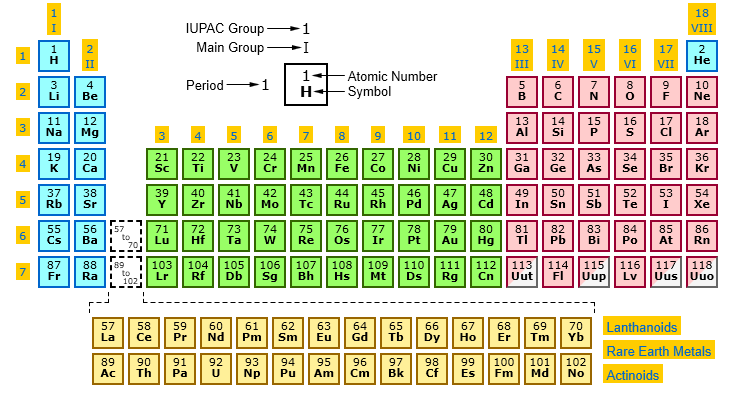
It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit a periodic dependence on their atomic numbers. The table is divided into four roughly rectangular areas called blocks. The rows of the table are called periods, and the columns are called groups.
Full Answer
What are the groups and families on the periodic table?
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties. The horizontal rows on the periodic table are called periods. Group (family): A vertical column in the periodic table.
What are groups and columns in the periodic table called?
- The horizontal rows are called periods.
- The vertical columns are called groups.
- Elements in the same group are similar to each other.
- The metals are on the left and the non-metals are on the right (hydrogen is a non-metal but is often put in the middle).
What does group in periodic table mean?
group, in chemistry, a column in the periodic table of the chemical elements. In a group, the chemical elements have atoms with identical valence electron counts and identical valence vacancy counts. This similarity in both the composition and structure of their atomic valence shells implies a corresponding similarity in both their chemical and physical properties.
What are the different element groups in the periodic table?
- Boron (B)
- Aluminum (Al)
- Gallium (Ga)
- Indium (In)
- Thallium (Tl)
- Nihonium (Nh)

What are the 7 groups of the periodic table?
What are the 7 groups in the periodic tableThe Alkali Metals.The Alkaline Earth Metals.The Transition Metals.The Metalloids.Other Metals.The Non-metals.The Halogens.The Noble Gases.More items...
What is the name of the groups on the periodic table?
These families are alkali metals, alkaline earth metals, transition metals, post-transition metals, metalloids, halogens, noble metals, and noble gases. Many of these families belong to a single group on the periodic table.
What are the 8 groups in periodic table?
The following are the 8 groups of the periodic table:Alkali metals.Alkaline earth metals.Rare earth metals.Crystallogens.Pnictogens.Chalcogens.Halogens.Noble gases.
What are the 13 groups on the periodic table?
Groups 13-16 | Periodic TableGroup 1: Hydrogen And Alkali Metals.Group 2: Alkaline Earth Metals.Groups 3-12: Transition Metals.Groups 13-16.Group 17: Halides.Group 18: Noble Gases.Lanthanoids.Actinoids.
What is Group 4 called in the periodic table?
titanium groupGroup 4 is the second group of transition metals in the periodic table. It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf). The group is also called the titanium group or titanium family after its lightest member.
What are group 6 elements called?
The ChalcogensGroup 6A (or VIA) of the periodic table are the chalcogens: the nonmetals oxygen (O), sulfur (S), and selenium (Se), the metalloid tellurium (Te), and the metal polonium (Po). The name "chalcogen" means "ore former," derived from the Greek words chalcos ("ore") and -gen ("formation").
What is Group 9 called in the periodic table?
transition metalsGroup 9 is a group of chemical elements in the periodic table. Members are cobalt, rhodium, iridium and meitnerium. These are all transition metals in the d-block.
What is Group 13 called?
boron group elementboron group element, any of the six chemical elements constituting Group 13 (IIIa) of the periodic table. The elements are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh).
What are the 11 groups on the periodic table?
