
Precautions
It is in the anticoagulant class of drugs. This activity reviews the indications, action, and contraindications for warfarin as a valuable agent in the prophylaxis and treatment of myocardial infarction, deep vein thrombosis, pulmonary embolism, and atrial fibrillation.
What class of drug is warfarin?
Canrenoic acid may increase the excretion rate of Warfarin which could result in a lower serum level and potentially a reduction in efficacy. Warfarin may increase the anticoagulant activities of Capecitabine.
Canrenoic acid and warfarin interact?
Warfarin has numerous drug interactions that can increase the risk of adverse effects or decrease warfarin's anticoagulant effect. Therefore dosing adjustments, close monitoring, and alternative agents should be considered when combining warfarin with certain medications.
How does warfarin interact with other medications?
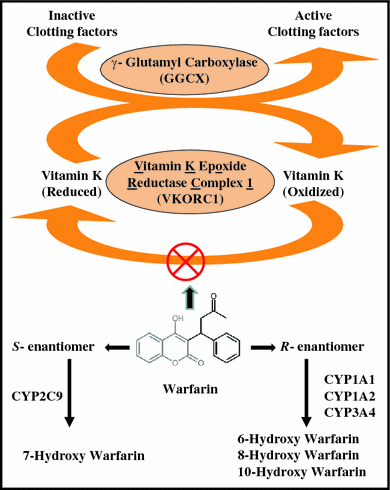
While warfarin is one of several drugs popularly referred to as a "blood thinner", this is a misnomer since it does not affect the viscosity of blood. Warfarin inhibits the vitamin K -dependent synthesis of biologically active forms of the clotting factors II, VII, IX and X, as well as the regulatory factors protein C, protein S, and protein Z.
Does warfarin affect the viscosity of blood?
What is the pH of warfarin?
Clinical Study. The average concentration of warfarin in the oral fluid was 2.5±1.6 ng/mL, with values ranging from 0.8 to 7.6 ng/mL, whereas the pH of the oral fluid was 6.7±0.4 in the range 6.0–7.5 (Fig.
Are medications basic or acidic?
As has been stated before, most of the drugs used in medicine behave in solution as weak acids, weak bases, or sometimes as both weak acids and weak bases.
What type of chemical is warfarin?
Warfarin is a synthetic anticoagulant. Warfarin inhibits the regeneration of vitamin K1 epoxide and so the synthesis of vitamin K dependent clotting factors, which include Factors II, VII, IX and X, and the anticoagulant proteins C and S.
What is chemical properties of warfarin?
SUMMARY AND EVALUATION 2.1 Identity, Physical and Chemical Properties, and Analytical Methods Warfarin is a colourless, crystalline compound with a melting point of 151-161°C. It is practically insoluble in water, readily soluble in acetone and dioxane, and moderately soluble in alcohols.
What are acidic medications?
They include:Dexlansoprazole (Dexilant)Esomeprazole (Nexium)Lansoprazole (Prevacid)Omeprazole (Prilosec and another with sodium bicarbonate, Zegerid)Pantoprazole (Protonix)Rabeprazole (Aciphex)
Is aspirin a basic or acidic drug?
Aspirin is a commonly used medicine for primary and secondary prevention of cardiovascular diseases. It is an acidic medicine associated with gastric irritation and acid reflux, which in turn can lead to low oral pH levels.
Is warfarin a salt?
The sodium salt form of warfarin, a coumarin and a vitamin K antagonist, with anticoagulant activity. Warfarin sodium inhibits both vitamin K and vitamin K epoxide reductases, thereby interfering with the cyclic interconversion of vitamin K epoxide to its reduced form, vitamin KH2.
What is warfarin made of?
Warfarin is derived from coumarin, a sweet-smelling anticoagulant (blood-clotting) chemical found naturally in sweet clover and many other plants. In 1954, warfarin was approved for clinical use and has remained a popular anticoagulant ever since.
What is warfarin an example of?
Warfarin belongs to a class of drugs called anticoagulants. A class of drugs is a group of medications that work in a similar way. These drugs are often used to treat similar conditions. Warfarin works by stopping your body from forming blood clots.
