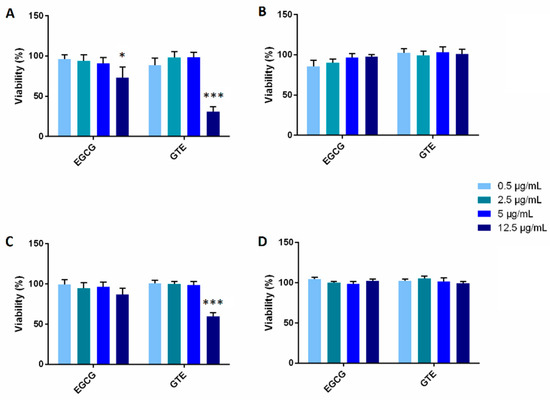
The substrates of catecholase are catechol and oxygen. The substrates react with one another within the active site of the enzyme. The products formed by this reaction are benzoquinone and water; since benzoquinone has a brown color, you can see that the reaction has taken place.
What is the substrate of catecholase?
We will be using the enzyme catecholase (or catechol oxidase) that is common in plants; it is present in the freshly squeezed potato juice that has been provided. The substrates of catecholase are catechol and oxygen. The substrates react with one another within the active site of the enzyme.
What are the uses of catechol substituents?
The chelating property of catechol substituent is the molecular basis for the adhesion properties of mussels to mineral surfaces. [17] Approximately 50% of the synthetic catechol is consumed in the production of pesticides, the remainder being used as a precursor to fine chemicals such as perfumes and pharmaceuticals. [9]
What is the nature of catechol?
Catechol occurs as feathery white crystals that are very rapidly soluble in water. Catechol was first isolated in 1839 by Edgar Hugo Emil Reinsch (1809–1884) by distilling it from the solid tannic preparation catechin, which is the residuum of catechu, the boiled or concentrated juice of Mimosa catechu ( Acacia catechu ). [4]
What happens when catechol is added to oxygen?
Upon mixing the enzyme with the substrate and exposure to oxygen (as when a potato or apple is cut and left out), the colorless catechol oxidizes to reddish-brown melanoid pigments, derivatives of benzoquinone. The enzyme is inactivated by adding an acid, such as citric acid contained in lemon juice.
Is catechol oxidase an enzyme or substrate?
Catechol oxidase (also known as catecholase) is an enzyme present in most fruits and vegetables. It facilitates the browning of cut or bruised produce by catalyzing a reaction between the substrate molecule catechol and atmospheric oxygen (O2).
What is the purpose of catechol?
Catechol is a derivative of benzene found in many fruits and other plant structures. Catecholase catalyzes the reaction of catechol and oxygen and is the enzyme that causes bruised or otherwise damaged fruit to turn brown.
What is catechol in biology?
Catechol (/ˈkætɪtʃɒl/ or /ˈkætɪkɒl/), also known as pyrocatechol or 1,2-dihydroxybenzene, is a toxic organic compound with the molecular formula C6H4(OH)2. It is the ortho isomer of the three isomeric benzenediols. This colorless compound occurs naturally in trace amounts.
Is catalase a substrate?
As mentioned, catalase is an enzyme, and it gets its name because it catalyzes (to cause or accelerate) a reaction involving hydrogen peroxide. Since catalase works on hydrogen peroxide, hydrogen peroxide is also known as the catalase's substrate.
What is the substrate that catalase breaks down?
When the enzyme catalase comes into contact with its substrate, hydrogen peroxide, it starts breaking it down into water and oxygen.
Is catechol a protein?
Catechol oxidase is a type 3 copper protein that exclusively catalyses the oxidation of catechols (e.g. o-diphenols) to the corresponding o-quinones.
Is catechol an acid or a base?
Catechol is a benzenediol comprising of a benzene core carrying two hydroxy substituents ortho to each other. It has a role as a genotoxin, an allelochemical and a plant metabolite. It is a conjugate acid of a catecholate(1-). Exposure to catechol may occur during its manufacture and use.
Is a potato a substrate?
