Chemical Property and physical property of element Neon
| Symbol of Neon: | Ne |
| Name: | Neon |
| Atomic Number of Neon: | 10 |
| Group of Neon: | Noble Gas |
| Crystal Structure of Neon: | Cubic: Face centered |
| Atomic number | 10 |
|---|---|
| Density | 0.9*10 -3 g.cm-3 at 20°C |
| Melting point | -249 °C |
| Boiling point | -246 °C |
| Vanderwaals radius | 0.16 nm |
What element has the same properties as neon?
What element is most similar to helium? Helium, neon, and xenon are in the same group, which are the noble gases (group 18/VIIIA). All of the atoms of the noble gases have filled valence shells–two valence electrons for helium, and eight valence electrons for the rest. This makes them very non-reactive, which is a hallmark of the noble gases.
What are chemical and physical properties does neon have?
Neon is an elemental inert gas. It’s chemical symbol is Ne, from the Greek, neos. Its atomic weight is 20.183. Density: 0.8990 g/l at 0 degrees Celsius. It’s atomic number is 10. It’s melting point is -248.67 degrees Celsius, and boiling point is -245.9 degrees Celsius. It is only slightly soluble in hot or cold water.
What determines chemical properties of an element?
What are the factors affecting property value?
- Location. The location of a property is the most obvious factor that affects how much a property is worth. …
- Supply and demand. …
- Interest rates. …
- Economic outlook. …
- Property market performance. …
- Population and demographics. …
- Size and facilities. …
- Aesthetics.
What are the characteristics of the element neon?
Characteristics: Neon is a light, very inert gas. Colorless under normal conditions, it glows a reddish-orange in a vacuum discharge tube. Neon forms no known stable compounds. It has the smallest liquid range of any element (2.6 o C). Uses of Neon. When a few thousand volts are applied to neon, it emits an orange/red light.

What are 5 chemical properties of neon?
Key PropertiesIt is a colorless, tasteless odorless inert gas.It changes to reddish-orange color in vacuum tube.It is chemically inactive.It has the lowest liquid range of any element.
What is the chemical stability of neon?
Neon is stable as it has a full octet or valence shell of 8 electrons. If you are in year 11 or beyond, this will change, but I assume that you are still touching the surface of chemistry and learning that all noble gases (last group on the periodic table) have a full valence electron and "are happy," thus stable.
Is neon chemically reactive?
Because it is a noble gas, neon is stable with a complete octet of electrons. Neon is an inert gas that does not react with any other elements.
What is the chemical bond of neon?
Neon (Ne) is the second of the noble gases. Just like all noble gases, it is very non-reactive. So much so, that it doesn't form compounds with anything. Just like helium (He) and argon (Ar), neon floats around all by itself.
Is neon physical or chemical property?
Neon is a chemical element with a chemical symbol Ne and atomic number 10. It is a noble gas that is colorless, odorless, inert and monatomic.
Is neon brittle or malleable?
ElementLusterMalleabilityNeonColorlessNot able to be stretchedSiliconDull, metallicBrittle, shatters when shapedSulfurDullCannot be stretchedManganeseShinyCan be hammered into thin sheets
How many chemical bonds can neon form?
The first thing it will be helpful to remember is that neon is one of the noble gases, which are well-known for not forming any bonds at all, with some exceptions. However, generally speaking, neon goes around as single atoms.
Is the element neon magnetic?
Neon (chemical symbol Ne, atomic number 10) is the fourth most abundant chemical element in the universe, but it is just a trace element in the air....Neon.GeneralMagnetic orderingnonmagneticThermal conductivity(300 K) 49.1 mW/(m·K)Speed of sound(gas, 0 °C) 435 m/sCAS registry number7440-01-930 more rows
Is neon a solid liquid or gas?
neon (Ne), chemical element, inert gas of Group 18 (noble gases) of the periodic table, used in electric signs and fluorescent lamps. Colourless, odourless, tasteless, and lighter than air, neon gas occurs in minute quantities in Earth's atmosphere and trapped within the rocks of Earth's crust.
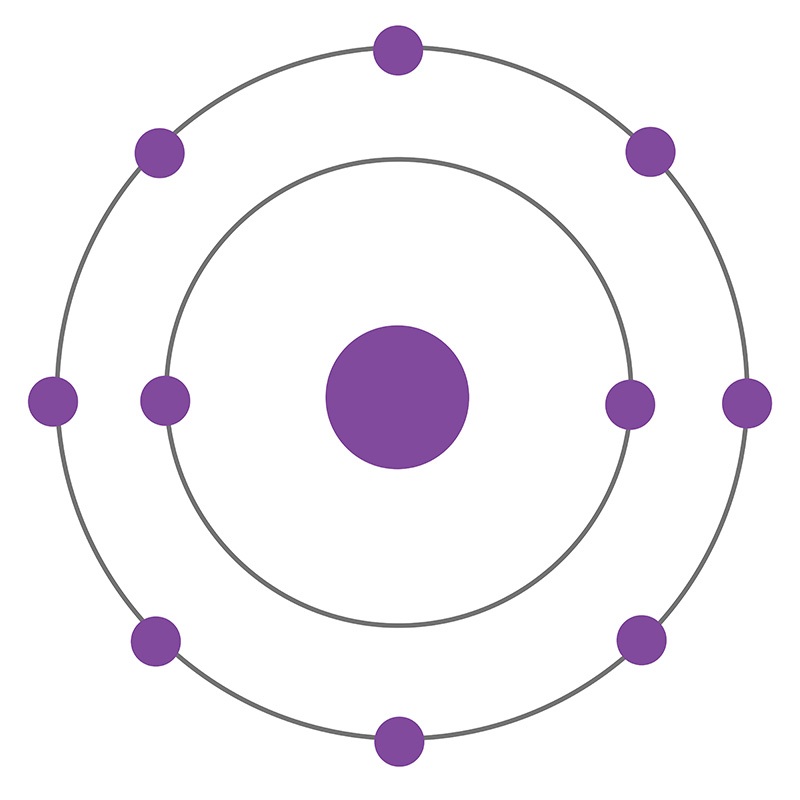
How many rings does neon have?
