
What is neuston in biology?
Neuston. Neuston, group of organisms found on top of or attached to the underside of the surface film of water. The neuston includes insects such as whirligig beetles and water striders, some spiders and protozoans, and occasional worms, snails, insect larvae, and hydras. It is distinguished from the plankton,...
What is neneuston?
Neuston, group of organisms found on top of or attached to the underside of the surface film of water.
What is an example of a neuston insect?
Mosquito larvae (Diptera, Culicidae) are examples of true neuston ( Figure 33.18 ). The water striders and whirligig beetles prey on organisms stuck to the water surface by the adhesive properties of the water molecules.
What is the habitat of neuston?
Consequently, most neuston is confined to lentic habitats or some lateral components of the riverscape. The neustonic food web is primarily supported by a thin bacterial film on the upper surface of the water, a concentration of phytoplankton near the surface, and allochthonous inputs from trapped terrestrial and aquatic organisms.
See more

What are examples of neuston?
The neuston includes insects such as whirligig beetles and water striders, some spiders and protozoans, and occasional worms, snails, insect larvae, and hydras. It is distinguished from the plankton, which only incidentally becomes associated with the surface film.
What is a neuston ecosystem?
Neuston are considered a cosmopolitan component of aquatic ecosystems and contains a diverse assemblage of taxa that have a direct relationship to the surface layer of water. These organisms may rest on, or move across the water surface, and be in contact with, or move below its surface.
What is pleuston in biology?
'Pleuston' is generally an old term or a term that describes the subset of macroscopic epineustonic organisms. Thus, pleuston refers to macroscopic organisms that live on the surface of the water by floating.
What are some of the different kinds of plankton?
There are two main types of plankton: phytoplankton, which are plants, and zooplankton, which are animals. Zooplankton and other small marine creatures eat phytoplankton and then become food for fish, crustaceans, and other larger species.
Why is the neuston layer important?
Furthermore, the neuston provides a food source to the zooplankton migrating from deeper layers to the surface, as well as to seabirds roaming over the oceans. For these reasons, the neustonic community is believed to play a critical role on the structure and function of marine food webs.
Is neuston a Sargassum?
Organisms that live freely at the surface, termed “neuston,” include keystone organisms like the golden seaweed Sargassum that makes up the Sargasso Sea, floating barnacles, snails, nudibranchs, and cnidarians.
What are examples of Nekton?
2 Nekton. Nekton (or swimmers) are living organisms that are able to swim and move independently of currents. Nekton are heterotrophic and have a large size range, with familiar examples such as fish, squid, octopus, sharks, and marine mammals.
What would happen if phytoplankton increased?
When too many nutrients are available, phytoplankton may grow out of control and form harmful algal blooms (HABs). These blooms can produce extremely toxic compounds that have harmful effects on fish, shellfish, mammals, birds, and even people.
What is marine ecosystem Upsc?
Marine ecosystems - The water bodies containing salt concentration equal to or above that of sea water (i.e., 35 ppt or above). Example: shallow seas and open ocean. Brackish water ecosystems - these water bodies have salt content in between 5 to 35 ppt. Example: estuaries, salt marshes, mangrove swamps and forests.
Is plankton an algae?
The term plankton is a collective name for all such organisms—including certain algae, bacteria, protozoans, crustaceans, mollusks, and coelenterates, as well as representatives from almost every other phylum of animals.
Where does plankton come from?
Most of the plankton in the ocean are plants. Phytoplankton produce their own food by lassoing the energy of the sun in a process called photosynthesis. So for sunlight to reach them, they need to be near the top layer of the ocean.
What is the largest plankton?
With their limited swimming capability, jellyfish are often referred to as the largest type of plankton. In addition to being categorized by life stages, plankton can be categorized into different groups based on size. These groups include: Femtoplankton - Organisms under 0.2 micrometers in size, e.g., viruses.
What are Epifauna Epiflora and Infauna?
Epiflora Lifestyle. Plants that live on seafloor (sea grass) Infauna Lifestyle. Organisms buried in sea floor (tubeworm, sand dollars)
What are examples of Nekton?
2 Nekton. Nekton (or swimmers) are living organisms that are able to swim and move independently of currents. Nekton are heterotrophic and have a large size range, with familiar examples such as fish, squid, octopus, sharks, and marine mammals.
