
See more
What class of drug is ipratropium bromide?
Ipratropium belongs to the family of medicines known as bronchodilators. Bronchodilators are medicines that are breathed in through the mouth to open up the bronchial tubes (air passages) in the lungs. This medicine is available only with your doctor's prescription.
Is ipratropium bromide a long acting anticholinergic?
Ipratropium bromide is an anticholinergic drug frequently administered for the treatment of COPD. Unfortunately, ipratropium has a short duration of action, requiring administration every 6 hours; this regimen affects adherence to drug therapy.
What is the most common anticholinergic bronchodilator?
The 3 most widely used bronchodilators are: beta-2 agonists, such as salbutamol, salmeterol, formoterol and vilanterol. anticholinergics, such as ipratropium and tiotropium.
When should you not use ipratropium?
Previous severe allergic reaction symptoms upon using ipratropium or atropine and its other derivatives, such as angioedema, urticaria, severe shortness of breath, oropharyngeal edema, and ultimately anaphylaxis is a contraindication to ipratropium use.
Can you use ipratropium bromide long term?
For the 0.06% nasal spray: For runny nose associated with the common cold: Adults, teenagers, and children 5 years of age and older—2 sprays in each nostril three or four times a day. Do not use the medicine for more than 4 days.
Which neurological system will ipratropium effect?
Ipratropium acts as an antagonist of the muscarinic acetylcholine receptor. This effect produces the inhibition of the parasympathetic nervous system in the airways and hence, inhibit their function.
What are the 4 most common anticholinergic drugs?
It was found the most common anticholinergic drug classes used by the elderly in the study were: tricyclic antidepressants (for example: amitriptyline) first generation antihistamines (for example: chlorpheniramine, diphenhydramine) overactive bladder antimuscarinics (for example: oxybutynin)
What are two examples of anticholinergic drugs?
The following are some examples of anticholinergic medications:Dicyclomine.Hyoscyamine (Levsin)Scopolamine (Transderm Scop)Glycopyrrolate (Robinul, Cuvposa)Qbrexza (glycopyrronium)Trihexyphenidyl.Benztropine (Cogentin)Solifenacin (Vesicare)More items...•
What are strong anticholinergic drugs?
Some of the strongest anticholinergic drugs include antihistamines, sleeping pills, tricyclic antidepressants, and drugs to treat an overactive bladder.
What are the adverse effects of ipratropium?
Ipratropium may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:dizziness.nausea.heartburn.constipation.dry mouth.difficulty urinating.pain when urinating.frequent need to urinate.More items...•
What does ipratropium bromide do to the body?
Ipratropium is used to control and prevent symptoms (wheezing and shortness of breath) caused by ongoing lung disease (chronic obstructive pulmonary disease-COPD which includes bronchitis and emphysema). It works by relaxing the muscles around the airways so that they open up and you can breathe more easily.
Does ipratropium affect the heart?
Heart rate already started to rise during the injection of ipratropiumbromide. It increased by an average of 78% three minutes following the administration as compared to pretreatment control values. Mean heart rate still was markedly increased by an average of 26% at 120 min following the injection of the drug.
Is ipratropium bromide short or long-acting?
Background: Short-acting anticholinergic bronchodilator, ipratropium bromide has been recommended as first-line drug in chronic obstructive pulmonary disease (COPD).
What are long-acting anticholinergics?
Long-acting anticholinergics are effective and convenient for preventing and treating COPD in a person whose symptoms do not go away (persistent symptoms).
What is the duration of action for ipratropium bromide?
In acute exacerbations, ipratropium bromide is useful but may not be the preferred agent because of a delayed onset of action (within 15 minutes; mean dose-dependent duration of effect, three to five hours).
How long does ipratropium bromide stay in your system?
Steroids Used for AsthmaHalf-Lives of Common Asthma MedicationsClassMedicationHalf-LifeAnticholinergicipratropium bromide3 to 5 hoursMethylxanthinetheophylline3 to 13 hoursControllers8 more rows•Jan 6, 2020
How long does it take for ipratropium to work?
