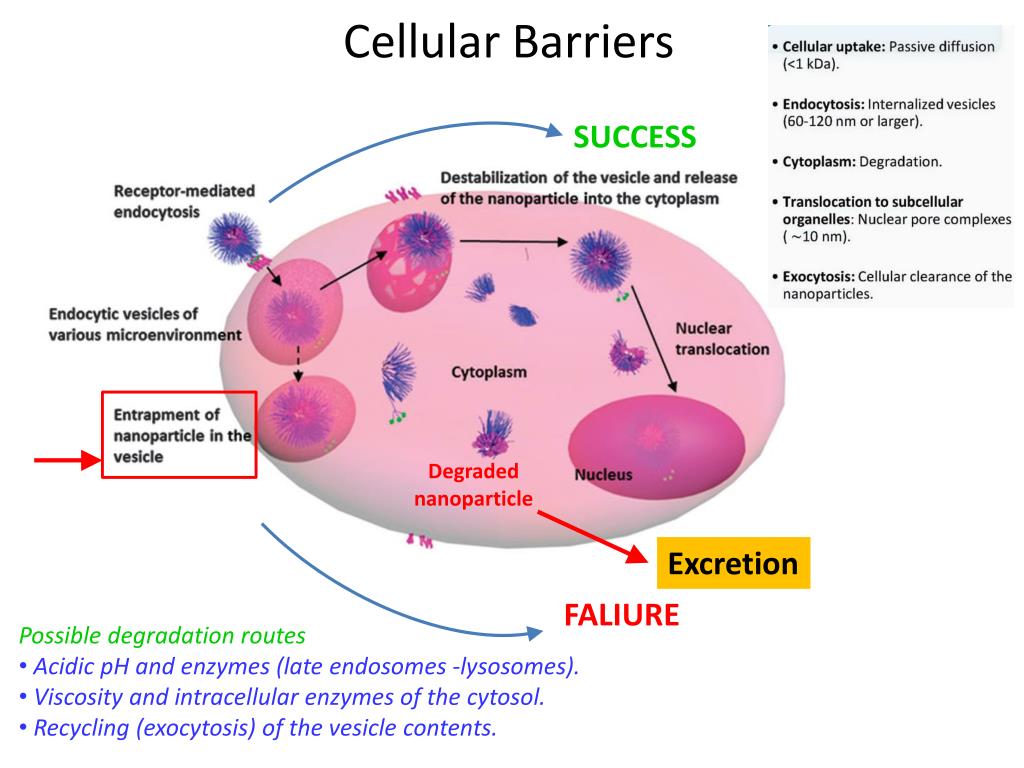
Precautions
Ibuprofen is a relatively non-polar molecule, so it can be absorbed easily through the outer skin layer, but has difficulty penetrating the inner layer. When mixed with a more polar solvent such as 2-propanol (propan-2-ol), the mixture is sufficiently polar to carry the ibuprofen through the inner layer of the skin, but not so polar that it ...
Is ibuprofen a polar or nonpolar molecule?
Is ibuprofen polar or nonpolar? Ibuprofen has a polar end, the carboxylic acid group, which can act both as a hydrogen donor and a hydrogen acceptor establishing moderate to strong interactions with other polar molecules. The other part of ibuprofen is non-polar.
Is ibuprofen a polar molecule?
The third component, caffeine, which is the most polar, is eluted last with acetone and observed by TLC. What is the polarity of ibuprofen? Unlike acetaminophen with three polar groups, ibuprofen only has one polar group, the carboxylic acid functional group, making this over-the-counter painkiller somewhat soluble in water and soluble in organic solvents.
Is caffeine or ibuprofen more polar?
• Ibuprofen. This over-the-counter pain reliever can cause a drug test to show up as positive for barbiturates, benzodiazepines, and marijuana. 12000 mg of ibuprofen, it is thought, may interfere with the enzyme used in the testing method. Ibuprofen is in a class of pain relievers called NSAIDs (non-steroidal anti-inflammatory drugs).
Does ibuprofen contain benzodiazepines?
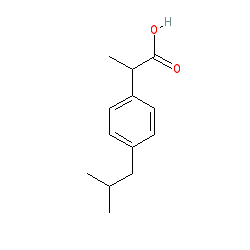
What type of molecule is ibuprofen?
Ibuprofen is a monocarboxylic acid that is propionic acid in which one of the hydrogens at position 2 is substituted by a 4-(2-methylpropyl)phenyl group.
Why is ibuprofen not very polar?
As with aspirin and paracetomol, two other pain-killing pharmaceuticals, ibuprofen features a six-membered ring structure, which avoids the polar environment of water.
What type of bond is ibuprofen?
One of the key features defining the ibuprofen structure is the doubly intermolecular O-HO[double bond, length as m-dash]C hydrogen bond in cyclic dimers as know from carboxylic acids and confirmed by X-ray analysis.
