Precautions
Insulin is produced by cells in the pancreas, called the islets of Langerhans. These cells continuously release a small amount of insulin into the body, but they release surges of the hormone in response to a rise in the blood glucose level. Certain cells in the body change the food ingested into energy, or blood glucose, that cells can use.
Where does insulin come from and what does it do?
- Insulin, glucagon and somatostatin stores in the pancreas of subjects with type-2 diabetes and their lean and obese non-diabetic controls
- Girl With Diabetes and Insulin Allergy to Receive Pancreas Transplant
- A Patient With Diabetes No Longer Needs Insulin After Receiving A Bioengineered “Pancreas”
What organ in the human body produces insulin?
Usual Adult Dose for Diabetes Type 1. Note: Regular human insulin is available in 2 concentrations: 100 units of insulin per mL (U-100) and 500 units of insulin per mL (U-500) Doses should be individualized based on patient's metabolic needs, blood glucose monitoring results, and glycemic goals. -Total daily insulin requirements are generally between 0.5 to 1 unit/kg/day.
How much insulin should you use?
Types of Insulin
- Rapid-acting: These include Apidra, Humalog, and Novolog. ...
- Regular (short-acting): These include Humulin R and Novolin R. ...
- Intermediate-acting: These include Humulin N and Novolin N. ...
- Long-acting: These include Levemir, Lantus, Semglee, and Tresiba. ...
- Ultra long-acting: These include Toujeo. ...
What is the most common type of insulin?

What is the main source of insulin?
Insulin is a peptide hormone secreted by β cells of the pancreas. The human pancreas contains one to two million pancreatic islets [17] housing different endocrine cells, primarily insulin-secreting β cells, glucagon-producing α cells, and somatostatin-secreting δ cells [18].
How do you make insulin?
Scientists make insulin by inserting a gene that codes for the insulin protein into either yeast or bacteria. These organisms become mini bio-factories and start to spit out the protein, which can then be harvested and purified.
How is insulin made naturally?
insulin production naturally by activating beta cells of pancreas. The corosolic acid present in the leaves induces insulin production and thus controls hyperglycaemia in the blood. That's not all, it's also hypolipidemic, diuretic, antioxidant, anti-microbial and anti-cancerous.
What is the chemical formula of insulin?
Insulin human | C257H383N65O77S6 - PubChem.
How is insulin made artificially?
Recombinant DNA is a technology scientists developed that made it possible to insert a human gene into the genetic material of a common bacterium. This “recombinant” micro-organism could now produce the protein encoded by the human gene. Scientists build the human insulin gene in the laboratory.
Can anyone make insulin?
Generic drugs cannot be made until a patent on a brand-name drug expires. One expert pointed out the possible repercussions. "This is a big issue. Some patients simply cannot afford to pay for the insulin that keeps their blood sugar down, even people who have health insurance," explained Dr.
Can pancreas start producing insulin again?
Researchers have discovered that patients with type 1 diabetes can regain the ability to produce insulin. They showed that insulin-producing cells can recover outside the body. Hand-picked beta cells from the islets of Langerhans in the pancreas.
What foods naturally produce insulin?
But increased amounts of natural insulin may be released if you consume certain dietary nutrients....Foods to Boost Natural InsulinAvocados.Nuts like almonds, peanuts, or cashews.Oils including olive, canola, or flaxseed oils.Some types of fish, such as herring, salmon, and sardines.Sunflower, pumpkin, or sesame seeds.
What are the components of insulin?
Components of insulin preparations (e.g., zinc, protamine) and subcutaneous insulin aggregates are also thought to contribute to antibody formation (17). Commercially available human insulins are now virtually free of contaminants and contain <1 ppm of proinsulin (1).
How many amino acids are in insulin?
The insulin molecule consists of 51 amino acids arranged in two chains, an A chain (21 amino acids) and B chain (30 amino acids) that are linked by two disulfide bonds (1) (Figure 1).
Why are there no animal source insulins?