What are the chemical properties of K?
Chemical properties of potassium - Health effects of potassium - Environmental effects of potassiumAtomic number19Density0.86 g.cm -3 at 0 °CMelting point63.2 °CBoiling point760 °CVanderwaals radius0.235 nm7 more rows
Why is it called warfarin?
The name warfarin is derived from WARF (Wisconsin Alumni Research Foundation) and –arin from coumarin. Warfarin is now the most widely used anticoagulant in the world.
What are the two types of warfarin?
There are 2 two brands of warfarin available; Marevan and Coumadin. Marevan and Coumadin are not the same. Always use the same brand unless your doctor specifically tells you to change. They come in different tablet strengths and colours.
What are medications classified as?
The 4 Categories of MedicationGeneral Sales List (GSL) GSLs are a type of medicine that have few legal restrictions. ... Pharmacy Medicines. Pharmacy Medicines are only available to purchase behind the counter at a pharmacy. ... Prescription Only Medicines. ... Controlled Drugs.
Are all drugs acidic?
Most drugs are weakly acidic or basic substances and are thus ionized at physiologic pH. Passive diffusion across lipophilic membranes depends on the degree of ionization.
Why most of the drugs are weak acid or base?
Most drugs are weak acids or bases that are present in solution as both the ionized and unionized forms. Ionized molecules are usually unable to penetrate lipid cell membranes because they are hydrophilic and poorly lipid soluble. Unionized molecules are usually lipid soluble and can diffuse across cell membranes.
On what basis are drugs classified?
Classification of Drugs on the basis of the Pharmacological Effect: How a drug or medicine affects or influences the cells of an organism is referred to as the pharmacological effect. Different types of drugs have various pharmacological effects on an organism.
What causes purple toes?
Purple toe syndrome is a complication characterized by cholesterol microembolization that causes purple lesions to develop on the toes and sides of the feet. Purple toe syndrome usually develops 3 to 8 weeks after the initiation of warfarin therapy. Warfarin-induced skin necrosis is a serious condition in which the subcutaneous tissue necrosis occurs due to an acquired protein C deficiency following treatment with warfarin. The risk of necrosis increases in patients with protein C or protein S deficiency. The skin necrosis usually occurs within the first week of therapy and management strategies, including discontinuing treatment with warfarin, administering fresh frozen plasma and vitamin K, and initiating anticoagulation therapy with either unfractionated heparin or low molecular weight heparin. Calciphylaxis or calcium uremic arteriolopathy is another rare adverse effect that can occur in patients with or without end-stage renal disease.
What is the composition of Warfarin?
Drug Composition: Warfarin is a racemic mixture composed of R and S enantiomers of the drug. Each enantiomer gets metabolized through unique pathways, and the S-enantiomer is approximately three to five times more potent than the R-enantiomer.
What is the best way to monitor warfarin?
Patients receiving treatment with warfarin should have close monitoring to ensure the safety and efficacy of the medication. Periodic blood testing is recommended to assess the patient’s prothrombin time (PT) and the international normalized ratio (INR).
What is an unsupervised patient?
Unsupervised patients with conditions associated with a high potential of non-adherence
What is an active ulcer?
Active ulceration or overt bleeding of the gastrointestinal, genitourinary, or respiratory tract
What is a hemorrhagic tendency?
Hemorrhagic tendencies (e.g., active GI ulceration, patients bleeding from the GI, respiratory, or GU tract ; a cerebral aneurysm; central nervous system (CNS) hemorrhage; dissecting aortic aneurysm; spinal puncture and other diagnostic or therapeutic procedures with the potential for significant bleeding)
Is warfarin an anticoagulant?
Warfarin is a medication used in the prophylaxis and treatment of venous thrombosis and thromboembolic events. It is in the anticoagulant class of drugs . This activity reviews the indications, action, and contraindications for warfarin as a valuable agent in the prophylaxis and treatment of myocardial infarction, deep vein thrombosis, pulmonary embolism, and atrial fibrillation. This activity will highlight the mechanism of action, adverse event profile, pharmacokinetics, and drug interactions pertinent for members of the interprofessional team to treat patients with venous thrombosis and thromboembolic events.
