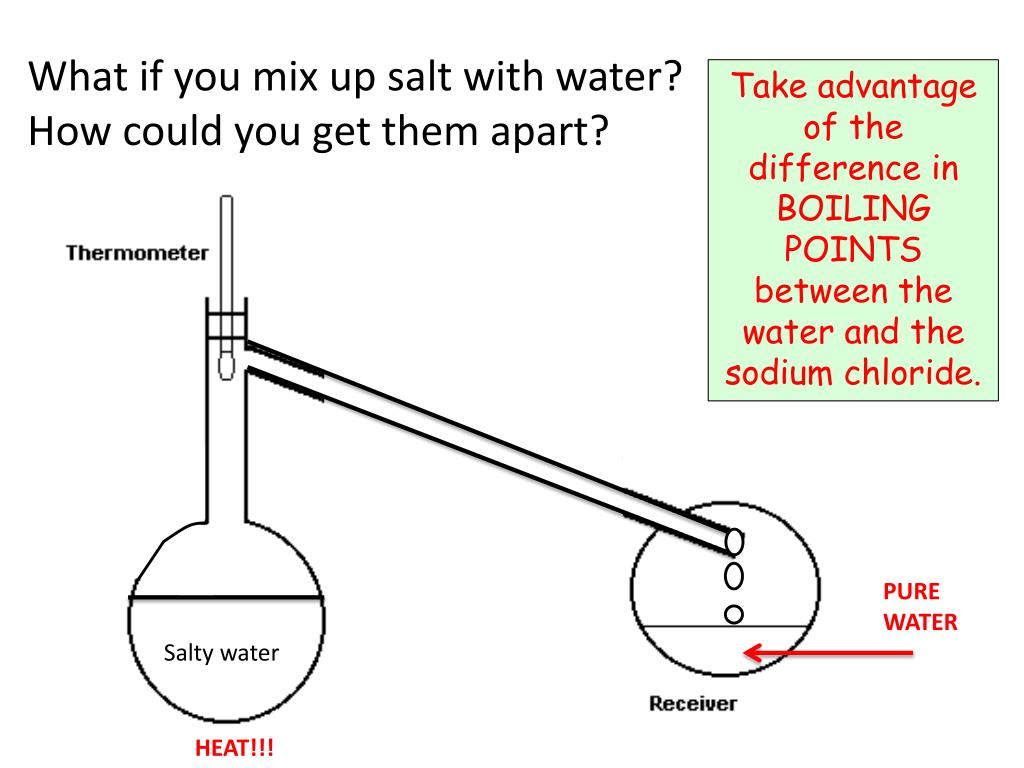
What is the freezing point of salt water?
Water freezes at 0°C (32°F), but it's most dense—that is, its molecules are most tightly packed—at 4°C (39°F). Salt water freezes at a lower temperature than fresh water does—and the saltier the water, the more it resists freezing. That's why we put salt on sidewalks in the winter.
What is the phase diagram of water?
At different temperatures and pressures, a phase diagram depicts the preferred physical states of matter. Water is a liquid at typical room temperatures and pressures, but it becomes solid (i.e. ice) if the temperature is reduced below 273 K, and gaseous (i.e. steam) if the temperature is increased above 373 K at the same pressure.
How does pressure affect the two phases of water?
In liquid water, ice floats. As a result, a change in pressure has the opposite effect on those two phases. When ice is near its melting point, it can be converted into liquid water by applying pressure. In the liquid phase, water molecules are actually closer together than in the solid phase.
Does salt water freeze faster than fresh water?
Salt water freezes at a lower temperature than fresh water does-and the saltier the water, the more it resists freezing. That's why we put salt on sidewalks in the winter. But melted salt water can hurt plants, so many people are switching to sand or other salt substitutes-even sugar will lower the freezing point and melt ice.
How many atoms are in water?
How does salt affect the ocean?
Why do we put salt on sidewalks?
What temperature does water freeze?
Why does water stick to other water molecules?
What does H2O mean in water?
When liquids freeze, they become more dense?
See 4 more
About this website
How many phases are present in an unsaturated salt solution?
one phaseCalculate the number of phases in an unsaturated solution of NaCl and water. Explanation: Since the salt is completely miscible in water, it forms a homogeneous mixture and hence it has only one phase (miscible liquids form only one phase and only one component).
Is salt a solid phase?
Salt, in its solid form, does not melt until 800°C. That proves the incredibly strong bonds between the negatively charged chloride ion and positively charged sodium ion. The electrostatic attraction between the oppositely charged ions creates an ionic bond that is in solid form at room temperature.
How many phases and components are present in an aqueous NaCl solution?
The saturated solution of sodium chloride consists of three phases—solid sodium chloride, salt solution and water vapour in equilibrium.
How many phases does a saturated solution have?
There are 3 phases.
Is salty water a solid liquid or gas?
In a chemistry class, we take solid salt NaCl (s) and dissolve it into pure liquid water H2O (l). The result is a salt water solution, denoted NaCl (aq). The solution is neither solid nor liquid. A solution is the 4th state of matter denoted (aq).