Because of the availability of human insulin and the increased potential for animal source insulin to be immunogenic, animal source insulins are no longer available in the United States. Rare hypersensitivity responses to insulin can be immediate-type, local or systemic IgE-mediated reactions (17).
Why is insulin injected before meal?
Regular insulin is injected pre-meal to blunt the postprandial rise in glucose levels. It forms hexamers after injection into the SQ space slowing its absorption. Hexameric insulin progressively dissociates into absorbable insulin dimers and monomers. For this reason, regular insulin has a delayed onset of action of 30-60 minutes, and should be injected approximately 30 minutes before the meal to blunt the postprandial rise in blood glucose. Adherence to a 30-minute pre-meal schedule is inconvenient and difficult for many patients.
Where is insulin absorbed?
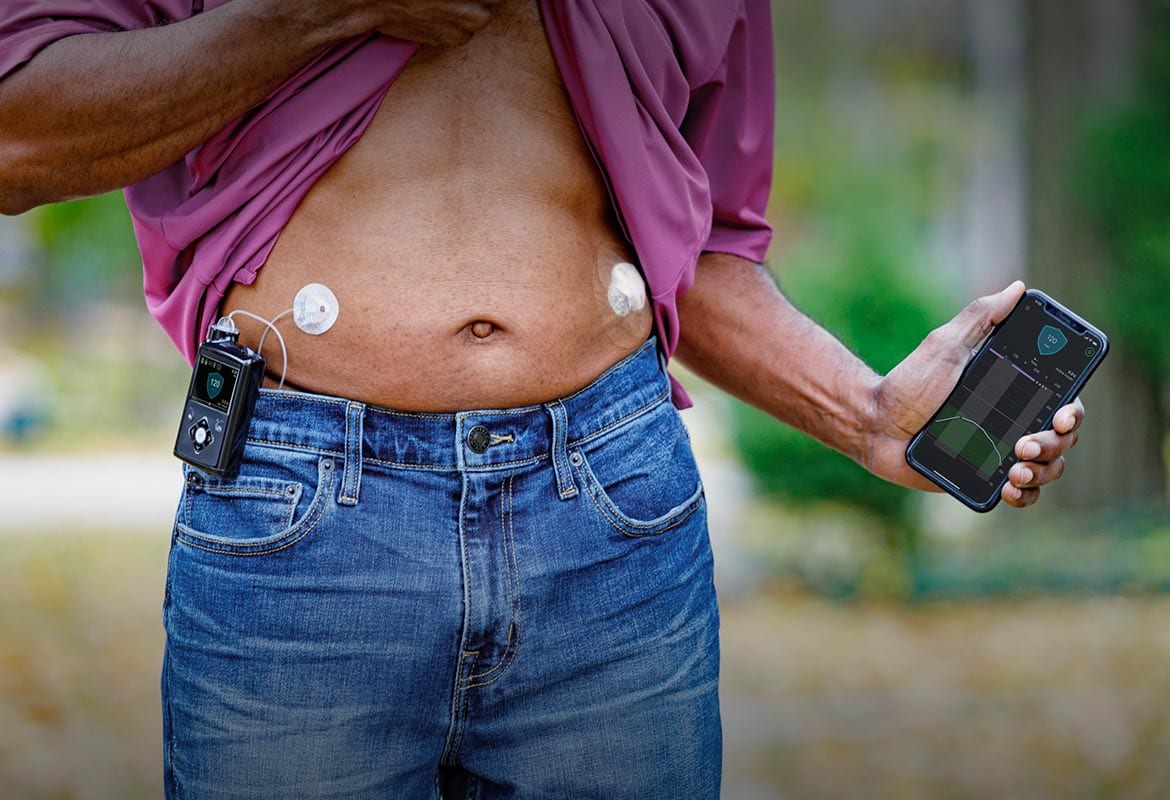
Insulin administered via SQ injection is absorbed directly into the bloodstream, with the lymphatic system playing a minor role in transport (1). The absorption of human insulin into the bloodstream after SQ absorption is the rate limiting step of insulin activity. This absorption is inconsistent with the coefficients of variation of T50% (time for 50% of the insulin dose to be absorbed) varying ~ 25% within an individual and up to 50% between patients (1,20). Most of this variability of insulin absorption is correlated to blood flow differences at the various sites of injection (abdomen, deltoid, gluteus, and thigh) (1). For regular insulin, the impact of this is a ~ 2 times faster rate of absorption from the abdomen than other subcutaneous sites (1). The clinical significance of this is that patients should avoid random use of different body regions for their injections. For example, if a patient prefers to use their thigh for a noontime injection, this site should be used consistently for this injection. The abdomen is the preferred site of injection because it is the least susceptible to factors affecting insulin absorption (see Table 1). Insulin aspart, glulisine and lispro appear to have less day-to-day variation in absorption rates and also less absorption variation from the different body regions (3,5,8,21). Insulin glargine’s pharmacokinetic profile is similar after abdominal, deltoid or thigh SQ administration (7). Similarly, the glucose-lowering effect of insulin degludec has not been found to vary between abdominal, upper arm, or thigh SQ sites (22).
