
Why is nitrogen a gas whereas phosphorus is a solid?
Why nitrogen is gas while phosphorus is solid? Nitrogen is gas but phosphorus is a solid because Nitrogen pairs up, into the N2 molecule which has a very strong triple bond. In phosphorus, the atoms do not pair up, they bond to their neighboring atoms, thus forming a network of bonding that holds the atoms together into a solid.
What are facts about phosphorus?
Facts About Phosphorus. Phosphorus is in Period 3 and Group 15 of the periodic table. It has 15 protons, so it has the atomic number 15. This also means it is the 15th element on the periodic table.
Why does my body need phosphorus?
You need phosphorus to:
- keep your bones strong and healthy
- help make energy
- move your muscles
Is strontium a liquid a solid or a gas?
Strontium is the chemical element with the symbol Sr and atomic number 38. An alkaline earth metal, strontium is a soft silver-white yellowish metallic element that is highly chemically reactive. The metal forms a dark oxide layer when it is exposed to air.
See more
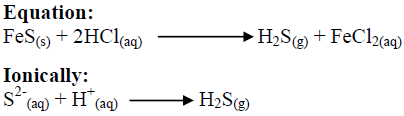
Can phosphorus be a gas?
Phosphorus can be found on earth in water, soil and sediments. Unlike the compounds of other matter cycles phosphorus cannot be found in air in the gaseous state. This is because phosphorus is usually liquid at normal temperatures and pressures.
Is a phosphorus a solid?
The two main forms of phosphorus are white phosphorus and red phosphorus. White phosphorus is a poisonous waxy solid and contact with skin can cause severe burns. It glows in the dark and is spontaneously flammable when exposed to air. Red phosphorus is an amorphous non-toxic solid.
What state of matter is phosphorus?
Phosphorus is a chemical element with symbol P and atomic number 15. Classified as a nonmetal, Phosphorus is a solid at room temperature.
What are properties of phosphorus?
It is colorless and transparent in its pure form. Phosphorus is insoluble in water, but soluble in carbon disulfide. Phosphorus burns spontaneously in air to its pentoxide. It is highly poisonous, with a lethal dose of ~50 mg.
Why is phosphorus a solid?
In phosphorus, the atoms do not pair up, they bond to their neighboring atoms, thus forming a network of bonding that holds the atoms together into a solid.
Is phosphorus a solid liquid or gas at room temperature?
solidphosphorus (P), nonmetallic chemical element of the nitrogen family (Group 15 [Va] of the periodic table) that at room temperature is a colourless, semitransparent, soft, waxy solid that glows in the dark.
Is phosphorus a gas in its standard state?
4:185:36Aleks Standard chemical and physical states of the elements - YouTubeYouTubeStart of suggested clipEnd of suggested clipThen you have phosphorus which is p4. It's a solid and sulfur which is s8. It's a solid thenMoreThen you have phosphorus which is p4. It's a solid and sulfur which is s8. It's a solid then everything else is going to be a solid.
Is Potassium a gas?
Potassium is a chemical element with symbol K and atomic number 19. Classified as an alkali metal, Potassium is a solid at room temperature.
What is phosphorus made of?
The world phosphorus comes from a Greek word meaning "bearer of light," and this element delivers on that promise. The most common forms are white phosphorus, made up of phosphorus atoms arranged like a tetrahedron (a four-sided pyramid), and red phosphorus, a solid but non-crystalline form of the element.
What is liquid phosphorus?
Pro-Germinator (PrG) is a liquid phosphorus fertilizer supplying nitrogen, potassium, and iron in addition to phosphate phosphorus. This combination can supply your crop's complete phosphorus needs in a single application, early in the season.
Is phosphorus heavier than water?
Density approximately 15.2 lb / gal. Phosphorus, white, molten appears as a white or yellow colored semi-liquid. Transported at high temperatures. Insoluble in water and denser than water.
Is phosphorus an acid?
Phosphorus is a soft base so the complexes most often involve a soft metal atom or ion.
What is liquid phosphorus?
Pro-Germinator (PrG) is a liquid phosphorus fertilizer supplying nitrogen, potassium, and iron in addition to phosphate phosphorus. This combination can supply your crop's complete phosphorus needs in a single application, early in the season.
Is potassium a solid liquid or gas?
solidPotassium is a chemical element with symbol K and atomic number 19. Classified as an alkali metal, Potassium is a solid at room temperature.
Why is phosphorus called the devil's element?
Some texts refer to phosphorus as the "Devil's Element" because of its eerie glow, tendency to burst into flame, and because it was the 13th known element. Like other nonmetals, pure phosphorus assumes markedly different forms. There are at least five phosphorus allotropes.
What are the types of phosphorus?
Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Earth. It has a concentration in the Earth's crust of about one gram per kilogram (compare copper at about 0.06 grams).
Overview
Characteristics
Phosphorus has several allotropes that exhibit strikingly diverse properties. The two most common allotropes are white phosphorus and red phosphorus.
From the perspective of applications and chemical literature, the most important form of elemental phosphorus is white phosphorus, often abbreviated as WP. It is a soft, waxy solid which consists of tetrahedral P 4 molecules, in which eac…
Occurrence
In 2013, astronomers detected phosphorus in Cassiopeia A, which confirmed that this element is produced in supernovae as a byproduct of supernova nucleosynthesis. The phosphorus-to-iron ratio in material from the supernova remnant could be up to 100 times higher than in the Milky Way in general.
In 2020, astronomers analysed ALMA and ROSINA data from the massive star-forming region AFG…
Compounds
The most prevalent compounds of phosphorus are derivatives of phosphate (PO4 ), a tetrahedral anion. Phosphate is the conjugate base of phosphoric acid, which is produced on a massive scale for use in fertilisers. Being triprotic, phosphoric acid converts stepwise to three conjugate bases:
H3PO4 + H2O ⇌ H3O + H2PO4 Ka1 = 7.25×10
History
The name Phosphorus in Ancient Greece was the name for the planet Venus and is derived from the Greek words (φῶς = light, φέρω = carry), which roughly translates as light-bringer or light carrier. (In Greek mythology and tradition, Augerinus (Αυγερινός = morning star, still in use today), Hesperus or Hesperinus (΄Εσπερος or Εσπερινός or Αποσπερίτης = evening star, still in us…
Production
In 2017, the USGS estimated 68 billion tons of world reserves, where reserve figures refer to the amount assumed recoverable at current market prices; 0.261 billion tons were mined in 2016. Critical to contemporary agriculture, its annual demand is rising nearly twice as fast as the growth of the human population. The production of phosphorus may have peaked before 2011 and some scienti…
Applications
Phosphorus is an essential plant nutrient (the most often limiting nutrient, after nitrogen), and the bulk of all phosphorus production is in concentrated phosphoric acids for agriculture fertilisers, containing as much as 70% to 75% P2O5. That led to large increase in phosphate (PO4 ) production in the second half of the 20th century. Artificial phosphate fertilisation is necessary because phosphorus is essential to all living organisms; it is involved in energy transfers, strengt…
Biological role
Inorganic phosphorus in the form of the phosphate PO 4 is required for all known forms of life. Phosphorus plays a major role in the structural framework of DNA and RNA. Living cells use phosphate to transport cellular energy with adenosine triphosphate (ATP), necessary for every cellular process that uses energy. ATP is also important for phosphorylation, a key regulatory event in cells. Phospholipids are the main structural components of all cellular membranes. Calciu…