As with the beta 2-adrenoceptor agonists, the onset of maximum effect with ipratropium (about 1.5 to 2 hours) is slower than with isoprenaline (although significant bronchodilation usually occurs within seconds or minutes of ipratropium inhalation), and the duration of effect (about 4 to 6 hours) is longer.
Is ipratropium a beta agonist?
Ipratropium thus appears to be a suitable alternative to beta 2-adrenoceptor agonist drugs in patients not fully responding to these agents, and combined therapy with ipratropium and other bronchodilating drugs may prove to be an important area of use in patients failing to respond adequately to a single drug regimen.
Is ipratropium bromide effective in asthma?
Ipratropium bromide: a review of its pharmacological properties and therapeutic efficacy in asthma and chronic bronchitis. Ipratropium bromide is an anticholinergc bronchodilator administered by inhalation. Although producing bronchodilation in most patients with obstructive airways disease, it is somewhat less effective than beta 2-adrenoceptor ...
Is ipratropium bromide an anticholinergc?
Ipratropium bromide is an anticholinergc bronchodilator administered by inhalation. Although producing bronchodilation in most patients with obstructive airways disease, it is somewhat less effective than beta 2-adrenoceptor agonist drugs such as salbutamol or fenoterol in patients with asthma, but is at least as effective as these agents in ...
How much ipratropium is used for asthma?
It has anticholinergic properties and is used to treat asthma. The metered dose of ipratropium is some 20 micrograms. Its limited availability accounts for its relative bronchial selectivity when ipratropium is given in low doses by the aerosol route.
How long does it take for ipratropium to work?
The onset of action of ipratropium is relatively slow (20 minutes), and the peak effect occurs at 60 minutes.1 Ipratropium, unlike atropine, is poorly absorbed across mucous membranes and has little toxicity at the usual doses. In particular, ipratropium does not inhibit mucociliary clearance.
What is the anticholinergic agent for asthma?
The anticholinergic agent ipratropium bromide (Atrovent) is useful in the management of children with moderate to severe asthma exacerbations.36–39 Ipratropium is similar to atropine, but contains an ammonium group, which prevents systemic absorption.
Is ipratropium a beta 2 agonist?
Ipratropium has established a place in the therapy of asthma as an alternative to beta 2 -adrenoceptor agonist aerosols for patients who fail to respond adequately to these agents. Ipratropium may be more effective than beta 2 -adrenoceptor agonists for patients with non-atopic asthma and chronic bronchitis.
Is ipratropium bromide a cholinergic antagonist?
Ipratropium Bromide. Ipratropium bromide is a muscarinic cholinergic antagonist and is used in asthma, primarily in patients who either are intolerant of β2- agonists or are experiencing limited benefit from SABA use.84. From: Middleton's Allergy Essentials, 2017. Download as PDF.
Is Budesonide better than Ipratropium?
Budesonide achieved better control of sneezing than ipratropium throughout the study period and better control of congestion than ipratropium in the later part of the study. Of 36 patients who did not complete the study, largely for administrative reasons, six using ipratropium withdrew owing to lack of efficacy.
Is ipratropium bromide better than budesonide?
Both treatments resulted in significant improvements in rhinorrhea, sneezing, and congestion (as rated by both patients and physicians) and improved quality of life. Budesonide achieved better control of sneezing than ipratropium throughout the study period and better control of congestion than ipratropium in the later part of the study. Of 36 patients who did not complete the study, largely for administrative reasons, six using ipratropium withdrew owing to lack of efficacy. There were no reports of systemic anticholinergic adverse effects. The commonest nasal adverse events in both groups were nasal congestion (23% with ipratropium and 18% with budesonide) and rhinitis (13% with ipratropium and 7% with budesonide). Of nasal adverse events considered to be related to treatment, epistaxis and nasal irritation were more common with budesonide (10% and 4% respectively) than with ipratropium (8 and 0%). Sneezing occurred in 5% of those using ipratropium and in none of those using budesonide, probably reflecting the better efficacy of budesonide for control of sneezing. Overall both drugs were well tolerated.