When was insulin first used?
In 1922, Canadian researchers were the first to demonstrate a physiologic response to injected animal insulin in a patient with type 1 diabetes. In 1955, insulin was the first protein to be fully sequenced. The insulin molecule consists of 51 amino acids arranged in two chains, an A chain (21 amino acids) and B chain (30 amino acids) that are linked by two disulfide bonds (1) (Figure 1). Proinsulin is the insulin precursor that is transported to the Golgi apparatus of the beta cell where it is processed and packaged into granules. Proinsulin, a single-chain 86 amino acid peptide, is cleaved into insulin and C-peptide (a connecting peptide); both are secreted in equimolar portions from the beta cell upon stimulation from glucose and other insulin secretagogues. While C-peptide has no known physiologic function, it can be measured to provide an estimate of endogenous insulin secretion.
Which organ is involved in the degradation of insulin?
The kidney has a greater role in insulin degradation with SQ insulin (~60%), with the liver degrading ~30-40% (25). Because the kidneys are involved in the degradation of insulin, renal dysfunction will reduce the clearance of insulin and prolong its effect.
What is the clinical syndrome of insulin?
However, use of insulin in poorly controlled type 1 diabetes can result in a clinical syndrome known as glycogenosis or glycogenic hepatopathy, marked by varying degrees of hepatomegaly, abdominal pain and serum aminotransferase elevations.
Is insulin hepatotoxic?
Insulin is a pancreatic hormone that plays an essential role in regulation of blood glucose as well as lipid and carbohydrate metabolism. Both natural and recombinant forms of insulin are used therapeutically to treat type 1 diabetes. While insulin itself is not hepatotoxic and has not been linked to serum enzyme elevations or instances ...
Does insulin cause liver damage?
Insulin in typical therapeutic doses is not associated with serum enzyme elevations or with episodes of clinically apparent liver injury. However, use of insulin in poorly controlled type 1 diabetes can result in a clinical syndrome known as glycogenosis or glycogenic hepatopathy, marked by varying degrees of hepatomegaly, abdominal pain and serum aminotransferase elevations. Serum ALT and AST levels range from normal to 20 to 30 times the upper limit or normal. Alkaline phosphatase and bilirubin levels are minimally increased or normal. Serum glucose and hemoglobin A1c levels are invariably elevated, and the liver and metabolic abnormalities resolve rapidly with better glycemic control. A severe form of glycogenosis associated with hepatomegaly, growth retardation, delayed puberty and Cushingoid facies in children is known as Mauriac syndrome. Glycogenosis with serum enzyme elevations can also occur in patients with insulin overdose during treatment with high doses of intravenous glucose (Case 1). Glycogenosis has also been reported in patients recieving short-term, high-dose corticosteroids..
Where is insulin produced?