One under-exploited source of en- zymes for classroom use is the potato (Solanum tuberosum), which tradition- ally serves as the substrate for class- room amylase studies.
What is a catechol group?
A catechol is an unsaturated six-carbon ring (phenolic group) with two hydroxyl groups attached to adjacent carbons (dihydroxyphenol). From: Vertebrate Endocrinology (Fifth Edition), 2013.
What is a substrate in biology?
Definition of substrate 1 : substratum. 2 : the base on which an organism lives the soil is the substrate of most seed plants. 3 : a substance acted upon (as by an enzyme)
What is the substrate for catalase quizlet?
What is the substrate of the catalase reaction? hydrogen peroxide.
What is the substrate for lipase?
So, the correct answer is 'Lipid'
What is catechol in onion?
Catechol, along with protocatechuic acid (3, 4-dihydroxybenzoic acid), appears to be the chief toxic substance that enables the pigmented onion to resist the invasion of the fungus Colletotrichum circinans, the organism responsible for the onion disease known as smudge.
Is catechol a drug?
A group of 1,2-benzenediols that contain the general formula R-C6H5O2. Used for the treatment of actinic keratoses (precancerous skin growths that can become malignant if left untreated).
What is the structure of catechol?
C6H6O2Catechol / Formula
How do you make catechol?
Catechol can be prepared by the oxidation of salicylaldehyde1 and of phenol;2 by the demethylation of guaiacol with aluminum chloride3 and hydriodic acid;4 by hydrolysis of ortho halogen-substituted phenols or ortho dihalobenzenes under suitable conditions;5 and by the replacement of sulfonic groups by alkali fusion.
What is the moiety of catechol?
Catechol is the adhesive moiety on the amino acid, 3,4-dihydroxyphenylalanine (DOPA), found in mussel adhesive proteins, which is capable of binding to both tissue and inorganic surfaces (i.e., polymer, metal) (Forooshani and Lee, 2017).
What is catechol hydrolyzed to?
Catechol hydrolyses in supercritical water to phenol as the main decomposition product and a number of other compounds as can be seen from the chromatogram shown in Figure 8.62. Other identified compounds are cyclopentanone, 2-cyclopentenone, 1,2-benzenedicarboxylic acid, nonylphenol, 1,4-dipropylbenzene, acetophenone, 2,6-di- tert -butylnaphthalene, 4-butoxyphenol, o -ethoxyphenol, 2-methylterephthalaldehyde, p -diethoxybenzene, and 5-methoxy-2,3,4-trimethylphenol [91].
What is the decomposition product of catechol?
In flash pyrolysis instruments, the catechol sample is easily volatilized and entrained by the flowing gas before reaching the decomposition temperature, and the yield of the decomposed compound is lower. The pyrolysis of catechol generates compounds such as CO, benzene, phenol, cyclopentadiene, acetylene, 1,3-butadiene, and ethylene. Also, considerable levels of PAHs are formed in the reaction. The variation with temperature of the yield of the main gas decomposition products of catechol (except for CO) is shown in Fig. 4.4.2, and the variation in the levels of benzene, cyclopentadiene, and phenol is shown in Fig. 4.4.3 [6].
What temperature does catechol decompose?
Catechol (1,2-dihydroxybenzene C6 H 4 (OH) 2) has a fairly high thermal stability. Only above 600°C does the compound start decomposing to a noticeable level, as shown in Fig. 4.4.1, where the percentage of unreacted catechol and that of generated CO at different temperatures are shown. The results were obtained from an experiment in which catechol vapors at a concentration of 0.72% mol carbon in nitrogen were heated at various temperatures in a quartz flow reactor with a residence time of 0.4 s and quenching of the reaction products at room temperature [6]. Due to the relatively high volatility of the compound, flash pyrolysis performed in a pyrolyzer for solid samples does not give the same decomposition yield as shown in Fig. 4.4.1.
Why is catechol a model compound for solid fuels?
Catechol is a model compound for solid fuels because it is present in biomass tar and tobacco smoke and is related to the chemical structure of lignin and brown coal.