What is ipratropium bromide?
Ipratropium Bromide Inhalation Solution is an anticholinergic (parasympatholytic) agent that, based on animal studies, appears to inhibit vagally-mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released from the vagus nerve.
What is the effect of anticholinergics on bronchial smooth muscle?
Anticholinergics prevent the increases in intracellular concentration of cyclic guanosine monophosphate (cyclic GMP) which are caused by interaction of acetylcholine with the muscarinic receptor on bronchial smooth muscle.
Is ipratropium bromide an adverse reaction?
Adverse reaction information concerning Ipratropium Bromide Inhalation Solution is derived from 12-week active-controlled clinical trials. Additional information is derived from foreign postmarketing experience and the published literature.#N#All adverse events, regardless of drug relationship, reported by three percent or more patients in the 12-week controlled clinical trials appear in the table.#N#Additional adverse reactions reported in less than three percent of the patients treated with Ipratropium bromide include tachycardia, palpitations, eye pain, urinary retention, urinary tract infection and urticaria. Cases of precipitation or worsening of narrow-angle glaucoma, mydriasis, and acute eye pain have been reported.#N#Lower respiratory adverse reactions (bronchitis, dyspnea and bronchospasm) were the most common events leading to discontinuation of Ipratropium bromide therapy in the 12-week trials. Headache, mouth dryness and aggravation of COPD symptoms are more common when the total daily dose of Ipratropium bromide equals or exceeds 2,000 mcg. Allergic-type reactions such as skin rash, angioedema of tongue, lips and face, urticaria, laryngospasm and anaphylactic reaction have been reported. Many of the patients had a history of allergies to other drugs and/or foods.
Is ipratropium bromide excreted in human milk?
It is not known whether Ipratropium bromide is excreted in human milk. Although lipid-insoluble quaternary bases pass into breast milk, it is unlikely that Ipratropium bromide would reach the infant to a significant extent, especially when taken by inhalation since Ipratropium bromide is not well absorbed systemically after inhalation or oral administration. However, because many drugs are excreted in human milk, caution should be exercised when Ipratropium bromide is administered to a nursing woman.
Does ipratropium bromide cause teratogenic effects?
Oral reproduction studies performed in mice, rats and rabbits at doses of 10, 100, and 125 mg/kg respectively and inhalation reproduction studies in rats and rabbits at doses of 1.5 and 1.8 mg/kg (or approximately 38 and 45 times the recommended human daily dose) respectively, have demonstrated no evidence of teratogenic effects as a result of Ipratropium bromide. However, no adequate or well controlled studies have been conducted in pregnant women. Because animal reproduction studies are not always predictive of human response, Ipratropium bromide should be used during pregnancy only if clearly needed.
Is ipratropium bromide a single agent?
The use of Ipratropium Bromide Inhalation Solution as a single agent for the relief of bronchospasm in acute COPD exacerbation has not been adequately studied. Drugs with faster onset of action may be preferable as initial therapy in this situation. Combination of Ipratropium Bromide Inhalation Solution and beta agonists has not been shown to be more effective than either drug alone in reversing the bronchospasm associated with acute COPD exacerbation.#N#Immediate hypersensitivity reactions may occur after administration of Ipratropium bromide, as demonstrated by rare cases of urticaria, angioedema, rash, bronchospasm and oropharyngeal edema.
Is ipratropium a bronchodilator?
Ipratropium Bromide Inhalation Solution administered either alone or with other bronchodilators, especially beta adrenergics, is indicated as a bronchodilator for maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease, including chronic bronchitis and emphysema.
What is ipratropium bromide?
New Window. Ipratropium Bromide is the bromide salt form of ipratropium, a synthetic derivative of the alkaloid atropine with anticholinergic properties. Ipratropium antagonizes the actions of acetylcholine at parasympathetic, postganglionic, effector-cell junctions.