Does ibuprofen have high polarity?
Ibuprofen contains only one ester group and highest Rf value. So it is the least polar.
Is ibuprofen non-polar or polar?
non-polarIbuprofen has a polar end, the carboxylic acid group, which can act both as a hydrogen donor and a hydrogen acceptor establishing moderate to strong interactions with other polar molecules. The other part of ibuprofen is non-polar.
What makes a drug polar or nonpolar?
Polar drugs entrapped within liposomes are found in the internal aqueous compartments of these particles, and are readily released when the liposome membrane is ruptured by ultrasonic energy. Non-polar drugs are found or intercalated into the liposome membrane as well as being found in the aqueous compartments.
Is ibuprofen covalent?
Ibuprofen is synthesized when a chemist binds together carbon, hydrogen, and oxygen atoms by covalent bonds.
Does ibuprofen dissolve in water?
Ibuprofen is a commonly used pain reliever. Its chemical properties include: Ibuprofen has a poor water solubility with less than 1 mg of ibuprofen dissolving in 1 ml water. It is more soluble in aqueous alcohol mixtures.
Is ibuprofen a molecule or compound?
Ibuprofen is a frequently used over-the-counter drug for treating pain, inflammation, and fever. It is a relatively simple molecule that was discovered and developed in the 1950s and 1960s at Boots Pure Drug in Nottingham, UK (now Boots UK in Beeston).
What drugs are polar?
Gentamicin, vancomycin, and calcitonin are polar drugs that have poor oral availability.
Is ibuprofen more polar than aspirin?
a. (Acetaminophen; Aspirin; Caffeine, and Ibuprofen) D cafteine > Acetaminophen > phenacetin > Aspirin> ibuprofen Aspirin is more polar than ibuprofen more H bond acceptors and donators acetamingghen phenacetin it has a OH that can H bond .
Are NSAIDs polar?
Although NSAIDs can vary significantly in terms of structure, size, and complexity, in general, all NSAIDs are amphiphilic with most possessing carboxylic acid groups or other such polar groups (Fig. 1).
Is ibuprofen less polar than aspirin?
a. (Acetaminophen; Aspirin; Caffeine, and Ibuprofen) D cafteine > Acetaminophen > phenacetin > Aspirin> ibuprofen Aspirin is more polar than ibuprofen more H bond acceptors and donators acetamingghen phenacetin it has a OH that can H bond .
Is ibuprofen a hydrophobic drug?
Analysis of MD simulations indicated that ibuprofen molecules prefer to be located in the hydrophobic acyl chain region of DMPC/cholesterol bilayers.
Why is aspirin not very polar?
Answer and Explanation: Aspirin is considered less polar as compared to caffeine because in aspirin only one group of the ester is present along with only one group of carboxylic acid. On the contrary, caffeine has a high polar nature comparatively.
Is ibuprofen insoluble in water?
Ibuprofen has a poor water solubility with less than 1 mg of ibuprofen dissolving in 1 ml water. It is more soluble in aqueous alcohol mixtures.
What is the net dipole of a polar molecule?
A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. (Wikipedia)
Why do polar molecules have polar bonds?
Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms. A polar molecule with two or more polar bonds must have an asymmetric geometry so that the bond dipoles do not cancel each other.
Which bond is the shortest in ibuprofen?
3D Model of Ibuprofen (2) The O-H bond is the shortest bond because Oxygen and Hydrogen are two smallest elements in the molecule. The strongest bond is the C=O bond because it is a double [pi] bond, which is stronger than single [sigma] bonds.
How does ibuprofen work?
In order for it to take effect, ibuprofen must first travel through the body. It goes down the esophagus, eventually residing in the stomach. Since Ibuprofen is not polar (seen in the design page), it can safely travel through the polar bloodstream without being dissolved. The outside of the pill allows the fragile inside to safely through the stomach without being dissolved early. Once there, the gastric acid dissolves it. This allows the ibuprofen to be absorbed into the bloodstream through the intestinal wall, and go to the area that is releasing the prostaglandinas. Over 98% of the Ibuprofen is bound to plasma proteins, and it can also get access to the central nervous system. Ibuprofen will travel to almost anywhere in the body if needed, because of the high variability of where prostaglandinas can be produced. It works by latching onto to an enzyme called cyclooxygenase, blocking it out, which stops it from making the prostaglandins.
What is the formula for ibuprofen?
Ibuprofen – C13H18O2 – 206.285. Structural Formula (1) Ibuprofen is one of the most popular NSAID (Non-steroidal Anti-inflammataory Drug) Drugs, used to treat pain, fevers, and inflammation. It comes in several forms, but we are focusing on the tablets. Ibuprofen is derived from 2-methylpropylbenzene.
