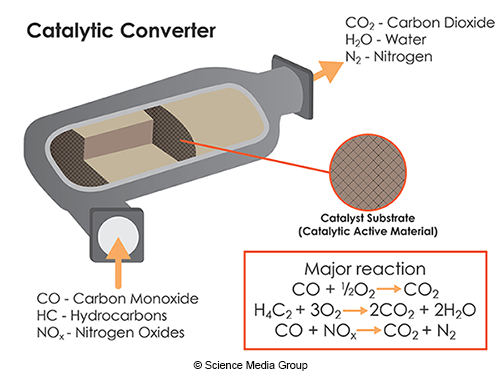
Are catalysts always permanently changed in a reaction?
Yes, a catalyst can be changed in a chemical reaction. But by definition a catalyst is not consumed as a reactant. It must be regenerated back into its original form after the reaction has taken place. Its function is to lower the reaction potential allowing the reaction between reactants to take place.
What affect does a catalyst have on a reaction?
- Catalysts are compounds that accelerate the rate of a reaction.
- Catalysts accelerate reactions by reducing the energy of the rate-limiting transition state.
- Catalysts do not affect the equilibrium state of a reaction.
What happens if a catalyst is used in a reaction?
What happens when a positive catalyst is used in a chemical reaction? The addition of a catalyst to a reaction lowers the activation energy, increasing the rate of the reaction. The activation energy of the uncatalyzed reaction is shown by Ea, while the catalyzed reaction is shown by Ea’.
Can a catalyst be used to slow down a reaction?
Under the Arrhenius equation, the reaction rate usually doubles for every 10 K rise in temperature. A catalyst acts to decrease the activation energy, which speeds up the rate of of reaction. Using more dilute solutions, or adding one or more reagents slowly will slow the rate of reaction.

Why do catalysts react with reactants?
Catalysts often react with reactants to form intermediates that eventually yield the same reaction products and regenerate the catalyst. Note that the catalyst may be consumed during one of the intermediate steps, but it will be created again before the reaction is completed.
How do catalysts work?
How Catalysts Work. Catalysts permit an alternate mechanism for the reactants to become products, with a lower activation energy and different transition state. A catalyst may allow a reaction to proceed at a lower temperature or increase the reaction rate or selectivity. Catalysts often react with reactants to form intermediates ...
What is the difference between a catalyzed reaction and an uncatalyzed reaction?
The only difference between a catalyzed reaction and an uncatalyzed reaction is that the activation energy is different . There is no effect on the energy of the reactants or the products.
What is a positive catalyst?
Positive and Negative Catalysts (Inhibitors) Usually when someone refers to a catalyst, they mean a positive catalyst, which is a catalyst that speeds up the rate of a chemical reaction by lowering its activation energy. There are also negative catalysts or inhibitors, which slow the rate of a chemical reaction or make it less likely to occur.
What is catalyst science?
Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. She has taught science courses at the high school, college, and graduate levels. A catalyst is a chemical substance that affects the rate of a chemical reaction by altering the activation energy required ...
What is the process of a chemical reaction called?
This process is called catalysis .
What happens when you mix two gases together?
However, if you add heat from a lighted match or a spark, you overcome the activation energy to get the reaction started. In this reaction, the two gases react to produce water (explosively). H 2 + O 2 ↔ H 2 O. The combustion reaction is similar.
What is a catalyst in chemistry?
What is a catalyst? A catalyst is a substance that is used to speed up a chemical reaction but it is not consumed by the reaction. What is catalysis? Catalysis is a process of increasing the rate of a chemical reaction by adding a chemical substance which is known as a catalyst. A very small amount of catalyst is required to alter the reaction rate.
What is a catalyzed reaction?
A catalyzed reaction is typically used to accelerate the rate by which specific chemistry is to proceed.
What happens at the reaction stage?
The reaction occurs and at this stage, both of the reactant molecules might attach themselves to the surface or one might get attached and hit by the other one that moves freely in the gas or liquid.
What is heterogeneous catalyzed reaction?
A heterogeneously catalyzed reaction occurs when the catalyst and the reactant exist in two different phases i.e. a solid catalyst in the presence of a reactant in solution.
Which metals are good catalysts?
Metals like platinum and nickel are considered as a good catalyst because they absorb strongly to hold and activate the reactants but not that much strongly that the products can’t break away.
Why are transition metal compounds used as catalysts?
Another good example of the use of the transition metal compounds as catalysts because of having an ability to change oxidation state.
Which type of catalysis goes through the same stages?
Majority of the examples of the heterogeneous catalysis go through the same stages