
Some waived tests have potential for serious health impacts if performed incorrectly. For example, results from waived tests can be used to adjust medication dosages, such as prothrombin time testing in patients undergoing anticoagulant therapy and glucose monitoring in diabetics.
What do you need to know about waived testing?
Waived test specimens. Waived tests are approved for use only with direct, unprocessed specimens that do not require operator manipulation . Specimens that are processed or manipulated by the user (e.g., serum or plasma) require centrifugation, dilution, extraction, or other preparation steps that require special training or instrumentation and ...
What is considered a CLIA waived test?
What is a waived test? As defined by CLIA, waived tests are categorized as “simple laboratory examinations and procedures that have an insignificant risk of an erroneous result.” The Food and Drug Administration (FDA) determines which tests meet these criteria when it reviews manufacturer’s applications for test system waiver.
Is 87811 a CLIA waived test?
FDA Emergency Use Authorization, CLIAwaived CPT is 87811-QW* The QuickVue SARS Antigen Test detects SARS-CoV-2 antigens directly from anterior nares swab specimens.
What tests are required?
Tests for SARS-CoV-2 tell you if you have an infection at the time of the test. This type of test is called a “viral” test because it looks for viral infection. Antigen or Nucleic Acid Amplification Tests (NAATs) are viral tests. Tests for antibodies may tell you if you have had a past infection with the virus that causes COVID-19. Your body creates antibodies after getting infected with SARS-CoV-2 or after getting vaccinated against COVID-19.
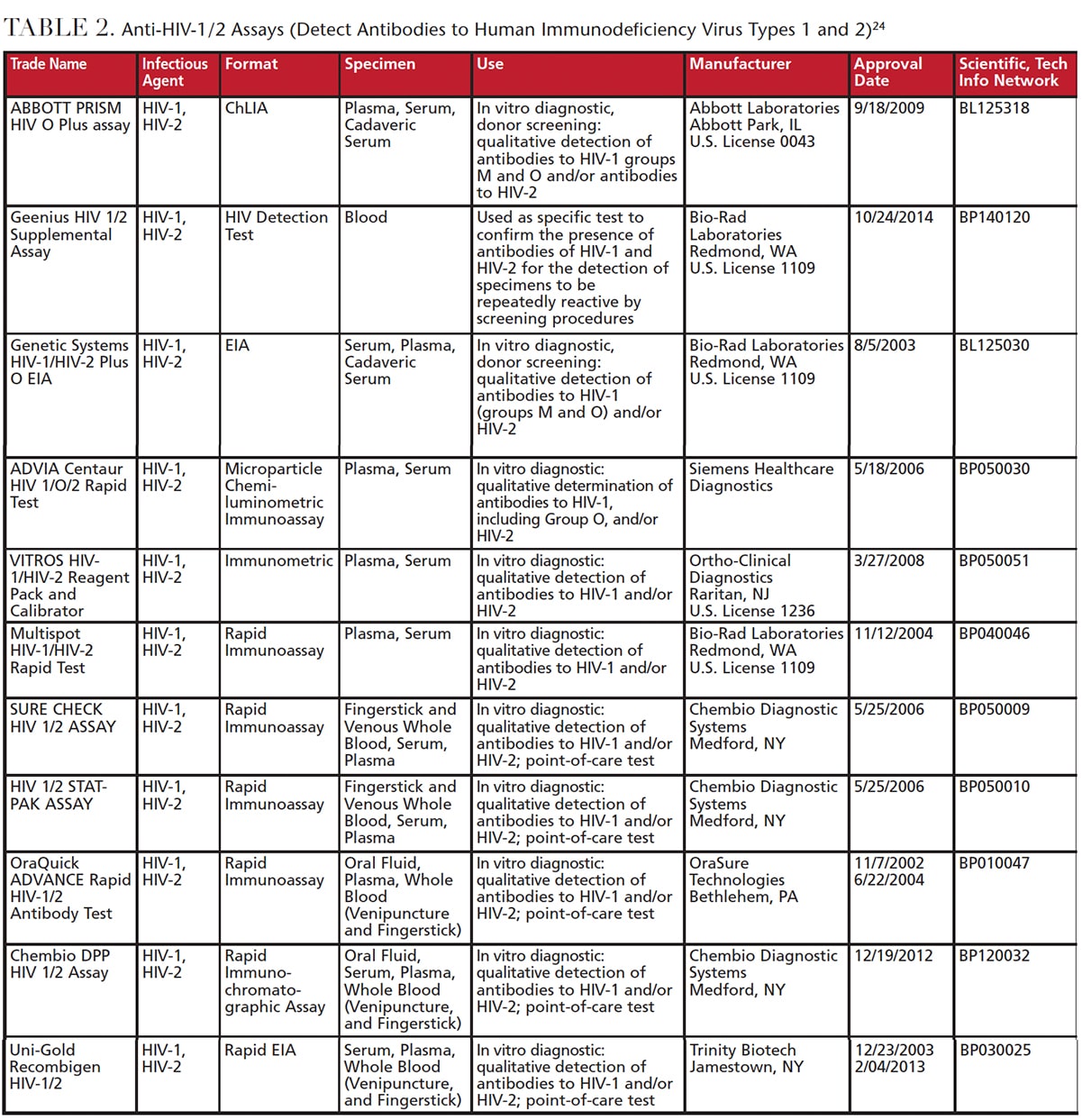
What are 4 examples of CLIA waived tests?