Is ClinicalTrials.gov copyrighted?
The ClinicalTrials.gov data carry an international copyright outside the United States and its Territories or Possessions. Some ClinicalTrials.gov data may be subject to the copyright of third parties; you should consult these entities for any additional terms of use.
Is ipratropium anhydrous or anhydrous?
Ipratropium Bromide Anhydrous is the bromide salt form of anhydrous ipratropium, a synthetic derivative of the alkaloid atropine with anticholinergic properties. Ipratropium antagonizes the actions of acetylcholine at parasympathetic, postganglionic, effector-cell junctions. When inhaled, ipratropium binds competitively to cholinergic receptors in the bronchial smooth muscle thereby blocking the bronchoconstrictor actions of the acetylcholine mediated vagal impulses. Inhibition of the vagal tone leads to dilation of the large central airways resulting in bronchodilation.
Is ipratropium a bronchodilator?
This opens the bronchi, so providing relief in chronic obstructive pulmonary disease and acute asthma. It has a role as a bronchodilator agent, a muscarinic antagonist and an antispasmodic drug. It contains an ipratropium.
Is the FDA website copyrighted?
Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphic s, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.
Does ipratropium block vagal impulses?
When inhaled, ipratropium binds competitively to cholinergic receptors in the bronchial smooth muscle thereby blocking the bronchoconstrictor actions of the acetylcholine mediated vagal impulses. Inhibition of the vagal tone leads to dilation of the large central airways resulting in bronchodilation.
What is ipratropium bromide?
Ipratropium bromide is a quaternary ammonium derivative of atropine sulfate that blocks muscarinic (M) receptors on airway smooth muscle and submucosal gland cells in a relatively nonselective fashion (Figure 40-4 ).
How long does it take for ipratropium bromide to work?
The onset of bronchodilatation is relatively slow and is usually maximal 30–60 min after inhalation, but may persist for more than 6 h. It is usually given by MDI three to four times daily on a regular basis, rather than intermittently for symptom relief, in view of its slow onset of action.
How long does tiotropium last?
Tiotropium is a long-acting anticholinergic bronchodilator that requires only once-daily dosing, because its effects last more than 24 h. Tiotropium binds with equal avidity to M 1, M 2, and M 3 receptors but dissociates fairly rapidly from the M 2 receptors but more slowly from M 1 and M 3 receptors. It is available as an 18-μg/dose dry powder inhaler (DPI) formulation. Reports of side effects indicate they are no greater than those seen with the short-acting anticholinergics (with the primary adverse effect being dry mouth). Tiotropium's onset of peak bronchodilation is between 1 and 3 h. In clinical studies, tiotropium is more effective than regularly scheduled ipratropium (or placebo) in terms of improving lung function, symptoms, quality of life, and decreasing acute exacerbations ( Figures 40-5 and 40-6 ). In addition, studies to date suggest that tiotropium produces better bronchodilation and improvements in dyspnea than salmeterol (a long-acting β 2 -agonist described later) in patients with COPD.
How much ipratropium is given?
The metered dose of ipratropium is some 20 micrograms. Its limited availability accounts for its relative bronchial selectivity when ipratropium is given in low doses by the aerosol route. In healthy volunteers, 120–280 micrograms given by intravenous injection reduced salivary secretion and increased heart rate. Inhaled doses, up to a total of 1.2 mg, had no significant effect on heart rate, although some patients reported dryness of the mouth [ 1 ]. Only if substantial overdosage occurs (for example 1 mg by inhalation) do traces of generalized atropine-like effects begin to appear.
Is ipratropium bromide better than budesonide?