What is a planktonic organism?
Planktonic organisms include protists, microorganisms, and certain types of small metazoan animals, all sharing a common liability to passive entrainment in water currents, generated by tide, wind, convection, gravity, and the rotation of the earth.
What are some examples of benthos?
The word benthos comes from a Greek term meaning “depths of the sea.” Benthic communities are complex and include a wide range of animals, plants and bacteria from all levels of the food web. Clams, worms, oysters, shrimp-like crustaceans and mussels are all examples of benthic organisms.
What are Neutrons?
Neutrons are subatomic particles that are one of the primary constituents of atomic nuclei. They are usually denoted by the symbol n or n o. Neutrons do not have any net electric charge associated with them. They do, however, have mass which is slightly greater in magnitude than that of a proton. Neutrons and protons are collectively referred to as nucleons, since they behave in a similar manner inside the nuclei of atoms. The mass of a neutron can be roughly approximated to one atomic mass unit (often abbreviated to amu). The branch of science that deals with the study of the properties of neutrons and the interactions of these subatomic particles with other matter and electromagnetic radiation is called nuclear physics.
What is the purpose of neutrons?
One of the most significant applications of neutrons is in the excitation of delayed and triggered gamma rays from material components. This forms the basis for study of neutron activation analysis, often abbreviated to NAA.
What is the mass of a neutron?
Charge and Mass of Neutrons. The electric charge that is associated with a neutron is 0. Therefore, neutrons are neutrally charged subatomic particles. The mass of a neutron is roughly equal to 1.008 atomic mass units. When converted into kilograms, the mass of the neutron can be approximated to 1.674*10-27 kg.
How to determine neutron mass?
The mass of the neutron can be calculated by subtracting the mass of a proton from the mass of a deuterium nucleus (deuterium is an isotope of hydrogen containing one proton, one electron, ...
What is the role of neutrons in nuclear reactions?
Neutron capture, for example, often results in activation of neutrons which, in turn, induces radioactivity. Knowledge of neutrons and their activity has been especially important in the past for the development of many nuclear reactors (and also several nuclear weapons). The nuclear fissioning of such elements as uranium-235 and plutonium-239 is almost always caused by their neutron absorption.
What are the two elements that are bound together in the nucleus?
Inside the nucleus of the atom, the protons and the neutrons are bound together via nuclear forces. For the stability of atomic nuclei, the presence of neutrons is a must. The only exception to this rule is the protium (hydrogen-1) nucleus. One of the most important applications of neutrons is in nuclear reactors to facilitate nuclear fission reactions, and in some cases, nuclear fusion reactions.
What is the name of the branch of science that deals with the study of the properties of neutrons?
The branch of science that deals with the study of the properties of neutrons and the interactions of these subatomic particles with other matter and electromagnetic radiation is called nuclear physics.
What is a Neuston?
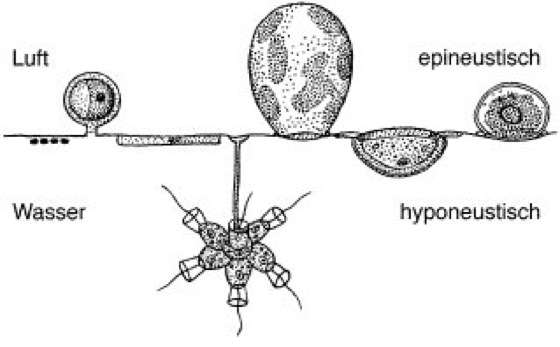
The term neuston refers to the assemblage of organisms associated with the surface film of lakes, oceans, and slow-moving portions of streams. It generally includes species that live just underneath the water surface (hyponeuston), individuals that are above but immersed in the water (epineuston), and taxa that travel over the surface on hydrophobic structures (superneuston or, more properly, a form of epineuston). This name is similar to, or a subset of, the older name, pleuston (sometimes neuston is used in reference to the microscopic components of the more encompassing pleuston). The density of neustonic organisms decreases with increasing turbulence.
What is Neuston in aquatic ecosystems?