The specific tests that are deemed to be CLIA waived are:Non-automated urinalysis using dipstick or tablet reagents.Ovulation tests.Urine pregnancy tests.Fecal occult blood.Hemoglobin-copper sulfate.Erythrocyte sedimentation rate.Blood glucose monitoring utilizing devices specifically for home use.Spun microhematocrit.More items...
What is considered a waived test?
What is waived testing? By the CLIA law, waived tests are those tests that are determined by CDC or FDA to be so simple that there is little risk of error. Some testing methods for glucose and cholesterol are waived along with pregnancy tests, fecal occult blood tests, some urine tests, etc.
What tests are CLIA waived?
As defined by CLIA, waived tests are categorized as “simple laboratory examinations and procedures that have an insignificant risk of an erroneous result.” The Food and Drug Administration (FDA) determines which tests meet these criteria when it reviews manufacturer's applications for test system waiver.
What are examples of CLIA waived tests that can be performed in a community pharmacy?
Some examples of CLIA-waived tests are influenza, Strep A, HIV, A1c, and INR testing. Many of these tests can be performed without specialized equipment and provide results within five to 20 minutes.
What are the 3 types of CLIA waivers?
CLIA regulatory requirements classify testing into three categories: high complexity, moderate complexity and waived.
Is a CBC a CLIA-waived test?
It is a CLIA-waived analyzer that is factory calibrated and it will not report values that might be inaccurate. Prior to or during the examination, the patient's blood is drawn and the CBC is performed in your office.
What is the difference between CLIA and CLIA-waived?
Waived tests include test systems cleared by the FDA for home use and those tests approved for waiver under the CLIA criteria. Although CLIA requires that waived tests must be simple and have a low risk for erroneous results, this does not mean that waived tests are completely error-proof.
What CPT codes are CLIA-waived?
Certain codes describe only CLIA-waived tests and therefore are exempt from the requirement to add the QW modifier. The CPT codes for the tests currently exempt from the requirement are 81002, 81025, 82270, 82272, 82962, 83026, 84830, 85013, and 85651.
Is urine dipstick CLIA-waived?
Urinalysis dipsticks fall into the CLIA-waived category and are generally very reliable, simple to use, and easy to interpret.
What is the difference between waived and non waived testing?
Waived tests are simple tests with a low risk for an incorrect result. Nonwaived tests, also called Moderately Complex tests, are those tests that require a number of criteria to be met prior to the operator running the tests.
Is rapid strep test CLIA waived?
Test Result The CLIAwaived,™ Inc. Rapid Strep A Test is a rapid, visual test for the presumptive in-vitro qualitative detection of Group A Streptococcal antigen from throat swab specimens. This test is for professional use only.
When should modifier QW be used?
Modifier QW is used to indicate that the diagnostic lab service is a Clinical Laboratory Improvement Amendment (CLIA) waived test and that the provider holds at least a Certificate of Waiver. The provider must be a certificate holder in order to legally perform clinical laboratory testing.
What is waived testing Joint Commission?
Waived testing is defined by the Clinical Laboratory Improvement Amendments (CLIA) of 1988 as tests which have the following characteristics: They employ methodologies that are so simple and accurate as to render the likelihood of erroneous results negligible.
What are some of the benefits of performing waived tests?
Some of the benefits of performing waived testing include: rapid availability of results while the patient is available for immediate follow-up, ● simple tests have minimal need for training, and ● portability of many waived tests allows for easier testing in nontraditional settings.
What is the first step for CLIA-waived tests?
You can enroll your laboratory or testing site in the CLIA program by completing an application (Form CMS-116 ) available on the CMS CLIA website or from your local State Agency . Send your completed application to the address of the local State Agency for the State in which your laboratory or testing site is located.
What is the most common coagulation test performed in a CLIA-waived pol?
Several testing methodologies are used in immunological testing; however, for CLIA-waived testing the most common methodology is lateral flow immunoassay.
What is a CLIA waived test? Is this the same as a CLIA Certificate of Waiver?
A CLIA certificate is still required to perform a CLIA waived test.
What are some common deficiencies that inspections find?
Some common deficiencies that may prevent a laboratory from receiving CLIA certification include:
What is a laboratory developed test (LDT)?