Both treatments resulted in significant improvements in rhinorrhea, sneezing, and congestion (as rated by both patients and physicians) and improved quality of life. Budesonide achieved better control of sneezing than ipratropium throughout the study period and better control of congestion than ipratropium in the later part of the study. Of 36 patients who did not complete the study, largely for administrative reasons, six using ipratropium withdrew owing to lack of efficacy. There were no reports of systemic anticholinergic adverse effects. The commonest nasal adverse events in both groups were nasal congestion (23% with ipratropium and 18% with budesonide) and rhinitis (13% with ipratropium and 7% with budesonide). Of nasal adverse events considered to be related to treatment, epistaxis and nasal irritation were more common with budesonide (10% and 4% respectively) than with ipratropium (8 and 0%). Sneezing occurred in 5% of those using ipratropium and in none of those using budesonide, probably reflecting the better efficacy of budesonide for control of sneezing. Overall both drugs were well tolerated.
Is ipratropium bromide a selective bronchodilator?
Ipratropium bromide is a quaternary ammonium derivative of atropine that when administered by inhalation into the airways is poorly absorbed into the circulation and can be used as a selective bronchodilator. From: Assisted Ventilation of the Neonate (Sixth Edition), 2017.
Is an aerosol nebulizer safe?
Therapeutic doses from an aerosol are free of systemic adverse effects because of the very low blood concentration after inhalation. Transient dryness of the mouth and scratching in the trachea can occur in up to 25% of patients receiving wet nebulizer treatment with the drug.
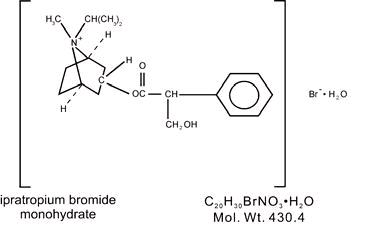
What is ipratropium bromide?
Ipratropium bromide ( N -isopropyl-notropine-tropic acid ester methylbromide), also known as Sch 1000, is an atropine- like agent which has been developed as an aerosol bronchodilator drug. This compound is believed to produce bronchodilatation by inhibiting increases in levels of cyclic guanosine monophosphate mediated by cholinergic receptors, ...
How long does it take for ipratropium bromide to improve FEV 1?
In the group with bronchial asthma, all doses of ipratropium and isoproterenol produced significant (P < 0.05) improvements in the FEV 1, compared with the placebo, within five minutes of administration. The duration of significant improvement in FEV 1 was less than 15 minutes after the 10µg dose of ipratropium bromide; it was 30 minutes after the 20µg dose of ipratropium bromide and after 75µg of isoproterenol, one hour for 150µg of isoproterenol, and up to four hours after the two higher doses of ipratropium ( Fig 1 ).
How many doses of isoproterenol were given?
All individuals received a total of seven treatments with an interval of at least 24 hours between each. Isoproterenol was administered in doses of 75µg and 150µg ; ipratropium bromide was administered in doses of 10µg, 20µg, 40µg, and 80µg. These two bronchodilator drugs, as well as a placebo, were given in random order, except that the doses of ipratropium were given in increasing strengths. In order to exclude diurnal variations, all treatments were given in the early morning. On each day of study, a coded aerosol was administered only if the baseline FEV 1 was within 10 percent of the qualifying baseline FEV 1. Otherwise, a test dose was not given on that day. Spirometric studies (using the Air-Shields Pulmonary Function Recorder) were performed 5, 15, 30, 60, 120, 180, 240, 300, and 360 minutes after the dose was administered. From the best of the three tracings obtained at each interval, the forced vital capacity (FVC), FEV 1 and the mean forced expiratory flow between 200 and 1,200 ml of the FVC (FEF 200-1200) were calculated. At the time of each spirometric test, the pulse and blood pressure were also recorded. A physical examination, as well as a complete blood cell count, urinalysis, and determinations of blood chemistry, were performed upon entering and concluding the study. Patients were encouraged to report any side effects.
What is an atropine derivative?
An atropine derivative as a metered dose aerosol for inhalation therapy on bronchial asthma. Inhalation of a new anticholinergic drug, Sch 1000, in asthma and chronic bronchitis: Effect on airway resistance, thoracic gas volume, blood gases and exercise-induced asthma.
How long does isoproterenol affect FEV?