What license is CAS Common Chemistry?
The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated.
What does percentage mean in hazard codes?
The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.
Is Warfarin an enantiomer?
It is an enantiomer of a (R)-warfarin. Warfarin consists of a racemic mixture of two active enantiomers—R- and S- forms—each of which is cleared by different pathways. S-warfarin is 2-5 times more potent than the R-isomer in producing an anticoagulant response.
Is H412 harmful to aquatic life?
H412 (100%): Harmful to aquatic life with long lasting effects [Hazardous to the aquatic environment, long-term hazard]
Is NCI copyright free?
Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source.
Is PDB free?
Data files contained in the PDB archive (ftp://ftp.wwpdb.org) are free of all copyright restrictions and made fully and freely available for both non-commercial and commercial use. Users of the data should attribute the original authors of that structural data.
Is the FDA website copyrighted?
Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphic s, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required.
What is the target INR for warfarin?
In particular, target INR may be 2.5–3.5 (or even 3.0–4.5) in patients with one or more mechanical heart valves.
How does warfarin kill vampire bats?
Warfarin is used to cull vampire bat populations in areas where human–wildlife conflict is a concern. Vampire bats are captured with mist nets and coated with a combination of petroleum jelly and warfarin. The bat returns to its roost and other members of the roost become poisoned as well by ingesting the warfarin after reciprocal grooming. Suspected vampire bat roosts may also be coated in the warfarin solution, though this kills other bat species and remains in the environment for years. The efficacy of killing vampire bats to reduce rabies transmission is questionable, however; a study in Peru showed that culling programs did not lead to lower transmission rates of rabies to livestock and humans.
What anticoagulant is used for warfarin?
Several types of anticoagulant drugs offering the efficacy of warfarin without a need for monitoring, such as dabigatran, apixaban, edoxaban, and rivaroxaban, have been approved in a number of countries for classical warfarin uses.
How long does Warfarin last?
A few days are required for full effect to occur, and these effects can last for up to five days. Warfarin first came into large-scale commercial use in 1948 as a rat poison. Warfarin was formally approved for human use by the US FDA to treat blood clots in 1954.
What is the best medicine for pulmonary embolism?
Warfarin, sold under the brand name Coumadin among others, is a medication that is used as an anticoagulant (blood thinner). It is commonly used to treat blood clots such as deep vein thrombosis and pulmonary embolism, and to prevent stroke in people who have atrial fibrillation, valvular heart disease or artificial heart valves. Less commonly it is used following ST-segment elevation myocardial infarction (STEMI) and orthopedic surgery. It is generally taken by mouth, but may also be used by injection into a vein.
When is warfarin given?
When warfarin (or another 4-hydroxycoumarin derivative) is given during the first trimester—particularly between the sixth and ninth weeks of pregnancy —a constellation of birth defects known variously as fetal warfarin syndrome (FWS), warfarin embryopathy, or coumarin embryopathy can occur.
What are the indications for warfarin?
Thus, common clinical indications for warfarin use are atrial fibrillation, the presence of artificial heart valves, deep venous thrombosis, and pulmonary embolism (where the embolized clots first form in veins).
How long does it take for warfarin to absorb?
Completely absorbed from the GI tract. The mean Tmax for warfarin sodium tablets is 4 hours. Label, 17, 9
What foods can you avoid with warfarin?
Avoid foods rich in vitamin K. Vitamin K in foods such as leafy vegetables can reduce warfarin efficacy.
What is the metabolic pathway of warfarin?