Insulin is produced in the pancreas and the Brockmann body (in some fish), and released when any of several stimuli are detected. These stimuli include the rise in plasma concentrations of amino acids and glucose resulting from the digestion of food. Carbohydrates can be polymers of simple sugars or the simple sugars themselves. If the carbohydrates include glucose, then that glucose will be absorbed into the bloodstream and blood glucose level will begin to rise. In target cells, insulin initiates a signal transduction, which has the effect of increasing glucose uptake and storage. Finally, insulin is degraded, terminating the response.
How many insulin genes are there in mammals?
In some mammals, such as rats and mice, there are two insulin genes, one of which is the homolog of most mammalian genes ( Ins2 ), and the other of which is a retroposed copy that includes promoter sequence but that is missing an intron ( Ins1 ). Both rodent insulin genes are functional.
What snails use insulin?
Cone snails Conus geographus and Conus tulipa, venomous sea snails that hunt small fish, use modified forms of insulin in their venom cocktails. The insulin toxin, closer in structure to fishes' than to snails' native insulin, slows down the prey fishes by lowering their blood glucose levels.
How many hexamers are in insulin?
Insulin is produced and stored in the body as a hexamer (a unit of six insulin molecules), while the active form is the monomer. The hexamer is about 36000 Da in size. The six molecules are linked together as three dimeric units to form symmetrical molecule.
What is the mechanism of glucose homeostasis?
The secretion of insulin and glucagon into the blood in response to the blood glucose concentration is the primary mechanism of glucose homeostasis. Decreased or absent insulin activity results in diabetes mellitus, a condition of high blood sugar level (hyperglycaemia). There are two types of the disease.
Why is type 2 diabetes so common?
It is common for morbidities such as essential hypertension, obesity, type 2 diabetes, and cardiovascular disease (CVD) to develop.
What is the name of the hormone that is produced by beta cells?
Wikidata. View/Edit Human. View/Edit Mouse. Insulin is a peptide hormone containing two chains cross-linked by disulfide bridges. Insulin ( / ˈɪn.sjʊ.lɪn /, from Latin insula, 'island') is a peptide hormone produced by beta cells of the pancreatic islets; it is considered to be the main anabolic hormone of the body.
What is the secretion of insulin?
Secretion of insulin follows a biphasic pattern – i.e. occurs in two phases: Short term blood glucose control: clearing absorbed nutrients from the blood following a meal. Long term insulin release for glucose uptake e.g. for cell growth, cell division, stimulating protein synthesis and DNA replication.
How many chains does insulin have?
Insulin consists of two polypeptide chains, an A chain and a B chain, covalently linked by two inter-chain disulfide bridges. There is a third, intra-chain disulfide bridge. By CNX OpenStax [CC BY 4.0 (http://creativecommons.org/licenses/by/4.0)], via Wikimedia Commons. Fig 1 – Diagram showing the structure of insulin.
What are the two phases of insulin secretion?
Secretion of insulin follows a biphasic pattern – i.e. occurs in two phases: 1 Pulsatile Release (rapid onset)#N#Short term blood glucose control: clearing absorbed nutrients from the blood following a meal. 2 Protracted release (longer)#N#Long term insulin release for glucose uptake e.g. for cell growth, cell division, stimulating protein synthesis and DNA replication.
How is glucose transported into the beta cell?
Glucose is transported into the Beta cell by facilitated diffusion through GLUT2 channels. Therefore a rise in glucose concentration in ECF = a rise in glucose concentration in Beta cells. This leads to membrane depolarisation of ATP sensitive K+ channels, opening Ca2+ channels. This triggers an influx of calcium.
What is the role of insulin in diabetes?
Insulin is a peptide hormone produced by beta cells within the pancreas. It is responsible for regulating movement of glucose from the blood into cells.
What happens when insulin is detected?
When insulin is detected, the alpha chains move together and fold around the insulin. This moves the beta chains together making them an active tyrosine kinase. This initiates a phosphorylation cascade which results in an increase of GLUT4 expression – a protein channel to allow glucose uptake.
Where does insulin bind to?
Insulin binds to a highly specific insulin receptor on cell surfaces.