Therefore, an atom of neon has two rings.
What is neon gas made of?
Neon atoms have 10 electrons and 10 protons with a full outer shell of 8 electrons. Under standard conditions the element neon is a colorless odorless gas. It is a completely inert gas, meaning that it will not combine with other elements or substances to create a compound.
Can neon be a liquid?
Liquid neon is an important cryogenic refrigerant. It has over 40 times more refrigerating capacity per unit volume than liquid helium, and more than 3 times that of liquid hydrogen. Neon has no known biological role. It is non-toxic.
What is the atomic number of neon?
Neon. Neon is a chemical element with symbol Ne and atomic number 10. Classified as a noble gas, Neon is a gas at room temperature. H.
What is neon used for?
Neon is also used to make high voltage indicators and is combined with helium to make helium-neon lasers. Liquid neon is used as a cryogenic refrigerant. Neon is highly inert and forms no known compounds, although there is some evidence that it could form a compound with fluorine.
How is neon obtained?
It is obtained by liquefaction of air and separated from the other gases by fractional distillation. Los Alamos National Laboratory, U.S. Department of Energy.
What is the fourth most abundant element in the universe?
Although neon is the fourth most abundant element in the universe, only 0.0018% of the earth's atmosphere is neon. Jefferson Lab, U.S. Department of Energy. From the Greek word neos, new. Discovered by Ramsay and Travers in 1898.
What is the cosmogenic component of 21 Ne?
The cosmogenic component is mainly detected in 21 Ne and can be used to determine cosmic-ray exposure ages of rock samples, including meteorites exposed during travel through space and boulders exposed by melting of glacial ice (Fig. IUPAC.10.1).
Where can neon isotopes be found?
In addition, neon isotopes can form at the surface of the Earth and in extraterrestrial bodies by cosmic-ray-induced spallation reactions on magnesium, silicon, aluminum, and sodium [103], [104]. Analyses of all three stable neon isotopes may be used to distinguish these sources from primordial neon.
Where did the name "Neos" come from?
The name derives from the Greek neos for "new". It was discovered from its bright orange spectral lines by the Scottish chemist William Ramsay and the English chemist Morris William Travers in 1898 from a liquefied air sample.
What is the color of neon?
Neon is the second-lightest noble gas, its colour is reddish-orange in a vacuum discharge tube and in neon lamps. The the refrigerating capacity of helium is over 40 times the one of liquid helium and three times that of liquid hydrogen (on a per unit volume basis). It is a less expensive refrigerant than helium in most applications.
How does neon affect the body?
Routes of exposure: The substance can be absorbed into the body by inhalation. Inhalation risk: On loss of containment this liquid evaporates very quickly causing supersaturation of the air with serious risk of suffocation when in confined areas.
What is neon used for?
Neon is also used generically for these types of lights when in reality many other gases are used to produce different colors of light. Other uses of neon include high-voltage indicators, lightning arrestors, wave meter tubes and television tubes. Neon and helium are used to make a type of gas laser.
Is neon toxic to the environment?
Neon is a rare atmospheric gas and as such is non-toxic and chemically inert. Neon poses no threat to the environment, and can have no impact at all because it's chemically unreactive and forms no compounds. No known ecological damage caused by this element. Back to periodic chart.
Is neon a rare element?
Although neon is the forth most abundant element in the universe, only 0.0018% in volume of the earth's atmosphere is neon. Neon is usually found in the form of a gas with molecules consisting of a single Neon atom. Neon is a rare gas that is found in the Earth's atmosphere at 1 part in 65,000. Health effects of neon.
What are the elements in the neon gas?
Basically, neon gas is one of the six noble gases featuring in the periodic table, while the other five happen to be helium, argon, krypton, xenon and radon. All these gases come together to form Group 18 of the periodic table.
What is the atomic number of neon?
Physical Properties. Neon is a colorless, odorless, tasteless noble gas – with symbol Ne and atomic number 10 , which is found in trace amounts in the atmosphere of the Earth.
Why are neon signs called neon signs?
While everyone finds these advertising signs appealing, only a few actually know that these signs are referred to as neon signs. The reddish-orange glow that these signs emit is attributed to the presence of neon gas in them, and hence they are referred to as neon signs.
Why did neon escape the atmosphere?
This rare nature of neon gas is also attributed to its physical and chemical properties, as this gas escaped the Earth’s atmosphere because of its relative lightness and inert nature instead of being trapped in the condensing gas and dust clouds during the formation of the planet. « Previous Post. Next Post ».
What is neon used for?
In fact, neon – in its liquefied form, is a popular cryogenic refrigerant. Given below are neon element facts – with special emphasis on its properties, which will shed some light on its importance in various ...
Is neon rare?
Neon Properties and Uses. Even though neon is found in abundance in the universe, it is relatively rare when it comes to the Earth’s atmosphere. This noble gas is commercially extracted from the air in which it is found in trace amounts on the planet.
Is neon an inert gas?
It was initially believed that neon was an inert gas i.e. chemically inactive in nature. However, recent studies have revealed that it does combine with fluorine to form a new compound. Similarly, it also has the tendency of forming ions by combining with other noble gases. These include Ne +, (NeAr) +, (NeH) +, and (HeNe + ). Neon gas also has the ability of forming unstable hydrates. All these chemical properties of neon hint at the fact that neon is not quite an inert gas as it was initially believed to be.
