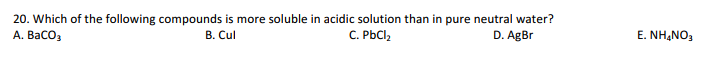
What is the minimum amount of solubility required to be insoluble?
The term insoluble is often applied to poorly or very poorly soluble compounds. A common threshold to describe something as insoluble is less than 0.1 g per 100 mL of solvent. If the answer is wrong.
Is anything completely insoluble in water?
Almost nothing is absolutely insoluble in water, however it is still useful to consider, for instance glass, as being insoluble even if some atoms do dissolve when a glass is filled with very pure water. In order for a salt to the soluble, theirs ions must be stable.
Why can't Al(OH)3 ionize and dissolve?
Al(OH)3 has a too highly charged cation: Al3+. There’s no way you can have that cation in presence of anything else without it stealing some electronic charge from other species. That is why Al(OH)3 cannot ionize and dissolve.
What is solubility in chemistry?
Solubility is the property of a solid, liquid, or gaseous chemical substance called solute to dissolve in a solid, liquid, or gaseous solvent. The solubility of a substance fundamentally depends on the physical and chemical properties of the solute and solvent as well as on temperature, pressure and the pH of the solution.

Is Al no3 soluble in water?
WaterAluminium nitrate / Soluble inThe chemical is both odorless and white as well as hygroscopic, meaning it readily absorbs water and moisture from the air. Due to this, it is easily soluble in water at a rate of 73.9 g/100ml at 20°C. The resulting aquatic solutions are acidic while aluminum nitrate does not dissolve well in alcohols.
Is Al no3 3 aqueous or solid?
Aluminum nitrate appears as a white, crystalline solid.
What happens when Al no3 3 is dissolved in water?
0:451:40Equation for Al(NO3)3 + H2O (Aluminum nitrate + Water) - YouTubeYouTubeStart of suggested clipEnd of suggested clipIt has a 3 plus charge and the no3 that's the nitrate ion it has a minus charge. So when we putMoreIt has a 3 plus charge and the no3 that's the nitrate ion it has a minus charge. So when we put solid alno3 in water liquid. Water it's going to break apart it's going to dissociate or dissolve.
Is Al OH 3 soluble or insoluble?
insolubleAluminium hydroxide, Al(OH)3 is insoluble in water, but dissolves readily in both acidic and basic solutions.
Is NO3 aqueous or liquid?
It is a nitrogen oxoanion, a member of reactive nitrogen species and a monovalent inorganic anion. It is a conjugate base of a nitric acid. Nitrates, inorganic, aqueous solution, n.o.s. is a liquid which is readily ignited when in contact with organic materials.
Is aluminum nitrate polar or nonpolar?
1:111:57Is Al(NO3)3 (Aluminum nitrate) Ionic or Covalent? - YouTubeYouTubeStart of suggested clipEnd of suggested clipThat's a non-metal.MoreThat's a non-metal.
Does Al NO3 3 form a basic solution?
0:011:30Is Al(NO3)3 acidic, basic, or neutral (dissolved in water)? - YouTubeYouTubeStart of suggested clipEnd of suggested clipIn this video we'll look at whether aluminum nitrate alno3 is acidic basic or neutral when it'sMoreIn this video we'll look at whether aluminum nitrate alno3 is acidic basic or neutral when it's dissolved in water to do that we need to know the neutralization reaction that produced the aluminum
Is Al NO3 3 ionic or covalent?
ionic compoundIs Al(NO3)3 covalent or ionic? Al(NO3)3 is an ionic compound that is made of a metal cation aluminum Al+3 and three nonmetal polyatomic anion NO3-. The net charge of Al(NO3)3 is equal to zero; one aluminum cation bonds with three nitrate anions.
Is Al soluble in water?
Aluminum chloride hydrolyses in water, and forms a mist when it comes in contact with air, because hydrochloric acid drops form when it reacts with water vapor. The most abundant aluminum compounds are aluminum oxide and aluminum hydroxide, and these are water insoluble.
Is Al2 co3 3 soluble?
About Aluminum Carbonate, Basic Basic Aluminum Carbonate is a water insoluble Aluminum source that can easily be converted to other Aluminum compounds, such as the oxide by heating (calcination).
Why is Aluminium hydroxide not soluble in water?
Because it reacts with both acids and bases. Like any conventional base, the OH radical in Al(OH)3 neutralizes H+ to make water and an aluminium salt.
Is Al2O3 soluble in NaOH?
07. Al2O3 is soluble in NaOH and HCl both.
Is Al NO3 3 an ionic compound?
Aluminium nitrateAluminium nitrate / IUPAC ID
Is Al NO3 3 covalent or ionic?
ionic compoundAl(NO3)3 is an ionic compound that is made of a metal cation aluminum Al+3 and three nonmetal polyatomic anion NO3-. The net charge of Al(NO3)3 is equal to zero; one aluminum cation bonds with three nitrate anions.
Does Al NO3 3 form a basic solution in water?
0:441:31Is Al(NO3)3 acidic, basic, or neutral (dissolved in water)? - YouTubeYouTubeStart of suggested clipEnd of suggested clipThat is a strong acid.MoreThat is a strong acid.
Is aluminum phosphate soluble in water?
Aluminum phosphate, solution appears as a colorless liquid. Insoluble in water.
Why can't hydrocarbons dissolve in water?
The reason why it is not able to dissolve in water is because the energy released in the formation of interaction between water and Hydrocarbon is insufficient to compensate for the energy abosrb to break the Instantaneous dipole-induced Dipole interaction ( Id-Id, Td-Id), hence it forms a suspension in water.
What is the energy of a solvent?
In a pure solvent, the molecules carry a great deal of energy in terms of their motion (Kinetic Energy). As they move from place to place, they have KE of translation: they also spin around so have KE of rotation and since their bonds behave much like springs they vibrate so they have KE of vibration.
Is glass insoluble in water?
Almost nothing is absolutely insoluble in water, however it is still useful to consider, for instance glass, as being insoluble even if some atoms do dissolve when a glass is filled with very pure water. 167 views. Quora User. , Highly electronegative!!
Can a substance be dissolved by an acid?
It is basic and can be dissolved by some acids. While not strictly true, it may be thought of as the acid reacting chemically to form another substance that is soluble.
Is aluminum hydroxide polymeric?
Aluminum hydroxide is polymeric and has extended bonding of Al and O atoms. And it has high lattice energy. Water cannot dissolve these type of compunds .
Does Al3+ ionize?
Al (OH)3 has a too highly charged cation: Al3+. There’s no way you can have that cation in presence of anything else without it stealing some electronic charge from other species. That is why Al (OH)3 cannot ionize and dissolve. Chin Yeh. , Electrons are players.
Is aluminum hydroxide amphoteric or amphoteric?
Aluminum hydroxide is amphoteric -- it reacts with both acids and bases. Like any conventional base, the OH radical in Al (OH)3 neutralizes H+ to make water and an aluminum salt. Al (OH)3 solid will also react with excess OH- to dissolve and form the complex ion [Al (OH)4]-. 7K views.