Which cells produce insulin?
The pancreatic cells that produce glucagon are called alpha cells. 1. Insulin is produced by specialized beta cells in the pancreas, which are clustered into groups called islets of Langerhans.
How does insulin work?
How Insulin Works. Insulin is the energy-storage hormone. After a meal, it helps the cells use carbs , fats, and protein as needed, and to store what's left (mainly as fat) for the future. The body breaks these nutrients down into sugar molecules, amino acid molecules, and lipid molecules, respectively. The body also can store and reassemble these ...
Why is insulin important?
Insulin is critical to your overall health, and even survival. Problems with insulin production or function can result in hypoglycemia (low blood sugars), hyperglycemia (high blood sugars), and diabetes. Verywell / Ellen Lindner.
How does insulin affect the liver?
Insulin stimulates the creation and storage of glycogen from glucose. High insulin levels cause the liver to get saturated with glycogen. When this happens, the liver resists further storage. Then, glucose is used instead to create fatty acids that are converted into lipoproteins and released into the bloodstream.
Why is my insulin not binding?
This can be caused by a problem with the shape of the insulin (preventing receptor binding), not having enough insulin receptors, signaling problems, or glucose transporters not working properly .
What is the function of insulin receptors?
Insulin helps transport glucose from the bloodstream with the help of glucose transporters. Insulin receptors have two main components—the exterior and interior portions. The exterior portion extends outside the cell and binds with insulin.
Why is insulin important for muscle growth?
Insulin helps the amino acids in protein to enter cells. Without adequate insulin production, this process is hindered, making it difficult to build muscle mass.
How is insulin made?
Insulin is a polypeptide hormone formed, after elimination of C peptide by hydrolysis, of two chains of 21 and 30 amino acids, connected by two disulfide bridges . It is secreted by the ß cells of the islets of Langerhans of the pancreas and exerts an hypoglycemic action. It belongs to the group of peptides called IGF (insulin like growth factors) or somatomedins. Biosynthesis Insulin is produced in beta cells which constitute 75% of the islets of Langerhans of the pancreas. Alpha cells secrete glucagon, delta cells somatostatin. Insulin is synthesized in the form of a single polypeptide chain, preproinsulin which is transformed into proinsulin which, itself, catalyzed by proteases called furines, gives insulin and C peptide (C for connecting, because connecting the two chains A and B). Bound to two zinc atoms, insulin is stored in granules as a polymer, probably a hexamer. Secretion Insulin, as well as C peptide, are released by exocytosis into the portal venous system which leads it directly to the liver, which takes up nearly 50%. The remainder of insulin is distributed throughout the body. With a basal secretion of approximately 40 microgram/h under fasting conditions, there are increases of secretion linked to meals. To these slow variations are superimposed peaks of pulsatile secretion. The aim of the treatments by exogenous insulin is to approach the physiological curve of secretion. The principal stimulant of insulin secretion is glucose; it elicits a biphasic release: an immediate effect of short duration and a sustained effect. The cells of the islets are connected by tight junctions, which allow the transfer of ions, of metabolites, secondary messengers from one cell to another, and thus play an important part in synchronizing the secretions. The stimulation Continue reading >>
What is the function of insulin?
Its main function is the regulation of blood sugar levels, by causing the liver and muscles to increase uptake of glucose [1]. Insulin is produced from a single gene which codes for the peptide proinsulin; a precursor molecule. Mutations in this gene can result in ...
How does insulin affect gene expression?
Insulin also has an effect on gene expression which is up or down regulated in the homeostasis process to maintain the optimum blood glucose levels.
What is insulin used for?