Does salt become liquid?
General Physical Properties of Salt Salt is a crystalline mineral. Like many minerals, it has a very high melting point. Salt's melting point is 800.8 degrees Celsius, or 1473.4 degrees Fahrenheit. At this temperature, salt turns into a liquid.
How do you calculate number of phases?
The phase rule states that F = C − P + 2. Thus, for a one-component system with one phase, the number of degrees of freedom is two, and any temperature and pressure, within limits, can be attained.
How many phases is sodium chloride?
An aqueous solution of a solid substance such as sodium chloride (or sugar) is uniform throughout. Therefore it is a 1-phase system. However, a saturated solution of sodium chloride in contact with excess solid sodium chloride is a 2-phase system.
How many phases of sugar and water are there?
two phasesAt all temperatures and compositions to the left of this line, there is one phase - a solution of sugar in water. To the right of this line, there are two phases present, the solution and solid sugar (that settles at the bottom of the cup). This could be considered to be a part of the sugar-water phase diagram.
Which of the following system has number of phases 3?
What is the degree of freedom for a water system? Explanation: We know that the number of phases and components in a water system is 3 and 1 respectively.
What is the number of phases in a mixture of benzene and water?
A mixture of two immiscible liquids such as water and benzene, will exist in two distinct liquid phases and in addition there will be a vapour phase. Thus there will be three phases each separated from the other by a well-defined bounding surface. A system consisting of more than one phase is said to be heterogeneous.
What is the difference between saturated and supersaturated solutions?
The solution obtained by dissolving maximum amount of solute at a given temperature is known as saturated solution. A solution which contains more amount of solute than that is required to saturate it is known as supersaturated solution. Was this answer helpful?
What type of solid is salt?
Ionic solids—Made up of positive and negative ions and held together by electrostatic attractions. They're characterized by very high melting points and brittleness and are poor conductors in the solid state. An example of an ionic solid is table salt, NaCl.
What type of crystal is salt?
Sodium chloride crystals are cubic in form. Table salt consists of tiny cubes tightly bound together through ionic bonding of the sodium and chloride ions. The salt crystal is often used as an example of crystalline structure.
Is salt a crystalline solid?
Salt is a clear, white, crystalline solid with a high melting point of 801°C. It shatters when hit with a hammer, forming many smaller crystals.
Is sugar a solid?
Sugar has close packing of constituent particles have its own volume and shape therefore it can be said to be solid whereas in water the constituent particles are not as closely packed as in solid. It has definite volume but not definite shape. Therefore it is a liquid.
THE 6 CHANGES OF STATE OF WATER
METEOROLOGIST JEFF HABY There are 3 states of water which are liquid, solid and gas. All three states exist on earth. Water has been called the life force.
Density of Water in its Three Phases - Study.com
Learn what a water phase diagram is and how to interpret the PT diagram for water. Know how to compare the density of water in its three phases....
3 states of water – interactive simulations – eduMedia
Water can exist in three states (or three phases): Solid phase: The particles in a solid are strongly bonded to one another. Ice cubes maintain their form regardless of the container that holds them. Liquid phase: The particles are no longer in an ordered state. The bonds between molecules are broken, and the liquid water takes the shape of its container.
How can water co-exist at three phases (solid, liquid and gas)?
How can water co-exist at three phases (solid, liquid and gas)? Asked by: Kenny Low Answer Water exists in three distinct phases at something called the triple point.
How many atoms are in water?
If you could peer into the submicroscopic world, you'd see that water consists of three atoms: two of hydrogen and one of oxygen. Water is a small molecule: A single living cell may contain billions of them. A water molecule is like a tiny magnet, with a negative charge on one side and a positive charge on the other.
How does salt affect the ocean?
Salt lowers the freezing temperature of water, which means that seawater, which is 3.5 percent salt, freezes at about -2.2°C (28°F). At the North and South poles, temperatures in the winter drop low enough to turn parts of the ocean into solid ice . This "sea ice" freezes and thaws with the seasons and plays a key role in regulating Earth's climate. The steady shrinking of Arctic Ocean sea ice in recent decades is strong evidence of climate change.
Why do we put salt on sidewalks?
But melted salt water can hurt plants, so many people are switching to sand or other salt substitutes—even sugar will lower the freezing point and melt ice.
What temperature does water freeze?
But as ice forms, the molecules arrange themselves in a crystal structure with empty spaces—and those spaces act as flotation devices. Water freezes at 0°C (32°F), but it's most dense—that is, its molecules are most tightly packed—at 4°C (39°F).
Why does water stick to other water molecules?
A water molecule clings to other water molecules because of its chemical structure. This "stickiness"—which comes from a force called hydrogen bonding—accounts for many of water's most amazing traits. Water Strider. © George Bernard/Animals Animals.
What does H2O mean in water?
Part of the Water: H2O = Life exhibition.
When liquids freeze, they become more dense?
As a rule, when liquids freeze, they become more dense. But forget about the rules when it comes to water: When frozen as ice, it becomes less dense. The result? Ice floats.