In the group with chronic bronchitis or chronic asthmatic bronchitis, the values for FEV 1 and FVC were significantly increased within five minutes after all doses of the active compounds ( Table 3 ). Both doses of isoproterenol remained effective at a significant level for one hour. The three lower doses of ipratropium had significant effects for three to four hours, while the 80 µ g dose significantly increased both the FEV 1 and FVC for five hours. The 80 µ g dose of ipratropium bromide had the largest and most prolonged increase ( Fig 2 and 3 ).
What is the role of atropine in bronchial obstruction?
The role of atropine as a mediator blocker of induced bronchial obstruction.
Is ipratropium bromide a bronchodilator?
Ipratropium bromide (also known as Sch 1000) is a new atropine-like bronchodilator drug whose mechanism of action is via an anticholinergic pathway and may decrease cyclic guanosine monophosphate. Although of established efficacy in asthma, there are no studies of the use of ipratropium in patients with chronic bronchitis.
How long does it take for ipratropium to peak?
Serial FEV1 measurements demonstrate that the median time to onset of ipratropium inhalation (i.e., a 15% increase in FEV1) is 15 to 30 minutes and the median time to peak FEV1 is 1 to 2 hours. The median duration of effect as measured by FEV1 is 4 to 5 hours. Nasal Spray.
What is quaternary amine?
Quaternary amine; short-acting antimuscarinic agent; available as an oral inhaler, nebulized solution, or nasal spray. Inhalations used primarily in adults for maintenance treatment of COPD; used in adult and pediatric patients for acute asthma exacerbation off-label.
How long does it take to inhale 136 mg of sab?
Adults. 136 mcg (8 actuations of 17 mcg/actuation) via oral inhalation every 20 minutes as needed for up to 3 hours has been recommended for severe asthma exacerbation in the emergency care setting. Used in addition to a short-acting beta-agonist (SABA).
Is ipratropium in milk?
There are no data regarding the presence of ipratropium in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. Although lipid-insoluble quaternary cations pass into breast milk, ipratropium concentrations in plasma after inhaled therapeutic doses are low; therefore, ipratropium levels in human breast milk are expected to be low. Consider the benefits of breast-feeding, the risk of potential infant drug exposure, and the risk of an untreated or inadequately treated condition. The National Asthma Education and Prevention Program (NAEPP) Asthma and Pregnancy Working Group recommend the use of ipratropium in the lactating mother as an additional therapy in severe exacerbations. Caution is advised when administering ipratropium nasal spray to a lactating woman for allergic rhinitis.
Is ipratropium safe for cardiac arrhythmias?
Use ipratropium with caution in patients at risk for cardiac arrhythmias. In a cohort of 283,429 asthmatics (age range: 5—24 years) with no history of arrhythmia or congenital heart disease, active use of an inhaled anticholinergic was associated with a 1.56-fold increase in arrhythmia risk compared to non-use or non-active use (adjusted OR 1.56, 95% CI 1.08—2.25). Risk was highest among active users of ipratropium; however, tiotropium sample size was limited. In addition, risk was increased in those receiving high-dose therapy (defined as > 0.114 mg of ipratropium equivalents; adjusted OR 1.69, 95% CI 1.1—2.59) and monotherapy (adjusted OR 1.59, 95% CI 1.08—2.33) compared to those receiving low-dose therapy (defined as <= 0.114 mg ipratropium equivalents; adjusted OR 1.22, 95% CI 0.53—2.65) and combination ipratropium/short-acting beta agonist therapy (adjusted OR 1.2, 95% CI 0.74—1.94), respectively.
Can ipratropium cause urinary retention?
Anticholinergics should be used with caution in patients with preexisting bladder obstruction (of the bladder neck) or other urinary tract obstruction, or in patients with prostatic hypertrophy. Ipratropium may precipitate urinary retention in patients with these conditions. Although inhaled ipratropium is only minimally absorbed into the systemic circulation, the effects of ipratropium may be additive to other concomitantly administered anticholinergic medications.
Is ipratropium bromide contraindicated?
