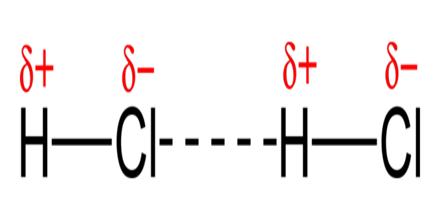
Why does CCl4 have a dipole moment?
The CCl4 is nonpolar in nature because of the symmetrical tetrahedral geometrical structure. Although the C-CL bond is polar in nature as Carbon and Chlorine atoms have a difference in their electronegativity. As a result, the C-Cl bond also has a dipole moment. But due to symmetrical structure, the net dipole moment gets canceled with each ...
Does CCl4 have a dipole moment?
The CCl4 is nonpolar in nature because of the symmetrical tetrahedral geometrical structure. Although the C-CL bond is polar in nature as Carbon and Chlorine atoms have a difference in their electronegativity. As a result, the C-Cl bond also has a dipole moment.
What are the intermolecular forces of CCl4?
Likewise, what kind of intermolecular forces are present in CCl4? Its strongest intermolecular forces are London dispersion forces. CCl4 is a tetrahedral molecule with a Cl-C-Cl bond angle of 109.5°. The two C-Cl bond dipoles in the plane of the paper have a resultant pointing to the right at an angle of 54.75° from the vertical.
What is the Lewis dot structure for CCl4?
CCl4 Lewis Structure. A CCL4 Lewis structure is a diagram that represents the electron configuration of covalently bonded compounds. Lewis structures are meant to provide a visualization of the atomic structure and the distribution of electrons in a given chemical compound.

Does CCl4 have dipole?
CCl4 has zero dipole moment whereas CHCl3 has non zero dipole moment.
Is CCl4 hydrogen bonding dipole-dipole or dispersion?
The answer is c. induced dipole-induced dipole. The molecule CCl4 C C l 4 is a nonpolar molecule. The intermolecular forces of attraction present for CCl4 C C l 4 is the London Dispersion Force of attraction which is also known as an induced dipole-induced dipole.
What is the intermolecular of CCl4?
CCl4 is a nonpolar molecule. Its strongest intermolecular forces are London dispersion forces. CH2Cl2 has a tetrahedral shape. The two C-Cl bond dipoles have a resultant that bisects the Cl-C-Cl bond angle.
Is ch4 dipole-dipole?
ch4 molecules are non polar, it has not dipole-dipole intraction. it has only london dispersion force. and ch4 molecules is not a dipole because there is no any negative pole and positive pole.
Why is CCl4 a dipole?
CCl4 has no net dipole moment because of its regular tetrahedral structure.
Why is there no dipole in CCl4?
Carbon tetrachloride is non polar molecule because the central carbon atom undergoes sp3 hybridization which results in regular tetrahedral geometry in which all the four C−Cl bond dipoles cancel each other. Hence, the molecule has no net dipole moment.
What type of dipole is CCl4 a polar compound?
The dipole moment of one bond cancels that of another placed opposite to it. Hence the two pairs of bonds in carbon tetrachloride cancel each other resulting in net zero dipole moment. Therefore carbon tetrachloride \[CC{l_4}\] is a nonpolar molecule.
Is CCl4 strongest intermolecular force?
The London dispersion force increases proportionally with the molar mass. Hence, CI4 C I 4 has the strongest intermolecular force.
How many intermolecular forces are in CCl4?
Because both Cl2 and CCl4 are nonpolar and have no other special identifying characteristics, the only intermolecular forces between the two molecules are London Dispersion Forces.
How do you identify dipole-dipole forces?
4:5912:15Dipole Dipole Forces of Attraction - Intermolecular Forces - YouTubeYouTubeStart of suggested clipEnd of suggested clipBetween two polar molecules is a dipole force. So these are dipole-dipole forces here we have oneMoreBetween two polar molecules is a dipole force. So these are dipole-dipole forces here we have one dipole.
Is co2 dipole-dipole?
Carbon dioxide does not have dipole-dipole forces due to symmetry of the dipoles found in the molecule as a result of the polar bonds. Carbon dioxide is not a polar molecule despite its polar bonds. Carbon dioxide also does not have hydrogen bond forces because it is a nonpolar molecule.
Does Cl2 have dipole-dipole forces?
Answer and Explanation: Cl2 does not have a dipole moment.
Is CCl4 hydrogen bonding?
Nonpolar molecules experience only induced dipole (dispersion or London) forces, and of the examples above, only CCl4 (l) and Br2 (l) are nonpolar. Only polar molecules with H-F, H-O, and H-N bonds experience hydrogen bonds. Of the examples above, only NH3 (l), H2O (l), and HF (l) experience hydrogen bonds.
Is there hydrogen bonding in CCl4?
The interaction between CH3OH and CCl4 at the molecular level can be twofold: hydrogen bond (O–H⋯Cl) and halogen bond (C–Cl⋯O) interaction. One halogen bonded minimum and two hydrogen bonded minima are identified in the dimer potential energy surface.
Does CCl4 show hydrogen bonding?
However, C-OH... PO2- hydrogen bonds can be identified in dry films of the sodium salt of 18:0-CL or in CCl4 solution.
Does CCl4 have only dispersion forces?
Because both Cl2 and CCl4 are nonpolar and have no other special identifying characteristics, the only intermolecular forces between the two molecules are London Dispersion Forces.