Neuston are considered a cosmopolitan component of aquatic ecosystems and contains a diverse assemblage of taxa that have a direct relationship to the surface layer of water. These organisms may rest on, or move across the water surface, and be in contact with, or move below its surface. The majority of these species originate from ...
How do you collect neuston?
Nets to collect neuston, the zooplankton that live within a few centimeters of the sea surface, by-and-large are non-opening/closing. The first net specifically designed to sample zooplankton neuston was built in about 1960. A rectangular mouth opening design is typical of most of the systems. Neuston nets come either with a single net which collects animals right at the water surface or vertically stacked sets of two to six nets extending from the surface to about 100 cm depth ( Figure 4 ). Normally they are towed from a vessel, but a ‘push-net’ was developed in the 1970s with a pair of rectangular nets positioned side-by-side in a framework and mounted in front of a small catamaran boat that pushed the frame through the water at ∼2.6 knots.
Where is Neuston found in the water?
Representation of neuston in water habitats will also vary when comparing shoreline and pelagic regions of lakes, rivers, and ponds, with the shoreline regions generally having a greater diversity and abundance of neuston.
What are the different types of invertebrates in lakes?
Lake invertebrate assemblages can be subdivided primarily into “zooplankton” (mostly protozoa, rotifers, cladocera, and cope pods) within the water column and “benthos” that live in, on, or just above the bottom (often in association with macrophytes). A third, smaller group encompasses the “neuston” which lives at the air–water interface. These first two categories remain quite distinct except in three general cases. A few species, such as larvae of the phantom midge Chaoborus, migrate upward at dusk into the plankton and return to the benthos near dawn; adults are winged terrestrial individuals. Meroplankton, in contrast to the much more diverse holoplankton, spend only a portion of their life cycle in the water column. For example, chironomid midges are benthic for most of the larval period (the short-lived adults are aerial) but swim within the water column during the earliest stages of their larval existence. The Ponto-Caspian dreissenid mussels, which have invaded much of Europe and North America (e.g., Gallardo et al., 2013) have a veliger stage that is planktonic for up to 3 weeks. Although they are not true members of the plankton, many near-shore species, such as some littoral-zone water fleas (cladocerans such as Bosmina ), alternate periods of resting or foraging on the bottom with intervals of swimming and foraging in the water column.
Do Neuston have compositional changes?
Seasonal composition and abundance changes will also occur among neuston and generally be similar to those compositional changes occurring in the plankton populations. As with the plankton, the neustic flora and fauna will be influenced over time by the trophic status of the water and any environmental perturbations.
Is Neuston freshwater?
Note: Although, neuston investigations have an early freshwater origin and history, the reader is encouraged to read marine-related publications on this topic for additional perspectives concerning collection protocols and the composition of neuston from oceanic habitats. There are also numerous marine publications on the physical and chemical properties of the surface layers of oceans, and the biological relationships that occur in this region.
How much does a neutron weigh?
A neutron has a mass of 1.67492729 x 10 -27 kg. A neutron is considered a type of fermion because it has a spin = 1/2. Although it's possible to eject neutrons from the nucleus, the free particles don't last long before reacting with other atoms. On average, a neutron survives on its own about 15 minutes.
What is the charge of a neutron?
The neutron is the particle in the atomic nucleus with a mass = 1 and charge = 0. Neutrons are found together with protons in the atomic nucleus. The number of neutrons in an atom determines its isotope. Although a neutron has a net neutral electrical charge, it does consist of charged components which cancel each other out with respect to charge.
How many quarks are in a neutron?
It consists of one up quark and two down quarks. Although the mass of a proton and a neutron are comparable, especially compared with the much lighter electron, a neutron is slightly more massive than a proton. A neutron has a mass of 1.67492729 x 10 -27 kg.
Why are neutrons needed?
Neutrons are required for the stability of nuclei, with the exception of the single-proton hydrogen nucleus. Neutrons are produced copiously in nuclear fission and fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes.
Which type of neutron has more energy?
High-energy neutrons have much more energy than fission energy neutrons and are generated as secondary particles by particle accelerators or in the atmosphere from cosmic rays. These high-energy neutrons are extremely efficient at ionization and far more likely to cause cell death than X-rays or protons.
How are the protons and neutrons formed?
