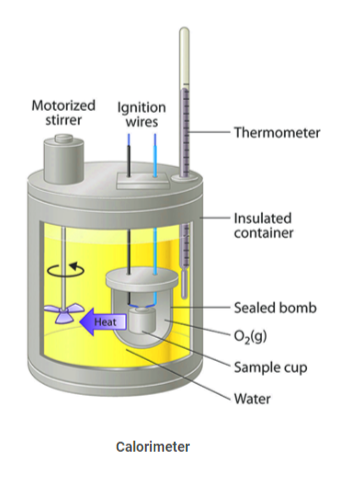
Calorimeter
- A calorimeter is what is used to measure the thermal changes of a body.
- Calorimetry is applied extensively in the fields of thermochemistry in calculating the enthalpy, stability, heat...
What are the advantages of using a calorimeter?
The main advantages of this sort of calibration are as follows:
- It is an absolute calibration.
- The use of standard materials for calibration is not necessary. The calibration is often performed at a continuing temperature, within the heating mode and within the cooling mode.
- It can be applied to any experimental vessel volume.
- It is a very accurate calibration.
How to make a simple calorimeter?
Materials
- 10 whole almonds
- 10 whole peanuts, out of the shell
- 10 whole cashews
- Light or match
- Kitchen or food scale that measures in grams
- Water
- Jug or large container
- Can opener
- Small metal can
- Large metal can, like a coffee can
How do you use a calorimeter?
Calorimetry is used to measure amounts of heat transferred to or from a substance. To do so, the heat is exchanged with a calibrated object (calorimeter). The temperature change measured by the calorimeter is used to derive the amount of heat transferred by the process under study.
Why is using a calorimeter important?
Expert Answers
- Understanding Coffee Cup Calorimetry. ...
- 5.5: Calorimetry - Chemistry LibreTexts. ...
- Solved Does stirring the water in your calorimeter | …. ...
- Experiment 5 Flashcards | Quizlet. ...
- chem 101L Flashcards | Quizlet. ...
- Lab 4 - Calorimetry. ...
- 5.6: Calorimetry - Chemistry LibreTexts. ...
- The Importance Of Calorimetry - 1575 Words | Cram. ...
- HEAT OF COMBUSTION: BOMB CALORIMETER. ...

What is the use of colorimeters in a biochemistry laboratory?
A laboratory colorimeter, also known as a digital colorimeter, is an instrument used to measure the absorption of wavelengths of light by a sample...
How do you use a colorimeter to measure concentration?
Choose the light filter or LED that gives full absorbance. Chart the data in order to get an absorbance graph versus concentration. Then use the in...
Why is colorimetry important?
Colorimetry, wavelength measurement and electromagnetic radiation intensity in spectrum area visible. It is commonly used to classify and assess co...
What are the applications of colorimetry?
Colorimeters are used for a wide variety of applications in the chemical and biological fields including, but not limited to, analysis of blood, wa...
How is colorimetry used in real life?
Unlike spectrophotometry, colorimetry is a method of calculating color values as perceived by the human visual system. This is used in applications...
What Does A Calorimeter Do?
A calorimeter measures the change in heat. Simple calorimeters are made with a metal container of water, positioned above a combustion chamber. A thermometer is used to measure the heat change in the amount of water. The simplest versions of the device can be made at home using two coffee cups or styrofoam cups, though it is not as accurate as lab equipment. There are, however, several other types that are much more complex.
What is the purpose of a calorimeter?
The calorimeter measures the mass of the liquid along with the temperature change, to determine the amount of energy change . It is different from a thermal analysis in that thermal analyzers measure properties of a material at various temperatures.
How does a thermometer measure temperature?
There is a thermometer to measure the temperature of the liquid in the inner container, and a stirrer to stir the liquid to distribute the heat throughout the container. If there is an exothermic reaction, one that releases thermal energy through heat or light, in the solution in the calorimeter, the temperature rises.
How is heat flow determined?
The heat of the reaction is determined by measuring the temperature difference between the heat transfer fluid and the process fluid. It is necessary to know the fill volumes, specific heat, and heat transfer coefficient before a correct answer can be found.
What is the temperature change used to calculate?
The temperature change is used to calculate the enthalpy change per mole of substance A when substances A and B are reacted . The formula is:
What is a constant pressure calorimeter?
It measures the thermodynamic change in a solution, under constant pressure.
How much does a calorimeter cost?
You can expect to spend anywhere from around $15,000 to $40,000 for a brand new calorimeter, depending on type and size. Used models can be found for as little as $9,000.
What is a bomb calorimeter?
One type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid, such as water, that absorbs the heat of the reaction and thus increases in temperature.
Is a bomb calorimeter efficient?
…burned (oxidized) in a bomb calorimeter. However, the human body is not as efficient as a calorimeter, and some potential energy is lost during digestion and metabolism. Corrected physiological values for the heats of combustion of the three energy-yielding nutrients, rounded to whole numbers, are as follows: carbohydrate, 4 kilocalories…
What is Colorimeter?
Colorimetry is one of the methods of spectro analytical methods. The variation of the color of a solution with change in concentration of some solute component in the solution, forms the basis of colorimetry.
What is the colorimeter optical system?
The colorimeter optical system isolates a narrow waveband of light at a selected absorption peak for the chemical that is measured.
How to make a colorimeter more stable?
A more stable colorimeter can be made if the dual beam principle is utilized. This is usually accomplished by comparing the light intensity at an absorption wavelength with the intensity of a reference beam from the same light source.
What is colorimetry in chemistry?
What is Colorimeter? Colorimetry is one of the methods of spectro analytical methods. The variation of the color of a solution with change in concentration of some solute component in the solution, forms the basis of colorimetry. The colour is either the inherent colour of the constituent itself or due to the formation of a coloured species by ...
Why use narrow wavelength?
Use of the narrow wavelength band usually makes possible the elimination of the absorption effects of other chemicals in the solution.