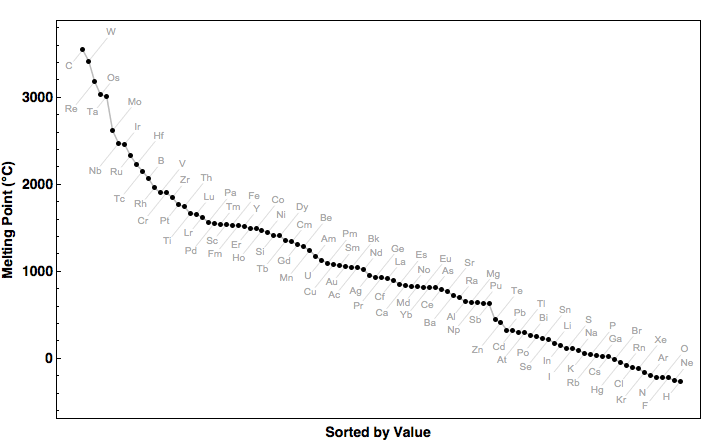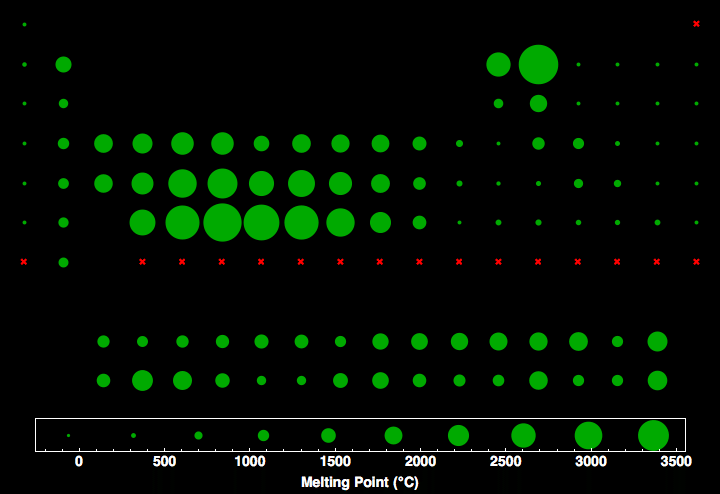
Absolute Melting Point Chart - Absolute Melting Point of the elements in table chart format
| Element Atomic Number | Element Symbol | Element Name | Element Absolute Melting Point |
| 1 | H | Hydrogen | 14.01 |
| 2 | He | Helium | 0 |
| 3 | Li | Lithium | 453.69 |
| 4 | Be | Beryllium | 1560 |
What factors affect melting point?
When molecules are tightly packed together, a substance has a higher melting point than a substance with molecules that do not pack well. Molecular size also affects the melting point. When other factors are equal, smaller molecules melt at lower temperatures than larger molecules.
What metal has a high melting point?
Tungsten is the refractory metal with the highest melting point (3420 °C) . As a refractory metal, the most important advantage of tungsten is that it has good high-temperature strength and good corrosion resistance to the molten alkali metal and steam.
What causes higher melting point?
What causes high melting point? The stronger the intermolecular forces are, the more energy is required, so the higher the melting point is. Many intermolecular forces depend on how strongly atoms in the molecule attract electrons — or their electronegativity.
What is the approximate melting point?
The melting point of a compound is the temperature at which it changes from a solid to a liquid. This is a physical property often used to identify compounds or to check the purity of the compound. It is difficult, though, to find a melting point. Usually, chemist can only obtain a melting point range of 2 - 3( C accuracy.

What determines the melting and boiling points of elements?
Intermolecular forces, relative molecular mass are factors which decides melting and boiling point of element.
What makes an element have a higher melting point?
The high melting point of elements is due to strong intermolecular forces between atoms. Metals are mainly involved in metallic bonds which are an electrostatic force of attraction between metal ions and free electrons in their crystal lattice.
How do you determine the melting point of a compound on the periodic table?
1:483:11Melting Points | A-level Chemistry | OCR, AQA, Edexcel - YouTubeYouTubeStart of suggested clipEnd of suggested clipFirst of all let's have a look at the trend between Group 1 and group 14. So from lithium to carbonMoreFirst of all let's have a look at the trend between Group 1 and group 14. So from lithium to carbon that's between group 1 and group 14 we see a steady increase in both the boiling and melting points.
What affects melting point of metals?
The strength of the bond — which is based on the material itself — is typically what determines the melting point of a metal. Some metal alloys will have higher or lower melting points than the individual metals themselves, and they may not always melt smoothly.
What factors cause melting?