How long does it take for ibuprofen to leave the body?
Usually the drug will be fully absorbed within 30 minutes to 2 hours after taken orally. The Half life of the drug varies between 1.8 to 2 hours. When it comes time for the drug to leave the body, it is excreted as urine.
Why is ibuprofen used for pain?
Ibuprofen is used to block the production of prostaglandins. These are chemicals that the body produces when someone is sick or injured. They make nearby nerves sensitive to pain, so your body can react and start the healing process. They can also swell tissue depending on the injury/sickness.
What is the negative side of ibuprofen?
There are 3 de localized pi orbitals. The negative region of the medicine is where it bonds with oxygen. The positive side is the side where Oxygen bonds with Hydrogen [specifically the Hydrogen]. The C-C bonds are the longest bonds because Carbon is the largest element in the molecule, and it is also the weakest because of the length of the bond.
Does ibuprofen travel to the central nervous system?
Over 98% of the Ibuprofen is bound to plasma proteins, and it can also get access to the central nervous system. Ibuprofen will travel to almost anywhere in the body if needed, because of the high variability of where prostaglandinas can be produced.
What is the melting point of ibuprofen?
Ibuprofen is a white crystalline solid with a melting point of 76 o C.
What is the skeletal formula of ibuprofen?
Ibuprofen is synthesized from 2-methylpropylbenzene which can be made from compounds separated from crude oil. The skeletal formula of 2-methylpropylbenzene is shown on the right. Notice that the only functional group is the benzene ring.
What is the reaction of ibuprofen to hydrogencarbonate?
Ibuprofen is a carboxylic acid, it will react with hydrogencarbonate solutions to produce a salt, water and carbon dioxide gas.
How to determine ibuprofen content?
The ibuprofen content of commercially available tablets can be determined using a titration with a strong base .
How many steps are involved in synthesis of ibuprofen?
The so-called "Green" synthesis of ibuprofen involves 3 steps 2 .
What is the trade name for ibuprofen?
Ibuprofen is sold under a large number of tradenames including Advil™, Brufen™, Motrin™ and Nurofen™.
Why is salt faster acting than ibuprofen?
Because the salt is more water soluble, it is faster acting than molecular ibuprofen.
How long does ibuprofen last?
Ibuprofen has a dose-dependent duration of action of approximately 4–8 hours, which is longer than suggested by its short half-life. The recommended dose varies with body mass and indication. Generally, the oral dose is 200-400 mg (5-10 ...
When was ibuprofen invented?
History Ibuprofen was developed by the research arm of Boots Group during the 1960s [1]. Its discoverer was Stewart Adams, with colleagues John Nicholson and Colin Burrows. Its patent was filed in 1961. The drug was launched as a treatment for rheumatoid arthritis in the United Kingdom in 1969, and in the United States in 1974.
What is the name of the drug that is used to treat arthritis?
Ibuprofen. Ibuprofen (from the earlier nomenclature i so- bu tyl- pro panoic- phen olic acid) is a non-steroidal anti-inflammatory drug (NSAID) originally marketed as Nurofen and since then under various other trademarks (See tradenames section). It is used for relief of symptoms of arthritis, primary dysmenorrhea, fever, and as an analgesic, ...
Is ibuprofen lysine safe for premature infants?
Ibuprofen lysine is indicated for closure of a patent ductus arteriosus in premature infants weighing between 500 and 1500 grams, who are no more than 32 weeks gestational age when usual medical management (e.g., fluid restriction, diuretics, respiratory support, etc.) is ineffective. [6]With regard to this indication, ibuprofen lysine is an effective alternative to intravenous indomethacin and may be advantageous in terms of renal function. [8]
Is ibuprofen lysine the same as ibuprofen?
In Europe, Australia, and New Zealand, ibuprofen lysine (ibuprofen lysinate, the lysine salt of ibuprofen) is licensed for treatment of the same conditions as ibuprofen. The lysine salt increases water solubility, allowing the medication to be administered intravenously. [6] Ibuprofen lysine has been shown to have a more rapid onset of action compared to base ibuprofen. [7]
Is ibuprofen a non-selective drug?
Adverse effects. Ibuprofen appears to have the lowest incidence of gastro intestinal adverse drug reactions (ADRs) of all the non-selective NSAIDs. However, this only holds true at lower doses of ibuprofen, so over-the-counter preparations of ibuprofen are generally labeled to advise a maximum daily dose of 1,200 mg.
Does ibuprofen help with Parkinson's?
Ibuprofen has been associated with a lower risk of Parkinson's disease, and may delay or prevent Parkinson's disease. Aspirin, other NSAIDs, and paracetamol had no effect on the risk for Parkinson's. [5] . Further research is warranted before recommending ibuprofen for this use.