Ipratropium is a derivative of and structurally similar to atropine; as such, use is contraindicated in patients with atropine hypersensitivity or atropine derivative hypersensitivity. Do not use ipratropium bromide products in those with ipratropium bromide hypersensitivity. In addition, ipratropium aerosols can produce a paradoxical bronchospasm that can be life-threatening in some patients. This rare problem, when it occurs, is usually seen with the first inhalation from a newly opened canister. Prescribers and patients should be aware of this precaution. It is recommended to 'test-spray' three times before using a new canister for the first time. If acute bronchospasm occurs during use, the ipratropium inhalation should be discontinued immediately and appropriate treatment measures instituted. In rare cases, severe allergic reactions, including urticaria, angioedema, oropharyngeal edema, respiratory difficulty, and anaphylaxis have occurred after the use of ipratropium.

Ipratropium Description
Ipratropium - Clinical Pharmacology
- Ipratropium Bromide Inhalation Solution is an anticholinergic (parasympatholytic) agent that, based on animal studies, appears to inhibit vagally-mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released from the vagus nerve. Anticholinergics prevent the increases in intracellular concentration of cyclic guanosine monophosphate (cyclic GMP) which …
Indications and Usage For Ipratropium
- Ipratropium Bromide Inhalation Solution administered either alone or with other bronchodilators, especially beta adrenergics, is indicated as a bronchodilator for maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease, including chronic bronchitis and emphysema.
Contraindications
- Ipratropium bromide is contraindicated in known or suspected cases of hypersensitivity to Ipratropium bromide, or to atropine and its derivatives.
Warnings
- The use of Ipratropium Bromide Inhalation Solution as a single agent for the relief of bronchospasm in acute COPD exacerbation has not been adequately studied. Drugs with faster onset of action may be preferable as initial therapy in this situation. Combination of Ipratropium Bromide Inhalation Solution and beta agonists has not been shown to be more effective than eit…
Precautions
- General
Ipratropium bromide should be used with caution in patients with narrow angle glaucoma, prostatic hypertrophy or bladder neck obstruction. - Information for Patients
Patients should be advised that mydriasis, temporary blurring of vision, precipitation or worsening of narrow-angle glaucoma or eye pain may result if the solution comes into direct contact with the eyes. Use of a nebulizer with mouthpiece rather than face mask may be preferable, to reduce th…
Adverse Reactions
- Adverse reaction information concerning Ipratropium Bromide Inhalation Solution is derived from 12-week active-controlled clinical trials. Additional information is derived from foreign postmarketing experience and the published literature. All adverse events, regardless of drug relationship, reported by three percent or more patients in the 12-week controlled clinical trials a…
Overdosage
- Acute systemic overdosage by inhalation is unlikely since Ipratropium bromide is not well absorbed after inhalation at up to four-fold the recommended dose, or after oral administration at up to forty-fold the recommended dose. The oral LD50of Ipratropium bromide ranged between 1,001 and 2,010 mg/kg in mice; between 1,667 and 4,000 mg/kg in rats; and between 400 and 1,…
Ipratropium Dosage and Administration
- The usual dosage of Ipratropium Bromide Inhalation Solution is 500 mcg (1 Unit-Dose Vial) administered three to four times a day by oral nebulization, with doses 6 to 8 hours apart. Ipratropium Bromide Inhalation Solution Unit-Dose Vials contain 500 mcg Ipratropium bromide, USP anhydrous in 2.5 mL normal saline. Ipratropium Bromide Inhalation Solution can be mixed i…
How Is Ipratropium Supplied
- Ipratropium Bromide Inhalation Solution, USP is a clear, colorless solution supplied in a unit-dose vial containing 2.5 mL. Supplied in cartons as listed below: NDC 47335-706-49 carton of 25 vials (5 vials per foil pouch) NDC 47335-706-52 carton of 30 vials (5 vials per foil pouch) NDC 47335-706-54 carton of 60 vials (5 vials per foil pouch) Each vial is made from a low density polyethyle…