
Pyruvate is an important "hub" metabolite that is a precursor for amino acids, sugars, cofactors, and lipids in extant metabolic networks. Pyruvate has been produced under simulated hydrothermal vent conditions from alkyl thiols and carbon monoxide in the presence of transition metal sulfides at 250 °C [Cody GD et al. (2000) Science 289(5483):1337-1340], so it is plausible that pyruvate was formed in hydrothermal systems on the early earth.
What are the 5 Universal metabolites of metabolism?
This dynamic process occurs through the intermediacy of just five universal metabolites made of carbon, hydrogen and oxygen: acetate, pyruvate, oxaloacetate, succinate and α-ketoglutarate 1.
Which intermediates carry the 13 C label from pyruvate?
We found the 13 C label present in pyruvate to be carried over to the following intermediates: levulinate, malonate, malate, fumarate, succinate, α-ketoglutarate, hydroxyketoglutarate/ oxopentenedioate, isocitrate, aconitate, tricarballylate and acetate.
What is the reaction between pyruvate and glyoxylate?
We discuss some of the key transformations here. An aldol reaction between pyruvate and glyoxylate gives hydroxyketoglutarate, followed by dehydration to oxopentenedioate and reduction to α-ketoglutarate.
Is there a universal metabolic precursor without phosphorus?
A recent theoretical analysis of all known metabolic reactions revealed a robust hypothetical metabolic network, containing all five of the universal metabolic precursors, that does not rely on phosphorus or on phosphorus-containing co-factors such as ATP 23. Of the molecules made up only of carbon, hydrogen and oxygen within this hypothetical p...
Is pyruvate a metabolite?
It acts as the main metabolite and cofactor. Pyruvate is commonly seen as one of the last products of glycolysis where a couple of its molecules can be produced by the relative breakdown of a single glucose molecule.
Is pyruvate a metabolite of glycolysis?
Pyruvate is an important chemical compound in biochemistry. It is the output of the anaerobic metabolism of glucose known as glycolysis.
Why is pyruvate a central metabolite?
Pyruvate occupies a central metabolic node by virtue of its position at the crossroads of glycolysis and the tricarboxylic acid (TCA) cycle and its production and fate being governed by numerous cell-intrinsic and extrinsic factors.
Is pyruvate a precursor for glucose?
Gluconeogenesis is the pathway by which glucose is synthesized from non-carbohydrate metabolites. The principal gluconeogenic precursors are pyruvate and lactate, certain gluconeogenic amino acids, and glycerol, which is derived mainly from fat metabolism.
What is pyruvate converted to?
Pyruvate—three carbons—is converted to acetyl CoA, a two-carbon molecule attached to coenzyme A. A molecule of coenzyme A is a necessary reactant for this reaction, which releases a molecule of carbon dioxide and reduces a NAD+ to NADH.
How can pyruvate be metabolized?
Pyruvate is metabolized by three major routes in animal tissues. It can be oxidized through the citrate cycle with concomitant synthesis of ATP. It can be metabolized to fatty acids, and it can also be converted into glucose.
Why acetyl CoA is called common metabolite?
Acetyl-CoA represents a key node in metabolism due to its intersection with many metabolic pathways and transformations. Emerging evidence reveals that cells monitor the levels of acetyl-CoA as a key indicator of their metabolic state, through distinctive protein acetylation modifications dependent on this metabolite.
What is the metabolic fate of pyruvate after glycolysis?
After glycolysis, it can be converted to acetyl- CoA, which has numerous metabolic destinations, including the TCA cycle. Pyruvate can also be converted into lactate, which enters the Cori cycle in absence of mitochondria or oxygen.
Why is pyruvate converted to lactic acid?
Our muscle cells can undergo this process when they are in oxygen debt. If enough oxygen is not present to undergo aerobic respiration, pyruvate will undergo lactic acid fermentation.