All group 11 elements are relatively inert, corrosion-resistant metals. Copper and gold are colored, but silver is not....Characteristics.ZElementNo. of electrons/shell29copper2, 8, 18, 147silver2, 8, 18, 18, 179gold2, 8, 18, 32, 18, 1111roentgenium2, 8, 18, 32, 32, 17, 2 (predicted)
What is group 14 called?
The carbon family consists of the elements carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl). Atoms of elements in this group have four valence electrons. The carbon family is also known as the carbon group, group 14, or the tetrels.
What is Group 15 called?
nitrogen familyGroup 15 elements are also known as pnictogens because in Greek pigeon means to cholk or stifle. In the absence of the oxygen, molecular nitrogen has this property. That's why group 15 elements are known as either nitrogen family or pnictogens.
What are group 14 elements called?
carbon group elementcarbon group element, any of the six chemical elements that make up Group 14 (IVa) of the periodic table—namely, carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl).
What are the 18 groups in a periodic table?
There are total 18 different groups in Periodic table.Group 1: Alkali metals group (hydrogen not included)Group 2: Alkaline earth metals group.Group 3-12: Transition and Inner transition metals group.Group 13: Boron group.Group 14: Carbon group.Group 15: Nitrogen group.Group 16: Oxygen group.Group 17: Halogen group.More items...•
What are the group 5 elements called?
Group 5 contains vanadium (V), niobium (Nb), tantalum (Ta) and dubnium (Db). This group lies in the d-block of the periodic table. The group itself has not acquired a trivial name; it belongs to the broader grouping of the transition metals.
What are Group 3 elements called?
What is this? The elements from Group 3 to 12 are called Transition Metals. They include the Scandium, Titanium, Vanadium, Chromium, Manganese, Iron, Cobalt, Nickel, Copper, and Zinc families of elements.
Why are there 18 groups in the periodic table?
Anyway, the community has pretty much settled on using 18 groups nowadays. The idea behind the periodic system is to find regularities (hence the name) between elements. It turns out, that individual groups and trends within them are similar in the 8 "main" groups (as you have learned).
How are nonmetals separated from metals?
Nonmetals are separated from metals by a line that cuts diagonally through the region of the periodic table. Nonmetals have high ionization energies and electronegativities. They are generally poor conductors of heat and electricity. Solid nonmetals are generally brittle, with little or no metallic luster.
What are the physical properties of halogens?
These reactive elements have seven valence electrons. As a group, halogens exhibit highly variable physical properties . Halogens range from solid to liquid to gaseous at room temperature . The chemical properties are more uniform. The halogens have very high electronegativities. Fluorine has the highest electronegativity of all elements. The halogens are particularly reactive with the alkali metals and alkaline earths, forming stable ionic crystals.
What is the Dnn87?
Dnn87/Creative Commons License. The alkali metals are the elements located in Group IA of the periodic table. The alkali metals exhibit many of the physical properties common to metals, although their densities are lower than those of other metals.
What is the first element in the 5D transition?
The lanthanides are metals that are located in block 5d of the periodic table. The first 5d transition element is either lanthanum or lutetium, depending on how you interpret the periodic trends of the elements. Sometimes only the lanthanides, and not the actinides, are classified as rare earths.
What are the properties of metals?
Many of the properties of metals, including large atomic radius, low ionization energy, and low electronegativity, are due to the fact that the electrons in the valence shell of a metal atoms can be removed easily. One characteristic of metals is their ability to be deformed without breaking.
What group is the alkaline earth in?
The alkaline earths are the elements located in Group IIA of the periodic table. The alkaline earths possess many of the characteristic properties of metals. Alkaline earths have low electron affinities and low electronegativities.
Where are the metalloids located?
The metalloids or semimetals are located along the line between the metals and nonmetals in the periodic table. The electronegativities and ionization energies of the metalloids are between those of the metals and nonmetals, so the metalloids exhibit characteristics of both classes.
What are the elements 57-71 and 89-103?
In addition the elements 57-71 (lanthanum-lutetium) are referred to as the lanthanoids (lanthanides) and the elements 89-103 (actinium-lawrencium) are referred to as the actinoids (act inides).
How many columns are there in the periodic table?
In the standard form of the periodic table the s-block, p-block, and d-block elements are organised into 18 vertical columns called groups. These are labelled from 1 to 18 under current IUPAC numenclature.