This article is about the insulin protein. For uses of insulin in treating diabetes, see insulin (medication). Not to be confused with Inulin. Insulin (from Latin insula, island) is a peptide hormone produced by beta cells of the pancreatic islets, and it is considered to be the main anabolic hormone of the body. [5] It regulates the metabolism of carbohydrates, fats and protein by promoting the absorption of, especially, glucose from the blood into fat, liver and skeletal muscle cells. [6] In these tissues the absorbed glucose is converted into either glycogen via glycogenesis or fats (triglycerides) via lipogenesis, or, in the case of the liver, into both. [6] Glucose production and secretion by the liver is strongly inhibited by high concentrations of insulin in the blood. [7] Circulating insulin also affects the synthesis of proteins in a wide variety of tissues. It is therefore an anabolic hormone, promoting the conversion of small molecules in the blood into large molecules inside the cells. Low insulin levels in the blood have the opposite effect by promoting widespread catabolism, especially of reserve body fat. Beta cells are sensitive to glucose concentrations, also known as blood sugar levels. When the glucose level is high, the beta cells secrete insulin into the blood; when glucose levels are low, secretion of insulin is inhibited. [8] Their neighboring alpha cells, by taking their cues from the beta cells, [8] secrete glucagon into the blood in the opposite manner: increased secretion when blood glucose is low, and decreased secretion when glucose concentrations are high. [6] [8] Glucagon, through stimulating the liver to release glucose by glycogenolysis and gluconeogenesis, has the opposite effect of insulin. [6] [8] The secretion of insulin and glucagon into the Continue reading >>
What are beta cells?
Beta cells are sensitive to glucose concentrations, also known as blood sugar levels. When the glucose level is high, the beta cells secrete insulin into the blood; when glucose levels are low, secretion of insulin is inhibited. [8] .
Why does the pancreas release insulin?
When blood sugar levels rise following a meal, the pancreas is stimulated to release insulin into the bloodstream. In order for tissues to absorb glucose from the blood, they must first bind insulin. Glucose metabolism is necessary for cell growth and energy needs associated with cell function.
Where is insulin stored?
Insulin is a small peptide (protein) consisting of fifty-one amino acids synthesized and stored within the pancreas, an organ situated behind the stomach . The protein itself consists of two chains, denoted A and B, linked by disulfide (sulfur-sulfur) bridges between cysteine residues (see Figure 1).
What are the determinants of insulin?
The structure of insulin contains determinants of foldability, trafficking, self-assembly, and receptor binding.
What is the role of insulin in the body?
Insulin plays a central role in the regulation of human metabolism . The hormone is a 51-residue anabolic protein that is secreted by the β-cells in the Islets of Langerhans. Containing two chains (A and B) connected by disulfide bonds, the mature hormone is the post-translational product of a single-chain precursor, designated proinsulin . Extensive studies of the three-dimensional structure of insulin, pioneered by D. C. Hodgkin , have enabled the development of therapeutic analogs for the treatment of the metabolic disorder diabetes mellitus (DM) . The insulin gene is the site of dominant mutations associated with DM . Although such mutations are uncommon, their molecular analysis has provided important insights into the biochemical bases of the hormone’s pathway of biosynthesis and mechanism of receptor binding . The largest class of mutations is associated with the impaired folding of proinsulin, which leads in turn to progressive endoplasmic-reticular (ER) stress, β-cell death and DM, usually with onset in the neonatal period .
How does glucose affect insulin secretion?
High ambient glucose concentration in the islets promotes insulin biosynthesis and is the primary regulator of secretion. Elevated glucose concentrations cause an increase in cAMP levels by a mechanism that does not appear to involve activation of adenylate cyclase . cAMP then exerts its effects via a mechanism involving protein kinase A (PKA), leading to the phosphorylation and activation of certain key proteins . Through this complex chain of events, glucose and cAMP (and possibly contributions from the rise in intracellular free calcium and IP3) rapidly increase translation and transcription of insulin mRNA . Insulin mRNA normally turns over slowly, with a half life of approximately 30 hours at normal or below normal levels. However, elevated ambient glucose concentrations increase the half life of insulin mRNA as much as threefold . Calcium dependent exocytosis of secretory granules is the main mechanism of secretion in both glucose-stimulated and basal states . Little or no direct secretion of proinsulin occurs from the rER to the plasma membrane by way of unregulated pathways . Other hormones and chemical substances also play an important role in the regulation of insulin secretion , including glucagon, which is secreted by α -cells in pancreatic islets as discussed in detail in another chapter; glucagon-like peptide (GLP-1: Ref. ); cholecystokinin ; and gastric inhibitory peptide all acting via specific receptors on the β-cell. Inhibitors of insulin secretion include catecholamines (adrenaline and noradrenaline) which interact with adrenergic receptors on the β-cell membrane , and somatostatin which is secreted by -cells of the pancreatic islets . Amylin is also secreted by the β -cell although the regulatory mechanisms for amylin co-secretion are not well understood.