Typically, these tests are not distributed or sold commercially. These are tests developed in-house in a healthcare facility to perform on their own.
What does CMS CLIA require for analytical validity for LDTs?
The federal government, through the CLIA program, thoroughly regulates the evaluation and use of all lab tests including lab-developed assays.
What are the three agencies and their roles in assuring quality laboratory testing?
FDA: Categorizes tests based on complexity; reviews requests for waivers by application; develops rules/guidance for CLI A complex ity categorization
What is modified testing?
Based on the testing site’s need and the unique population it serves, there may be instances when the site chooses to modify an FDA-cleared or approved test system. Modification means using a test system in a way other than that described in the intended use, precautions, limitations, or other sections of the manufacturer’s instructions (See Appendix B for an explanation of the common components found in a manufacturer’s instructions). The modified use of a test system is considered “off-label use” because it is not supported by the manufacturer’s clinical data and it is not part of the FDA-cleared or approved instructions. “Off-label use”, or modified use of a test system, defaults the test to the high-complexity testing category under CLIA regulations, and will require sites using the modified test system to meet all applicable CLIA requirements for high-complexity testing. These include requirements for proficiency testing (PT), establishing performance characteristics, quality control (QC), quality assessment, and adherence to personnel qualifications. Laboratories with a CLIA Certificate of Waiver that are using modified test systems will need to upgrade to a CLIA Certificate of Compliance or a CLIA Certificate of Accreditation if they continue to use modified test systems.
How to perform a 99 test?
When performing a test, some important points to remember are: 99Follow the testing steps in the exact order as they are in the manufacturer’s instructions. 99Test QC following the manufacturer’s instructions. 99Have the manufacturer’s instructions, site specific procedure, or a quick reference guide at the testing area. 99Use timers and follow the required timing intervals before reading test results.
How to interpret test results?
Interpret test results according to the manufacturer’s instructions. Keep the quick reference guide or color charts available to help interpret results. Test results are either quantitative, qualitative, or a combination of the two with a number result that is interpreted into a non-numeric result.
Why is QC testing important?
QC testing gives confidence that your results are accurate and reliable. The manufacturer’s instructions or site specific procedure explain what the controls are checking, the steps for performing QC testing, and when to do QC testing. Incorrect QC results alert the user about potential problems such as reagent/test kit deterioration, equipment failure, environmental conditions, or human error.
How to prepare for a 99pretest?
Consult with the patient regarding: 99Pretest instructions — some tests require preparation by the patient such as fasting for a glucose test. Verify these instructions were followed before collecting the sample. 99Pretest information — discuss factors such as medical indications, medications, or other interfering substances that can affect test results with the patient. This information can often be found in the Limitationssection of the manufacturer’s instructions. 99The test(s) — make sure the patient understands what the test(s) and result(s) will mean to their health. 99Patient counseling — some tests, such as HIV tests, benefit from counseling on what the test results will mean for the patient.
What steps should be taken before a test is begun?
Certain steps need to be taken even before a test is begun to be sure results are accurate. Most importantly, follow the manufacturer’s instructions throughout the testing process. Problems found in testing sites that perform waived tests are most often the result of not following this critical step.
Can a blood glucose test be waived?
If the patient’s hematocrit and oxygenation level are within the manufacturer’s stated limits, then performing a glucose test using the waived glucose monitoring system would not be considered off-label testing and the test system would still be considered waived.
What is nonwaived testing?
Nonwaived testing is subject to inspection, and must meet the CLIA quality system standards, such as those for proficiency testing, quality control and assessment, and personnel requirements . The standards for moderate and high complexity testing differ only in the personnel requirements.
Why do clinical laboratories need to know if a test system is waived, moderate, or high complexity?
Clinical laboratories or other testing sites need to know whether a test system is waived, moderate, or high complexity for each test on their menu because this determines the applicable CLIA requirements. In general, the more complicated the test, the more stringent the requirements under CLIA.
What are CLIA waivers?
As defined by CLIA, waived tests are simple tests with a low risk for an incorrect result. They include: 1 Certain tests listed in the CLIA regulations 2 Tests cleared by the FDA for home use 3 Tests that the manufacturer applies to the FDA for waived status by providing scientific data that verifies that the CLIA waiver criteria have been met
What is a PPM test?
PPM procedures are certain moderate complexity microscopy tests commonly performed by health care providers during patient office visits. The PPM subcategory includes a limited set of microscopic evaluations listed in the CLIA regulations that are performed on samples such as urine, skin scrapings and excretions. CLIA offers a PPM certification option for this limited set of moderate complexity tests to accommodate the unique needs of health care providers in clinical settings. Only certain types of providers qualify as testing personnel for PPM tests under a PPM certificate, including physicians, dentists and midlevel practitioners. See 42 CFR 493.19. Also, CMS’ list of PPMP tests (including CPT/HCPCS codes)#N#pdf icon external icon#N#.