Does the Modern Periodic Table Change? If So, How and Who Does That?
The periodic table as we know it today is managed by the International Union of Pure and Applied Chemistry, or IUPAC (eye-you-pack).
What is the CIAAW?
Since 1899 the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW) has been evaluating atomic weights and abundances. For example, Carbon had an atomic weight of 12.00 in 1902 but today it is [12.0096, 12.0116]! Times sure have changed as the source of the sample will determine the value.
What is PubChem working with?
PubChem is working with IUPAC to help make information about the elements and the periodic table machine-readable.
Who is responsible for deciding what needs to be changed?
While much of what is in the periodic table is stable and unlikely to change, the IUPAC organization is responsible for deciding what needs to be changed. They have created criteria for what constitutes the discovery of a new element.
Can periodic table games be used for grade?
The periodic table game available on this page is for entertainment purposes only, and should not be used to grade students on their knowledge of chemical elements.
Who created the periodic table?
The creator of the periodic table, Dmitri Mendeleev, in 1869 began collecting and sorting known properties of elements, like he was playing a game, while traveling by train.
Did Mendeleev's predictions get dismissed?
There were plenty of skeptics and it took years to gain international acceptance, but once newly-discovered elements matched the ones that Mendeleev predicted, his patterns could not be dismissed. In addition, some of the properties that he "fudged" were later recalculated and found to be much closer to his predictions.
What is the intermediate between metals and nonmetals?
Electronegativity and ionization energy intermediate between that of metals and nonmetals
Why is the periodic table useful?
One reason the periodic table of the elements is so useful is that it is a means of arranging elements according to their similar properties. This is what is meant by periodicity or periodic table trends . There are multiple ways of grouping the elements, but they are commonly divided into metals, semimetals (metalloids), and nonmetals.
What are the physical properties of halogens?
The halogens exhibit different physical properties from each other but do share chemical properties. Extremely high electronegativity. Very reactive. Seven valence electrons, so elements from this group typically exhibit a -1 oxidation state.
What are the different types of elements?
There are multiple ways of grouping the elements, but they are commonly divided into metals, semimetals (metalloids), and nonmetals. You'll find more specific groups, like transition metals, rare earths, alkali metals, alkaline earth, halogens, and noble gasses.
Do noble gases have valence electron shells?
The noble gasses have complete valence electron shells, so they act differently. Unlike other groups, noble gasses are unreactive and have very low electronegativity or electron affinity.
Is lanthanide a transition metal?
The lanthanides (rare earth) and actinides are also transition metals. The basic metals are similar to transition metals but tend to be softer and to hint at nonmetallic properties. In their pure state, all of these elements tend to have a shiny, metallic appearance.
Is halogen a metal?
The halogens and noble gases are nonmetals, although they have their own groups, too.
What family are alkali metals in?
Alkali Metals or Group 1 Family of Elements. The highlighted elements of the periodic table belong to the alkali metal element family. Todd Helmenstine. The alkali metals are recognized as a group and family of elements. These elements are metals. Sodium and potassium are examples of elements in this family.
What is the Arabic number system?
The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right. Many periodic tables include both Roman and Arabic numbers. The Arabic numbering system is the most widely accepted today. 02.
What are the columns of the periodic table?
Columns of the periodic table typically mark groups or families. Three systems have been used to number families and groups: The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.
What is the carbon group?
Carbon Group or Tetrels Family of Elements. The highlighted elements belong the carbon family of elements. These elements are collectively known as the tetrels. Todd Helmenstine. The carbon group is made up of elements called tetrels, which refers to their ability to carry a charge of 4. Group 14 or IVA.
What family does the highlighted element belong to?
The highlighted elements of this periodic table belong to the alkaline earth element family. Todd Helmenstine
How are elements categorized?
Elements may be categorized according to element families. Knowing how to identify families, which elements are included, and their properties helps predict behavior of unknown elements and their chemical reactions.
What is the Boron family?
Boron Group or Earth Metal Family of Elements. These are the elements belonging to the boron family. Todd Helmenstine. The boron group or earth metal family is not as well-known as some of the other element families. Group 13 or IIIA. Boron Group or Earth Metals.