An atomic nucleus is formed by a number of protons, Z (the atomic number ), and a number of neutrons, N (the neutron number ), bound together by the nuclear force. The atomic number determines the chemical properties of the atom, and the neutron number determines the isotope or nuclide.
What are the atoms of an element that differ only in neutron number?
Atoms of a chemical element that differ only in neutron number are called isotopes. For example, carbon, with atomic number 6, has an abundant isotope carbon-12 with 6 neutrons and a rare isotope carbon-13 with 7 neutrons. Some elements occur in nature with only one stable isotope, such as fluorine.
Why is the magnetic moment of a neutron negative?
The neutron's magnetic moment has a negative value, because its orientation is opposite to the neutron's spin. A free neutron is unstable, decaying to a proton, electron and antineutrino with a mean lifetime of just under 15 minutes ( 879.6 ± 0.8 s ).
When was the neutron discovered?
In 1920, Rutherford suggested that the nucleus consisted of positive protons and neutrally charged particles, suggested to be a proton and an electron bound in some way. Electrons were assumed to reside within the nucleus because it was known that beta radiation consisted of electrons emitted from the nucleus. Rutherford called these uncharged particles neutrons, by the Latin root for neutralis (neuter) and the Greek suffix -on (a suffix used in the names of subatomic particles, i.e. electron and proton ). But references to the word neutron in connection with the atom can be found in the literature as early as 1899.
What is the name of the nucleus of the most common isotope of the hydrogen atom?
Nuclides with the same atomic mass number, but different atomic and neutron numbers, are called isobars . The nucleus of the most common isotope of the hydrogen atom (with the chemical symbol 1 H) is a lone proton.
What is the neutron in physics?
neutron, neutral subatomic particlethat is a constituentof every atomic nucleus except ordinary hydrogen. It has no electric chargeand a rest massequal to 1.67493 × 10−27kg—marginally greater than that of the protonbut nearly 1,839 times greater than that of the electron. Neutrons and protons, commonly called nucleons, are bound together in the dense inner core of an atom, the nucleus, where they account for 99.9 percent of the atom’s mass. Developments in high-energy particle physicsin the 20th century revealed that neither the neutron nor the proton is a true elementary particle; rather, they are composites of extremely small elementary particles called quarks. The nucleus is bound together by the residual effect of the strong force, a fundamental interactionthat governs the behaviour of the quarks that make up the individual protons and neutrons.
What is the mass of a neutron?
It has no electric charge and a rest mass equal to 1.67493 × 10 −27 kg —marginally greater than that of the proton but nearly 1,839 times greater than that of the electron.
When were neutrons discovered?
The neutron was discovered in 1932 by the English physicist James Chadwick. Within a few years after this discovery, many investigators throughout the world were studying the properties and interactions of the particle. It was found that various elements, when bombarded by neutrons, undergo fission—a type of nuclear reactionthat occurs when the nucleus of a heavy element is split into two nearly equal smaller fragments. During this reaction each fissioned nucleus gives off additional free neutrons, as well as those bound to the fission fragments. In 1942 a group of American researchers, under the leadership of the physicist Enrico Fermi, demonstrated that enough free neutrons are produced during the fission process to sustain a chain reaction. This development led to the construction of the atomic bomb. Subsequent technological breakthroughs resulted in the large-scale production of electric powerfrom nuclear energy. The absorptionof neutrons by nuclei exposed to the high neutron intensities available in nuclear reactors has also made it possible to produce large quantities of radioactive isotopesuseful for a wide variety of purposes. Furthermore, the neutron has become an important tool in pure research. Knowledge of its properties and structure is essential to an understanding of the structure of matter in general. Nuclear reactions induced by neutrons are valuable sources of information about the atomic nucleus and the forcethat binds it together.
What happens when a heavy element is fissioned?
It was found that various elements, when bombarded by neutrons, undergo fission —a type of nuclear reaction that occurs when the nucleus of a heavy element is split into two nearly equal smaller fragments. During this reaction each fissioned nucleus gives off additional free neutrons, as well as those bound to the fission fragments.
How long does it take for a neutrino to decay?
It breaks down into a proton, an electron, and an antineutrino (the antimatter counterpart of the neutrino, a particle with no charge and little or no mass); the half-life for this decay process is 614 seconds.