How does proinsulin convert to insulin?
The conversion of proinsulin to insulin occurs through the joint action of two types of proteases: one with trypsin- like endoprotease activity which cleave after the dibasic residues pairs at each end of the C domain, and another with exopeptidase activity resembling that of carboxypeptidase B to remove the basic residues left after tryptic-like cleavage . Previous studies have also demonstrated that mixtures of pancreatic trypsin and carboxypeptidase B could convert proinsulin to insulin in vitro . Two endoproteases were found within insulinoma secretory granules . Initially called Type I and Type II converting enzymes, each of these acidic endoproteases was found to be dependent on Ca 2+ions. Type I was active in 1 mM Ca 2+and cleaved at Arg31-Arg32 in proinsulin (the first two positions of the C domain) whereas Type II required 0.1 mM Ca 2+and cleaved predominantly at Lys64-Arg65 (the last two positions of the C domain) . Each was also found to have an acidic pH optimum near 6.0.
How is insulin stored in the plasma membrane?
Such release is a multistep process that consists of the transport of the secretory vesicles to the plasma membrane , then docking, priming, and finally fusion of the vesicle with the plasma membrane. This process is regulated cooperatively by nutrients, other hormones, and neurotransmitters in association with electrical depolarization of the β-cell and release of insulin. Only a small portion of the insulin stored in vesicles in the β-cell is released, however, even under maximum stimulation. This suggests that systemic insulin levels are regulated by secretion rather than by biosynthesis and is not ordinarily limited by the size of storage pools. However the mechanisms that regulate the directed transport of the insulin granules to the plasma membrane are also not well understood.
How does insulin affect glucose transport?
Insulin enhances glucose uptake by increasing the number of transporters in the plasma membrane of target cells. This was first demonstrated in adipocytes and subsequently in skeletal and cardiac muscle . Insulin stimulation of such cells mobilizes transporters from intracellular compartments to the plasma membrane to facilitate glucose transport. Translocation of receptors to the plasma membrane has been demonstrated to occur within 30 seconds of insulin stimulation ; as the stimulus dissipates the decrease in the number of plasma membrane receptors declines coincident with a decline in glucose transport . Whereas glucose transport via Glut4 is a passive process (limited only by the chemical potential of the glucose gradient and the V maxof the transporters), translocation and reverse reinternalization of receptors are energy-dependent processes . The impaired ability of insulin, on binding and activation of the IR, to signal Glut4 translocation from intracellular stores contributes to postprandial hyperglycemia in Type 2 DM . Animal studies have also demonstrated that insulin resistance is associated with a decreased translocation of glucose transporters to the plasma membrane in muscle cells . In fact, decreased insulin levels in animal models of DM have been shown not only to decrease transporter translocation, but also to attenuate expression of Glut4 in muscle cells. Thus, it appears that insulin provides both a short-term signal to increase glucose-transporter translocation and a long-term signal to maintain a basal level of expression of such transporters in target cells. The combination of acute and basal actions provides a common mechanism in Type 1 DM (characterized by low or vanishing endogenous insulin levels) or Type 2 DM (characterized by insulin resistance) could cause pathologically high plasma glucose levels: loss of regulation and expression of transmembrane glucose transporters. Glut2, expressed on surface of β-cells, contributes to the regulation of insulin secretion . Accordingly, a β-cell specific IR knock-out (KO) model indicated that insulin likely positively regulates its own secretion from the β-cell .
