
REGULATORY CONSIDERATIONS To identify the potential hazards associated with human exposure to chemicals in general at the work place, through the environment or during the use of a chemical-based product, chemical classification and labelling schemes have been formulated to help reduce potential risks.
What are the legal and regulatory considerations for clinical trials?
Major legal and regulatory considerations • Informed consent • Licensing • Clinical privileges and credentials • Internet prescribing • Conflicts of interest • Malpractice insurance
What are the Internet-based prescribing regulations?
States vary in their Internet-based prescribing regulations, especially when the prescriber resides out of state. Any policies from both the medical and pharmacy boards should be reviewed before implementing a telemedicine program in any state in which the care occurs. Like several areas discussed, federal law overlays state policy.
What are the FINRA rules for supervisory control systems?
FINRA rules require firms to establish and maintain reasonable supervisory policies and procedures related to supervisory control systems in accordance with applicable rules ( e.g., FINRA Rules 3110 and 3120 ).
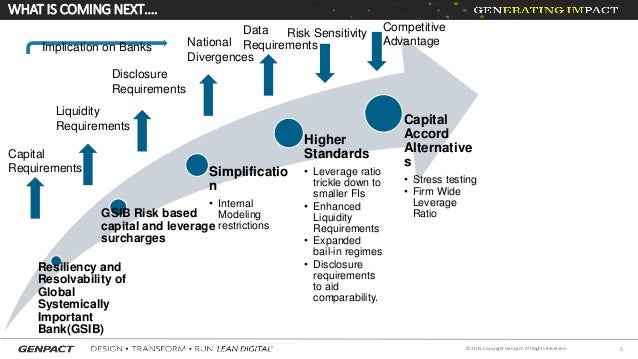
What is a regulatory requirement?
What Is a Regulatory Requirement? A regulatory requirement is a rule that a government entity imposes on an organization. Some federal and state laws govern virtually all organizations. Regulations govern how organizations manage their business and employees and how they interact with customers, among many other areas.
What are regulatory issues in law?
Regulatory Issue means any set of facts or circumstances under which either party's access to, provision of, or use of, the System results in a violation of any Law or gives rise to regulatory action, or a reasonable belief by a party to this Agreement that such a violation or regulatory action is likely to occur.
What is the importance of regulatory?
Regulations are indispensable to the proper functioning of economies and societies. They underpin markets, protect the rights and safety of citizens and ensure the delivery of public goods and services.
What impact if any do regulatory activities have on the ethics?
1) Regulatory activities allow business organization to sustain the ethical and environmental standard. 2) Every organization has obligation towards the environment protection and regulatory activities made it possible to implement the program which are aimed at managing the waste in best manner.
What are examples of regulatory requirements?
Some examples of regulatory compliance regulations include The U.S. Health Insurance Portability and Accountability Act of 1996 (HIPAA), the Sarbanes-Oxley Act of 2002, and the European Union's General Data Protection Regulation of 2016 (GDPR).
What are examples of regulatory law?
One example of regulatory law is the Consumer Product Safety Improvement Act, enacted in 2008. This law is designed to protect consumers from dangerous or defective products. It requires manufacturers to test their products for safety, and it also imposes fines and penalties for companies that sell unsafe products.
How do you ensure regulatory compliance?
Typical steps to achieve regulatory compliance include the following:Identify applicable regulations. Determine which laws and compliance regulations apply to the company's industry and operations. ... Determine requirements. ... Document compliance processes. ... Monitor changes, and determine whether they apply.
What is the difference between regulatory and compliance?
Compliance risk is the potential that you will be deemed to have violated a law or regulation. Regulatory risk is a potential that changes to laws, regulations or interpretations will cause you losses.
Why is regulatory risk important?
Regulatory risk is the risk that a change in regulations or legislation will affect a security, company, or industry. Companies must abide by regulations set by governing bodies that oversee their industry. Therefore, any change in regulations can cause a rippling effect across an industry.
What are ethical and legal considerations?
Legal actions are applicable to any individual present within a state or country that implements that particular set of laws. On the other hand, ethical rights are considered a voluntary and personal act of an individual based on their perception of right and wrong.
What are the 5 ethical considerations in research?
What are ethical considerations in research? Ethical considerations in research are a set of principles that guide your research designs and practices. These principles include voluntary participation, informed consent, anonymity, confidentiality, potential for harm, and results communication.
What are ethical regulations?
Definition: Ethical standards are a set of principles established by the founders of the organization to communicate its underlying moral values. This code provides a framework that can be used as a reference for decision making processes.
Why is regulatory risk important?
Regulatory risk is the risk that a change in regulations or legislation will affect a security, company, or industry. Companies must abide by regulations set by governing bodies that oversee their industry. Therefore, any change in regulations can cause a rippling effect across an industry.
What does the term case law mean?
Case law is law that is based on judicial decisions rather than law based on constitutions, statutes, or regulations. Case law concerns unique disputes resolved by courts using the concrete facts of a case. By contrast, statutes and regulations are written abstractly.
What is update validation process?
Update model validation processes to account for complexities of an ML model. 31 This includes reviewing the input data (e.g., review for potential bias), the algorithms ( e.g., review for errors), any parameters ( e.g., verify risk thresholds), and the output ( e.g., determine explainability of the output).
Why is model risk management important for AI?
Model risk management becomes even more critical for ML models due to their dynamic, self-learning nature.
Should firms update their policies and procedures?
Firms also should update their written policies and procedures with respect to customer data privacy, to reflect any changes in what customer data and information is being collected in association with AI applications, and how that data is collected, used, and shared. In this regard, below are some questions for firms to consider.
When telemedicine and treatment initiation are provided by the same person (as in Hageseth v?
When telemedicine and treatment initiation are provided by the same person (as in Hageseth v. Superior Court), more ethical dilemmas arise . The following ethical discussion focuses on provider-patient interactions at the most interactive end of the telemedicine spectrum: real-time, synchronous, clinical video telehealth (CVT). Four widely accepted principles of medical ethics should be respected in developing and sustaining any sleep CVT program:11
Is informed consent required in telemedicine?
Informed consent requirements vary by state; there is no federal policy. Some states require a written acknowledgment form completed and signed by the patient, whereas other states have no such requirements. As noted in the Ethics section later, informed consent is an important part of telemedicine initiation whether documentation to that effect is required or not. The Federation of State Medical Boards (FSMB) suggests the following elements be included in informed consent:3
Does Hageseth have a record of patient consent?
Hageseth v. Superior Courtreveals a potential vulnerability when no such documentation of risk acknowledgment exists. Among other deficiencies, Dr Hageseth had no record of patient consent to his method of care.
Main body
Patient-centered care is a frequently mentioned concept in modern health care. Its main idea is to consider patients’ needs and achieve desired health outcomes by inviting patient input into making specific decisions. This type of care promotes improved self-management, patient satisfaction, and adherence to medication (Elwyn et al., 2014).
Conclusion
In general, recent improvements in health care and the possibility of developing professional relationships between patients and healthcare workers are provoking an urgent need for such effective care models as shared decision-making.
