’s model, electrons’ only way to gain or lose energy is by jumping to another allowed orbit marked by receiving or releasing electromagnetic waves with specific discrete wavelengths. The Franck-Hertz experiment supported Bohr’s model.
Who was the first person to study quantum mechanics?
At that time, quantum mechanics was first studied and discovered as Physicist Max Planck proposed quantum theory by studying blackbody radiation (“Quantum Physics”).
What does the Bohr model say about the structure of an atom?
The structure of the atom is now a completely solved problem - so much so that the same model can be applied to describe the behaviour of molecules, rather than just single atoms, completely, as well as the behaviour of solid materials.
Where is the nucleus located in an atom?
The nucleus is at the centre of the atom and contains the protons and neutrons. Protons and neutrons are collectively known as nucleons. Virtually all the mass of the atom is concentrated in the nucleus, because the electrons weigh so little. Working out the numbers of protons and neutrons.
How many neutrons are in an atom?
If there are 9 protons, there must be 10 neutrons for the total to add up to 19. The atomic number is tied to the position of the element in the Periodic Table and therefore the number of protons defines what sort of element you are talking about. So if an atom has 8 protons (atomic number = 8), it must be oxygen.
How many electrons does an oxygen atom have?
So, if an oxygen atom (atomic number = 8) has 8 protons, it must also have 8 electrons; if a chlorine atom (atomic number = 17) has 17 protons, it must also have 17 electrons. The arrangement of the electrons. The electrons are found at considerable distances from the nucleus in a series of levels called energy levels.
What is the first quantum number?
The first, most important quantum number is n, the aptly-named principal quantum number. This quantum number determines the energy of the state - however, as you'll quickly see, there's a lot more to a state than just its energy. The second quantum number is ℓ, the azimuthal quantum number.
Which direction do electrons deflect?
Protons are deflected on a curved path towards the negative plate. Electrons are deflected on a curved path towards the positive plate. The amount of deflection is exactly the same in the electron beam as the proton beam if the energies are the same - but, of course, it is in the opposite direction.
How many electrons are in the first level of the nucleus?
Each energy level can only hold a certain number of electrons. The first level (nearest the nucleus) will only hold 2 electrons, the second holds 8, and the third also seems to be full when it has 8 electrons.
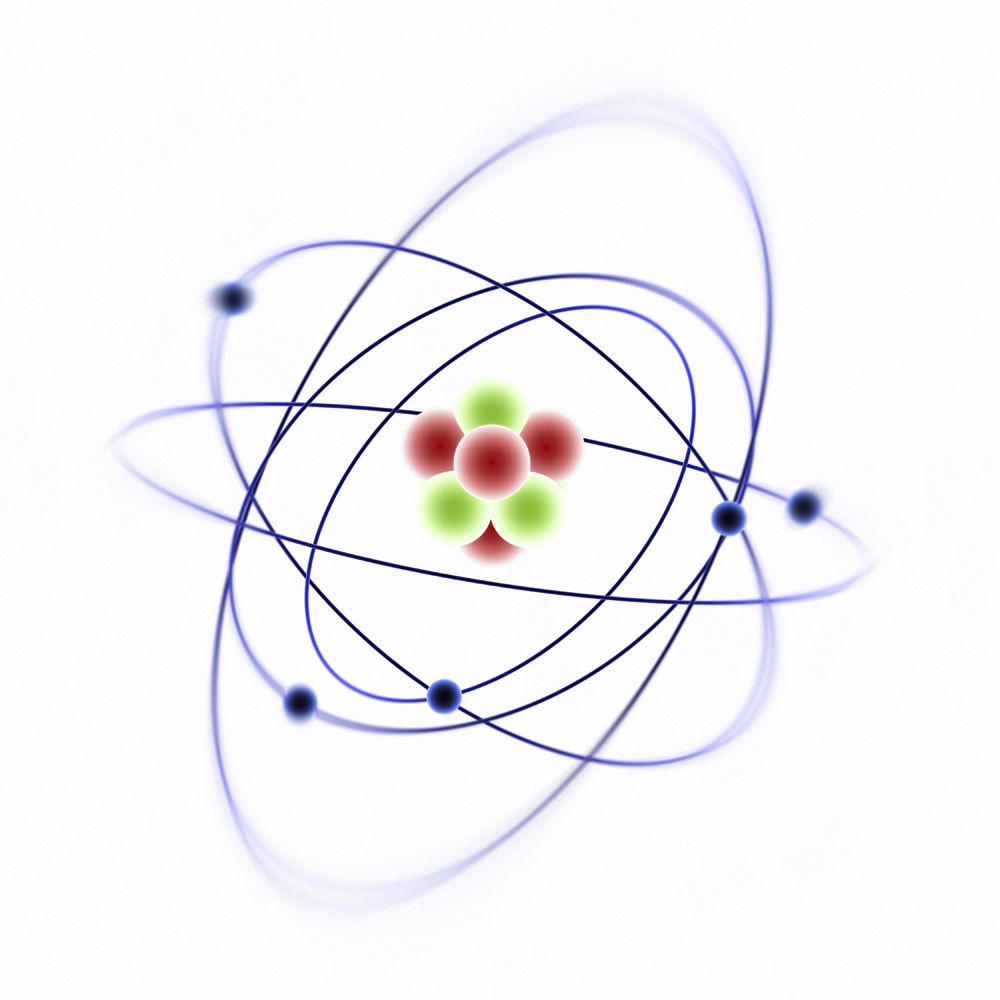
Overview
History
The idea that matter is made up of discrete units is a very old idea, appearing in many ancient cultures such as Greece and India. The word "atom" (Greek: ἄτομος; atomos), meaning "uncuttable", was coined by the Pre-Socratic Greek philosophers Leucippus and his pupil Democritus (c.460–c.370 BC). Democritus taught that atoms were infinite in number, uncreated, and eternal, and that the qualities of an object result from the kind of atoms that compose it. Democritus's at…
See also
• Spectroscopy
• History of molecular theory
• Timeline of chemical element discoveries
• Introduction to quantum mechanics
Footnotes
1. ^ Pullman, Bernard (1998). The Atom in the History of Human Thought. Oxford, England: Oxford University Press. pp. 31–33. ISBN 978-0-19-515040-7.
2. ^ Kenny, Anthony (2004). Ancient Philosophy. A New History of Western Philosophy. Vol. 1. Oxford, England: Oxford University Press. pp. 26–28. ISBN 0-19-875273-3.
Bibliography
• Andrew G. van Melsen (1960) [First published 1952]. From Atomos to Atom: The History of the Concept Atom. Translated by Henry J. Koren. Dover Publications. ISBN 0-486-49584-1.
• J. P. Millington (1906). John Dalton. J. M. Dent & Co. (London); E. P. Dutton & Co. (New York).
• Jaume Navarro (2012). A History of the Electron: J. J. and G. P. Thomson. Cambridge University Press. ISBN 978-1-107-00522-8.
Further reading
• Bernard Pullman (1998) The Atom in the History of Human Thought, trans. by Axel Reisinger. Oxford Univ. Press.
• Eric Scerri (2007) The Periodic Table, Its Story and Its Significance, Oxford University Press, New York.
• Charles Adolphe Wurtz (1881) The Atomic Theory, D. Appleton and Company, New York.
External links
• Atomism by S. Mark Cohen.
• Atomic Theory - detailed information on atomic theory with respect to electrons and electricity.