Where is proinsulin transported?
In the final series of steps proinsulin is transported to the Golgi apparatus where it is packaged into secretory granules and converted to native insulin and C-peptide. The conversion process may begin in the trans Golgi network but continues in the condensing vacuoles (early secretory granules), and the products are stored in mature secretory vesicles, and secreted in equimolar amounts along with small amounts (ca. 2-3%) of proinsulin and intermediate cleavage products . Glucose, in addition to stimulating insulin secretion by β-cells, also activates insulin gene transcription, enhances insulin mRNA stability, and stimulates its translation .
What is insulin used for?
Many types of insulin are used to treat diabetes. Although available choices may seem a bit overwhelming at first, this guide can help you discuss your treatment with your doctor. Insulin is classified by how fast and how long it works in your body.
How long does insulin cover?
Covers insulin needs for about a full day. Often used, when needed, with rapid- or short-acting insulin.
Who to talk to about insulin?
For more information about types of insulin and when to take them, talk to your doctor or diabetes educator.
Can you take insulin if you have type 1 diabetes?
If you have type 1 diabetes, you’ll likely take a combination of insulins. Some people with type 2 diabetes will also need to take insulin. Different brands of insulin vary in onset, peak time, and duration, even if they’re the same type, such as rapid acting.
Overview
Insulin regular is used with a proper diet and exercise program to control high blood sugar in people with diabetes.
May Treat: Type 2 diabetes mellitus · Type 1 diabetes mellitus · Diabetes mellitus · Diabetic coma · Diabetic ketoacidosis and more
Brand Names: Afrezza · Humulin R Regular U-100 Insuln · Novolin R Flexpen · Novolin R InnoLet · Novolin R PenFill and more
Drug Class: Human Insulins - Rapid Acting · Human Insulins - Short Acting
Availability: Prescription sometimes needed
Lactation: Does not adversely affect lactation
May Treat: Type 2 diabetes mellitus · Type 1 diabetes mellitus · Diabetes mellitus · Diabetic coma · Diabetic ketoacidosis and more
Brand Names: Afrezza · Humulin R Regular U-100 Insuln · Novolin R Flexpen · Novolin R InnoLet · Novolin R PenFill and more
Drug Class: Human Insulins - Rapid Acting · Human Insulins - Short Acting
Availability: Prescription sometimes needed
Lactation: Does not adversely affect lactation
Alcohol: Limit intake while taking this medication
Driving: May cause drowsiness or dizziness. Use caution
Evolution and species distribution
Production
Structure
Function
Insulin is a peptide hormone produced by beta cells of the pancreatic islets encoded in humans by the INS gene. It is considered to be the main anabolic hormone of the body. It regulates the metabolism of carbohydrates, fats and protein by promoting the absorption of glucose from the blood into liver, fat and skeletal muscle cells. In these tissues the absorbed glucose is converted into e…
Hypoglycemia
Insulin may have originated more than a billion years ago. The molecular origins of insulin go at least as far back as the simplest unicellular eukaryotes. Apart from animals, insulin-like proteins are also known to exist in the Fungi and Protista kingdoms.
Insulin is produced by beta cells of the pancreatic islets in most vertebrates and by the Brockmann body in some teleost fish. Cone snails Conus geographus and Conus tulipa, venomous sea snails t…
Diseases and syndromes
Insulin is produced exclusively in the beta cells of the pancreatic islets in mammals, and the Brockmann body in some fish. Human insulin is produced from the INS gene, located on chromosome 11. Rodents have two functional insulin genes; one is the homolog of most mammalian genes (Ins2), and the other is a retroposed copy that includes promoter sequence but that is missin…
Medical uses
Contrary to an initial belief that hormones would be generally small chemical molecules, as the first peptide hormone known of its structure, insulin was found to be quite large. A single protein (monomer) of human insulin is composed of 51 amino acids, and has a molecular mass of 5808 Da. The molecular formula of human insulin is C257H383N65O77S6. It is a combinatio…