Melting occurs when the Gibbs free energy of the liquid becomes lower than the solid for that material. The temperature at which this occurs is dependent on the ambient pressure.
Which elements has highest melting point?
Tungsten has the highest melting point, which is equal to 3422 degree Celsius.
What does it mean if something has a high melting point?
A high melting point results from a high heat of fusion, a low entropy of fusion, or a combination of both. In highly symmetrical molecules the crystal phase is densely packed with many efficient intermolecular interactions resulting in a higher enthalpy change on melting.
Which has higher melting point?
The chemical element with the lowest melting point is Helium and the element with the highest melting point is Carbon. The unity used for the melting point is Celsius (C).
What does a higher melting point than expected mean?
Impurities, even when present in small amounts, usually lower the melting point and broaden the melting point range. A wide melting point range (more than 5°C) usually indicates that the substance is impure; a narrow melting point range (O. 5-2°C) usually indicates that the substance is fairly pure.
How many protons does helium have?
Helium is a chemical element with atomic number 2 which means there are 2 protons and 2 electrons in the atomic structure. The chemical symbol for Helium is He.
How many electrons does a scandium have?
Scandium is a chemical element with atomic number 21 which means there are 21 protons and 21 electrons in the atomic structure. The chemical symbol for Scandium is Sc.
What is the atomic number of tennessine?
Tennessine is a chemical element with atomic number 117 which means there are 117 protons and 117 electrons in the atomic structure. The chemical symbol for Tennessine is Ts. Oganesson is a chemical element with atomic number 118 which means there are 118 protons and 118 electrons in the atomic structure.
What is the symbol for praseodymium?
The chemical symbol for Praseodymium is Pr. Praseodymium is a soft, silvery, malleable and ductile metal, valued for its magnetic, electrical, chemical, and optical properties. Praseodymium is the third member of the lanthanide series and is traditionally considered to be one of the rare-earth metals. 1204 K.
What is the lightest element on the periodic table?
With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Its monatomic form (H) is the most abundant chemical substance in the Universe, constituting roughly 75% of all baryonic mass.
What is the chemical symbol for caesium?
The chemical symbol for Caesium is Cs. Caesium is a soft, silvery-gold alkali metal with a melting point of 28.5 °C, which makes it one of only five elemental metals that are liquid at or near room temperature. Caesium has physical and chemical properties similar to those of rubidium and potassium. 301.6 K.
What is the most abundant element in the Earth's crust?
Aluminium is a silvery-white, soft, nonmagnetic, ductile metal in the boron group. By mass, aluminium makes up about 8% of the Earth’s crust; it is the third most abundant element after oxygen and silicon and the most abundant metal in the crust, though it is less common in the mantle below.
How many elements are in chemistry?
This list contains the 118 elements of chemistry.
How are the elements of the periodic table sorted?
The chemical elements of the periodic table sorted by melting point
What is the unity used for melting point?
The unity used for the melting point is Celsius (C). Click here: to convert Celsius to Fahrenheit or Kelvin.
How to take care of a benzoic acid and naphthalene sample?
Precautions to be taken during the experiment: Make sure the sample of Naphthalene and Benzoic acid are dry and powdered. Keep the capillary tube and the thermometer at a similar level. Tightly pack the powder into the capillary tube without any air gaps.
How to fill a capillary tube with naphthalene?
Dip the open end of the capillary tube in the finely powdered naphthalene. Gently tap the capillary tube on the table to fill the compound in the capillary tube to about a length of 1–2 cm. With the help of a thread, attach the capillary tube to a thermometer as shown in the figure below.
What acid to dip capillary tube in?
Dip the open end of the capillary tube in the finely powdered Benzoic acid.
How to determine melting point of naphthalene?
Procedure to determine the melting point of Naphthalene: 1 Place the capillary tube in the groove of the aluminium block. 2 Double check that the capillary tube holding the naphthalene is in the middle of the groove. 3 Place the Aluminium block on a tripod stand above the kerosene burner and start heating the block with the burner. 4 Keep continuous watch of the temperature and note the temperature as soon as the substance starts to melt. 5 Make note of the temperature (t1) as soon as the compound begins to melt. 6 Make note of the temperature (t2) as soon as the compound is melted completely. 7 The average of the two readings gives the correct melting point of the substance.
How to make Benzoic acid powder?
Take Benzoic acid on a tile and crush it into a fine powder .
How long to heat a capillary tube?
Take a capillary tube and close its one end by heating the end in the flame for 2-3 minutes while continuously rotating it.
Where is the capillary tube holding the naphthalene?
Double check that the capillary tube holding the naphthalene is in the middle of the groove.