Metabolism of warfarin is both stereo- and regio-selective. 9 The major metabolic pathway is oxidation to various hydroxywarfarins, comprising 80-85% of the total metabolites. CYP2C9 is the major enzyme catalyzing the 6- and 7-hydroxylation of S-warfarin while 4'-hydroxylation occurs through CYP2C18 with minor contributions from CYP2C19. R-warfarin is metabolized to 4'-hydroxywarfarin by CYP2C8 with some contirbuting by CYP2C19, 6- and 8-hydroxywarfarin by CYP1A2 and CYP2C19, 7-hydroxywarfarin by CYP1A2 and CYP2C8, and lastly to 10-hydroxywarfarin by CYP3A4. The 10-hydroxywarfarin metabolite as well as a benzylic alcohol metabolite undergo an elimination step to form dehydrowarfarin. The minor pathway of metabolism is the reduction of the ketone group to warfarin alcohols, comprising 20% of the metabolites. Limited conjugation occurs with sulfate and gluronic acid groups but these metabolites have only been confirmed for R-hydroxywarfarins.
How long does it take to die from warfarin?
Doses of 1-2 mg/kg/day over a period of 15 days have been fatal in humans. 19 Warfarin overdose is primarily associated with major bleeding particularly from the mucous membranes, gastrointestinal tract, and genitourinary system. Label, 19 Epistaxis, ecchymoses, as well as renal and hepatic bleeding are also associated. These symptoms become apparent within 2-4 days of overdose although increases in prothrombin time can be observed within 24 hours. Treatment for overdosed patients includes discontinuation of warfarin and administration of [vitamin K]. For more urgent reversal of anticoagulation prothrombin complex concentrate, blood plasma, or coagulation factor VIIa infusion can be used. Label Patients can be safely re-anticoagulated after reversal of the overdose.
What is the mechanism of action of warfarin?
Mechanism of action. Warfarin is a [vitamin K] antagonist which acts to inhibit the production of vitamin K by vitamin K epoxide reductase. Label,14,16The reduced form of vitamin K, vitamin KH2is a cofactor used in the γ-carboxylation of coagulation factors VII, IX, X, and thrombin.
What is the drug bank accession number for Warfarin?
DrugBank Accession Number. DB00682. Background. Warfarin is an anticoagulant drug normally used to prevent blood clot formation as well as migration. Although originally marketed as a pesticide (d-Con, Rodex, among others), Warfarin has since become the most frequently prescribed oral anticoagulant in North America.
Can warfarin be combined with acetazolamide?
The serum concentration of Warfarin can be increased when it is combined with Acetazolamide. Acetic acid. The risk or severity of bleeding can be increased when Acetic acid is combined with Warfarin. Acetohexamide. The therapeutic efficacy of Warfarin can be increased when used in combination with Acetohexamide.
What is Q3.1 Co-Trimoxazole?
Q3.1Co-trimoxazole tablets contain sulfamethoxazole and trimetho-prim and are used in the treatment of chest and urinary tractinfections. Classify sulfamethoxazole and trimethoprim as acidic,basic or neutral and hence describe how you could separate amixture of the two drugs in the laboratory using simple glasswareand reagents. See Fig. 3.24.
What is APC tablet?
Q 1 An ‘over-the-counter’ analgesic called APC Tabletscontains aspirin, paracetamol and codeine. An extract of thesetablets was dissolved in toluene (methylbenzene) and filtered toremove insoluble solids. Devise a separation scheme to isolate allthree drugs in a pure form.
Is phenytoin an anticonvulsant?
Phenytoin is an anticonvulsant widely used in the treatment of epilepsy. Theproperties of phenytoin resemble those of barbiturates. It is a cyclic imidewith apKaof 8.3. The anion is stabilised by resonance of the negative chargeonto the oxygens of the carbonyl group and the drug is usually administeredas the sodium salt to increase water solubility (Fig. 3.15).Phenytoin and barbiturates display tautomerism of the imine–imidetype, as shown in Fig. 3.16. The predominant tautomer is the imide form,although some older textbooks list the structure of the drug as the minortautomer.
Is Indometacin an NSAID?
Indometacin is another NSAID with a similar mode of action to that ofphenylbutazone. Indometacin is acidic due to ionisation of the carboxylicacid group and has apKa value of 4.5. The nitrogen atom in indometacinis present as an amide and is essentially neutral (Fig. 3.12).
Is warfarin acidic or neutral?