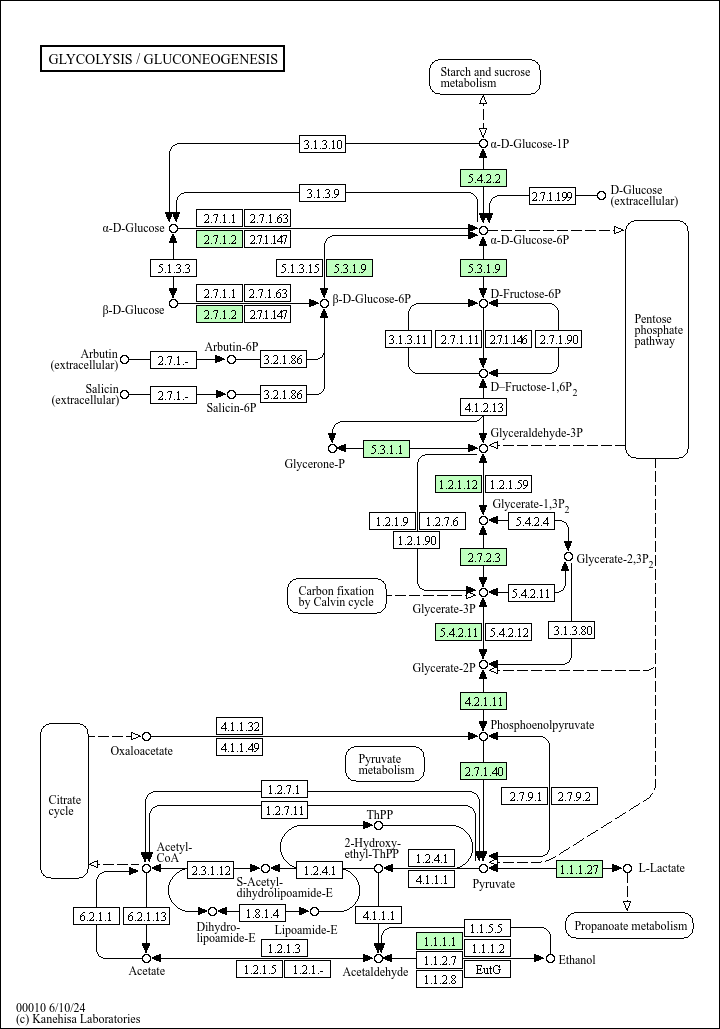
What are the precursors of glycolysis?
The intermediates of glycolysis, such as glucose 6-phosphate, fructose 6-P, glyceraldehyde 3-P, dihydroxyacetone-P, glycerate 3-P can be used as precursors for the synthesis of cell building material, such as nucleotides, amino acids, phospholipids and triglycerides.
What are the precursors of glucose?
In humans, lactate is probably the most important glucose precursor, especially during exercise. Others, in order of importance, are alanine, pyruvate, glycerol, and some glucogenic amino acids, including glutamate.
Which of the following is not the precursor of gluconeogenesis?
Which of the following is not the precursor of gluconeogenesis? Explanation: Only leucine or lysine is the substrate which is not used for gluconeogenesis as these amino acids produce only acetyl-CoA upon degradation. Animals cannot carry out gluconeogenesis by two acetyl carbon of acetyl-CoA.
Which is not a product of glycolysis?
Which of the following are not produced during the reactions of glycolysis? Explanation: The correct answer to this question is carbon dioxide. Carbon dioxide is not produced during glycolysis.
What happens to pyruvate after glycolysis?
In eukaryotic cells, the pyruvate molecules produced at the end of glycolysis are transported into mitochondria, which are the sites of cellular respiration. There, pyruvate will be transformed into an acetyl group that will be picked up and activated by a carrier compound called coenzyme A (CoA).
What is the product of glycolysis?
Pyruvic acidGlycolysis is defined as a sequence of reactions that convert glucose into pyruvate or lactate with the production of ATP (Adenosine triphosphate). Pyruvic acid is the end product of glycolysis. Two molecules of pyruvic acid are generated by the partial oxidation of one glucose molecule.
What is the fate of pyruvate after glycolysis?
Glycolysis is the process by which glucose is broken down within the cytoplasm of a cell to form pyruvate. Under aerobic conditions, pyruvate can diffuse into mitochondria, where it enters the citric acid cycle and generates reducing equivalents in the form of NADH and FADH2.
What is the reaction between pyruvate and glyoxylate?
We discuss some of the key transformations here. An aldol reaction between pyruvate and glyoxylate gives hydroxyketoglutarate, followed by dehydration to oxopentenedioate and reduction to α-ketoglutarate. The three intermediates and products of this sequence partially undergo oxidative decarboxylation to give malate, fumarate and succinate, respectively (Supplementary Figs. 4, 5 and 13 ). We found these oxidative processes to be more efficient in the presence of ferric iron (Fe 3+) than Fe 2+ —the former presumably being generated during the reduction of oxopentendioate (Supplementary Fig. 18 ). α-Ketoglutarate also undergoes an aldol/oxidative decarboxylation sequence to give isocitrate. Reminiscent of the glyoxylate cycle (or ‘glyoxylate shunt’), isocitrate slowly undergoes an Fe 2+ -catalysed retro-aldol reaction to return small quantities of glyoxylate and succinate, but can also slowly dehydrate to give aconitate (Supplementary Fig. 14 ). Trace amounts of fumarate are observed upon heating succinate under standard conditions, pointing to mechanistic redundancy within the network. Additional evidence for the occurrence of oxidation under the reaction conditions was our observation that malate produced a small amount of acetate, which presumably arises from oxidative decomposition of oxaloacetate (Supplementary Fig. 19 ). Cannizzaro-type disproportionation of glyoxylate results in the formation of glycolate and oxalate, the latter of which slowly breaks down to CO 2 under the reaction conditions (Supplementary Figs. 13 and 14 ). A prolonged reaction time (more than 24 hours) results in a slow accumulation of other thermodynamic end-products, including malonate, levulinate, succinate, malate and isocitrate (Supplementary Fig. 4 ). It is plausible that such a network could be kept in a nonequilibrium steady state by continuous input of pyruvate and glyoxylate, both of which can be produced abiotically 2, 3, 4, 19.