What is the rate constant for catechol decomposition?
First-order kinetics are appropriate to describe the catechol decomposition in supercritical water with a global rate constant value for the catechol decomposition of k = 3.0 × 10 − 4 to 11.0 × 10 − 4 min − 1 (see Figure 8.65 ).
What are the small molecules formed by pyrolysis?
Other small molecules are formed at levels depending on pyrolysis temperature but not exceeding 3% of the feed catechol. These include methane (0%–2.5%), ethane (0%–0.3%), propyne (0%–1.6%), propadiene (0%–0.5%), and propylene (0%–0.2%). The reaction mechanisms for catechol pyrolysis are similar to those for phenol.
What is the reaction that occurs when substrates react with one another?
This is called the fruit browning reaction.
What is the pH scale?
The pH scale measures the numbers of hydrogen ions (each with a +1 charge) present in solution. The scale in biological systems runs from 0 (strongly acidic) to 14 (strongly basic). 7, the neutral point, is the pH of pure water. Recall that the folded structure of protein molecules is in large part due to hydrogen bonding between oppositely charged parts of the polypeptide chain. Other ions in solution could replace the matching parts of the chain, and make it come undone. When this happens, what becomes of the active site?
What is the specificity of enzymes?
Enzymes have high specificity--only specific reactants will fit on their surfaces and balance the local surface charges of the place called the active site. The particular set of reactants for an enzyme are called its substrate(s). Enzymes, like all catalysts, are not changed by the reaction they catalyze; thus, ...
Is hydroquinone the same as catechol?
Hydroquinone is structurally similar to but not the same as catechol. In this experiment we will see if the enzyme can also convert it to benzoquinone, or if the enzyme is simply too specific in its design to interact with the similar but not identical molecule.
Does catecholase require copper?
V. Necessity of Cofactors for Enzyme Activity [Optional] Many enzymes work only if there is a cofactorassociated with them. Catecholase requires copper as a cofactor. PTU is a chemical that has a higher affinity for copper than does catecholase, and it takes the copper away from the enzyme.
What is the catalytic mechanism of catechol 1,2-dioxygenase?
The catalytic mechanism of catechol 1,2-dioxygenase was elucidated using a combination of O 18 labeling experiments and crystallography. Upon entering the active site, the hydroxyl group on the fourth carbon (C4) of catechol binds to Fe 3+; this binding is facilitated by the hydroxide ligand, which deprotonates the C4 hydroxyl group. The second catechol hydroxyl group on carbon 3 (C3) is coordinated to Fe 3+ after its deprotonation by the Tyr200 ligand. In the process of deprotonating the C3 hydroxyl group, Tyr200 dissociates from the catalytic complex.
What are the active sites of the catalytic domain?
Without heme, iron must be ligated to four amino acid residues (Tyr200, His226 , Tyr164, His224) to maintain is catalytically active conformation. With Tyr200 and His226 acting as the axial ligands and Tyr164, His224, and a solvent water molecule acting as equatorial ligands, the Fe 3+ complex displays trigonal bipyramidal geometry. Since the active sites of each catalytic domain are separated by a distance of 40 Å, they are not believed to allosterically effect one another.
How is oxygen bonded to the substrate?
Oxygen is bonded to the substrate through a series of trans influences and stabilizing hydrogen bonding between the substrate and other active site amino acid residues . His226 accepts electron density from the substrate, consequently decreasing the bond between Fe 3+ and the C4 hydroxyl.
What is the effect of the hydrogen bonding between Arg221 and C3 hydroxyl?
These distortions, coupled with the hydrogen bonding between Arg221 and the C3 hydroxyl, induces the C3 hydroxyl group to ketonize and increases the carbanion character of C4. The newly formed C4 carbanion attacks O2, thus binding it to the substrate.
Does 1,2-CTD bind to lipids?
1,2-CTD degrades phenolic hydrocarbons key to the synthesis lipid membranes. Therefore, 1,2-CTD may bind to the cell lipid membrane via its terminal phospholipids and thus have greater access to the phenolic hydrocarbons vital in lipid ...