Warfarin is used in the UK as the sodium salt, whichstrongly suggests that the drug is acidic, although the presence of the acidichydrogen may not be immediately apparent. The acidic hydrogen (drawnin bold in Fig. 3.7) is located between two electron-withdrawing carbonylgroups. Upon ionisation, the negative charge can be delocalised onto eachof the electronegative oxygen atoms of the dicarbonyl group to yield aresonance-stabilised anion. This enhanced stability of the anion allowswarfarin to lose a proton and renders the drug acidic with apKa of 5.0.Warfarin in the free acid form is not very soluble in water and is, therefore,always administered (and is official in theBritish Pharmacopoeia) as thesodium salt.
What is pKa in drugs?
The pKa is defined as the negative log of the dissociation constant. Where the dissociation constant is defined thus:-. Most drugs have pKa in the range 0-12, and whilst it is possible to calculate pKa it is desirable to experimentally measure the value for representative examples.
What is the pH range of the alimentary canal?
Applications like Marvin allow the user to calculate the pKa but also display the pH distribution profile, this can be valuable when thinking about absorption from the different regions of the alimentary canal where the pH ranges from 1-3 in the stomach to 7-8 in the ileum.
Where do weakly acidic drugs absorb?
64. Theoretically, weakly acidic drugs (eg, aspirin) are more readily absorbed from an acid medium (stomach) than are weakly basic drugs (eg, quinidin ). However, whether a drug is acidic or basic, much of the absorption can occur in the small intestine because the surface area is larger and membranes are more permeable.

Overview
This medication is used to treat blood clots (such as in deep vein thrombosis-DVT or pulmonary embolus-PE) and/or to prevent new clots from forming in your body.
May Treat: Cerebral thromboembolism · Deep vein thrombosis · Deep vein thrombosis with pulmonary embolism · Deep venous thrombosis · Myocardial reinfarction and more
Brand Names: Coumadin · Jantoven · Athrombin-K · Coufarin · Panwarfin and more
Drug Class: Anticoagulants - Coumarin
Availability: Prescription Required
Pregnancy: Do not use. This medication may be harmful to an unborn child.
May Treat: Cerebral thromboembolism · Deep vein thrombosis · Deep vein thrombosis with pulmonary embolism · Deep venous thrombosis · Myocardial reinfarction and more
Brand Names: Coumadin · Jantoven · Athrombin-K · Coufarin · Panwarfin and more
Drug Class: Anticoagulants - Coumarin
Availability: Prescription Required
Pregnancy: Do not use. This medication may be harmful to an unborn child.
Lactation: Does not adversely affect lactation
Alcohol: Limit intake while taking this medication
Medical uses
Contraindications
Adverse effects
Overdose
Warfarin, sold under the brand name Coumadin among others, is a medication that is used as an anticoagulant (blood thinner). It is commonly used to prevent blood clots such as deep vein thrombosis and pulmonary embolism, and to prevent stroke in people who have atrial fibrillation, valvular heart disease, or artificial heart valves. Less commonly, it is used following ST-segment elevatio…
Interactions
Warfarin is used to decrease the tendency for thrombosis, or as secondary prophylaxis (prevention of further episodes) in those individuals who have already formed a blood clot (thrombus). Warfarin treatment can help prevent formation of future blood clots and help reduce the risk of embolism (migration of a thrombus to a spot where it blocks blood supply to a vital organ).
Chemistry
All anticoagulants are generally contraindicated in situations in which the reduction in clotting that they cause might lead to serious and potentially life-threatening bleeds. This includes people with active bleeding conditions (such as gastrointestinal ulcers), or disease states with increased risk of bleeding (e.g., low platelets, severe liver disease, uncontrolled hypertension). For patients undergoing surgery, treatment with anticoagulants is generally suspended. Similarly, spinal and l…
Pharmacology
The only common side effect of warfarin is hemorrhage. The risk of severe bleeding is small but definite (a typical yearly rate of 1–3% has been reported), and any benefit needs to outweigh this risk when warfarin is considered. All types of bleeding occur more commonly, but the most severe ones are those involving the brain (intracerebral hemorrhage/hemorrhagic stroke) and the spinal cord. Risk of bleeding is increased if the INR is out of range (due to accidental or deliberate over…