What are the five universal metabolites?
This dynamic process occurs through the intermediacy of just five universal metabolites made of carbon, hydrogen and oxygen: acetate, pyruvate, oxaloacetate, succinate and α-ketoglutarate 1. These five compounds are found directly on or near life’s core anabolic and catabolic pathways (which, respectively, build up and break down complex ...
What are the two main components of a metabolic network?
Of the molecules made up only of carbon, hydrogen and oxygen within this hypothetical phosphorus-free network, the two that represent the biggest branching points are pyruvate and glyoxylate, suggesting that a primitive pre-ATP metabolism, if it existed, would have relied on these two compounds. Pyruvate and glyoxylate are attractive as starting materials for prebiotic chemistry because they can be accessed through abiotic CO 2 fixation 2, 3, 19 as well as by other plausible means 4, 24, yet their reactivity with each other has hardly been studied with regards to the origins of metabolism. A lone example used the strong oxidant hydrogen peroxide to drive a bicyclic reaction network containing two other TCA-cycle intermediates 25, but the influence of transition metal ions as potential naturally occurring catalysts has not yet been explored in this context. Here we show that pyruvate and glyoxylate spontaneously produce a reaction network in warm iron-rich water that recapitulates most of the intermediates and reactions of core biological pathways such as the Krebs cycle and glyoxylate cycle, in addition to amino-acid biosynthesis.
How are carboxylic acids quantified?
Carboxylic acids and amino acids were quantified according to a literature procedure 9, using six-point calibration curves prepared from ECF/ethanol-derivatized solutions of authentic samples (0.006 M, 0.013 M, 0.020 M, 0.027 M, 0.033 M and 0.040 M) as described above. Each data point was obtained from three independent measurements and the correlation line was obtained from the least-squares fitting (intercept = 0). Error bars on graphs are shown as plus/minus standard deviation (s.d.) for each data point. For the linear fits, the overall percentage error of the response factor corresponds to ± s.d. for each slope value. For the polynomial fits, 95% confidence envelopes are shown.
How to determine acetate and formate?
Formate and acetate were determined using an NMR procedure and a GC–MS procedure , both reported previously 2. For the NMR procedure, to a 1.5 ml plastic microtube we added roughly 1 ml of the reaction mixture and about 50 mg solid KOH (Merck EMSURE). The resulting thick suspension was centrifuged at 10,000 r.p.m. for 20 min. To 600 µl of the supernatant we added 100 µl of a 0.05 M solution of internal standard (4,4,-dimethyl-4-silapentane-1-sulfonic acid (DSS) in D 2 O). The solution was analysed by NMR using the Bruker ZGESGP pulse program, as described above. For the GC–MS detection of formate and acetate as their amides with N -methylphenylethylamine, to a 120 μl aliquot of a reaction mixture in a 1.5 ml plastic microtube we added 50 μl of a 0.12 M solution of 1-hydroxybenzotriazole in H 2 O, 75 μl 0.08 M 1-ethyl-3- (3-dimethyl-aminopropyl)carbodiimide solution (EDC) in acetonitrile, and 75 μl 0.06 M N -methylphenylethylamine (MPEA) in acetonitrile. The resulting mixture was vortex mixed for 30 s and incubated at 60 °C for 45 min. After cooling to room temperature, 200 μl of CHCl 3 was added and the mixture was vortex mixed for 30 s. The CHCl 3 layer was removed and dried over anhydrous magnesium sulfate. We added 50 μl of the CHCl 3 supernatant to a vial together with 150 μl of ethyl acetate, and analysed this by GC–MS.
How to identify reaction products derivatized to ethyl esters?
We identified reaction products derivatized to ethyl esters by comparing the mass spectra and retention times with those of analogously derivatized authentic samples, as shown below and described elsewhere 9.
How many metabolites does life have?
Life builds its molecules from carbon dioxide (CO 2) and breaks them back down again through the intermediacy of just five metabolites, which are the universal hubs of biochemistry 1. However, it is unclear how core biological metabolism began and why it uses the intermediates, reactions and pathways that it does.