Who was the first person to discover that neutrons are produced during the fission process?
In 1942 a group of American researchers, under the leadership of the physicist Enrico Fermi, demonstrated that enough free neutrons are produced during the fission process to sustain a chain reaction. This development led to the construction of the atomic bomb.
Who discovered the neutron?
The neutron was discovered in 1932 by the English physicist James Chadwick. Within a few years after this discovery, many investigators throughout the world were studying the properties and interactions of the particle.
Where do neurons originate?
For instance, until recently, researchers believed that neuron creation occurred in adults in a region of the brain called the hippocampus. The hippocampus is involved in memory and learning.
How many types of neurons are there?
Given the sheer number of neurons, there are thousands of different types, much like there are thousands of species of living organisms on Earth. In terms of function, scientists classify neurons into three broad types: sensory, motor, and interneurons.
How do action potentials affect other neurons?
In a chemical synapse, action potentials affect other neurons via a gap between neurons called a synapse. Synapses consist of a presynaptic ending, a synaptic cleft, and a postsynaptic ending. When an action potential is generated, it’s carried along the axon to a presynaptic ending.
How many dendrites can a neuron have?
Neurons can have more than one set of dendrites, known as dendritic trees. How many they have generally depends on their role. For instance, Purkinje cells are a special type of neuron found in the cerebellum. These cells have highly developed dendritic trees which allow them to receive thousands of signals.
How do neuronal signals work?
Neurons send signals using action potentials. An action potential is a shift in the neuron’s electric potential caused by the flow of ions in and out of the neural membrane. Action potentials can trigger both chemical and electrical synapses.
Where are the interneurons located?
Interneurons are neural intermediaries found in your brain and spinal cord. They’re the most common type of neuron. They pass signals from sensory neurons and other interneurons to motor neurons and other interneurons. Often, they form complex circuits that help you to react to external stimuli.
What are the cells that send and receive signals from the brain?
Neurons, also known as nerve cells, send and receive signals from your brain. While neurons have a lot in common with other types of cells, they’re structurally and functionally unique.
What Are neutrons?
- Neutrons are subatomic particles that are one of the primary constituents of atomic nuclei. They are usually denoted by the symbol n or no. Neutrons do not have any net electric charge associated with them. They do, however, have a mass which is slightly greater in magnitude than that of a proton. Neutrons and protons are collectively referred to a...
Discovery of Neutrons
- Neutrons were first theorized by the New Zealand born British physicist Ernest Rutherford in the year 1920.
- The discovery of neutrons is credited to the British physicist James Chadwick in the year 1932.
- He was awarded the Nobel prize in physics for this discovery in the year 1935.
Charge and Mass of Neutrons
- The electric charge that is associated with a neutron is 0. Therefore, neutrons are neutrally charged subatomic particles.
- The mass of a neutron is roughly equal to 1.008 atomic mass units. When converted into kilograms, the mass of the neutron can be approximated to 1.674*10-27 kg.
- Since neutrons lack electric charge, their mass cannot be directly determined via the analytic…
- The electric charge that is associated with a neutron is 0. Therefore, neutrons are neutrally charged subatomic particles.
- The mass of a neutron is roughly equal to 1.008 atomic mass units. When converted into kilograms, the mass of the neutron can be approximated to 1.674*10-27 kg.
- Since neutrons lack electric charge, their mass cannot be directly determined via the analytical technique of mass spectrometry.
- The mass of the neutron can be calculated by subtracting the mass of a proton from the mass of a deuterium nucleus (deuterium is an isotope of hydrogen containing one proton, one electron, and one...
Properties of Neutrons
- Despite the fact that the neutron is considered to be a neutral particle, the magnetic moment of neutrons is not equal to zero. Even though electric fields have no effects on neutrons, these subatomic particles are affected by the presence of magnetic fields. The magnetic moment associated with the neutron can be considered as an indication of its quark substructure and th…
Applications of Neutrons
- In several nuclear reactions, the subatomic particle known as the neutron plays a significant role. Neutron capture, for example, often results in the activation of neutrons which, in turn, induces radioactivity. Knowledge of neutrons and their activity has been especially important in the past for the development of many nuclear reactors (and also several nuclear weapons). The nuclear f…