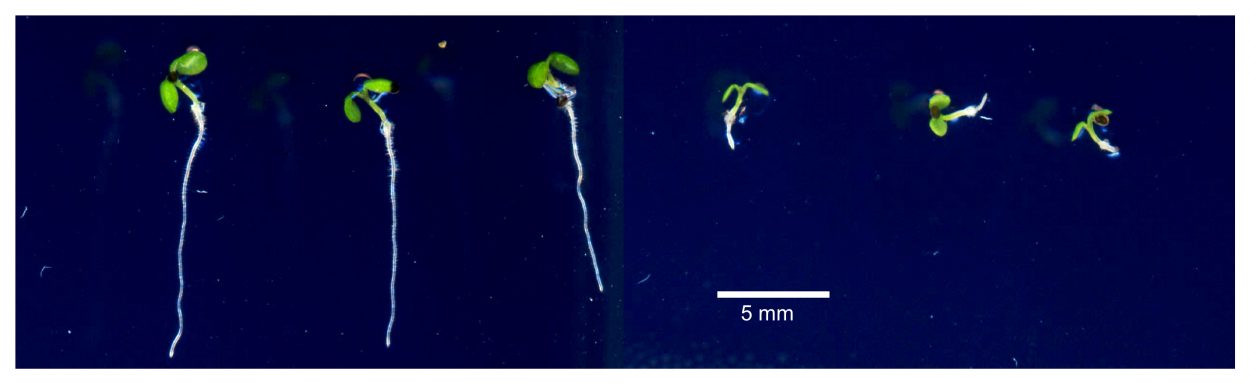
Iron is an essential plant nutrient as it serves as one of the main catalysts for a plant's production of chlorophyll
Chlorophyll
Chlorophyll is any of several related green pigments found in the mesosomes of cyanobacteria, as well as in the chloroplasts of algae and plants. Its name is derived from the Greek words χλωρός, khloros and φύλλον, phyllon. Chlorophyll is essential in photosynthesis, allowi…
How do I add iron to my soil?
How to Add Iron to Garden Soil
- Chelated Iron. As you may have guessed, simply adding pure elemental iron to your garden may not help with an iron deficiency in the soil.
- Iron Sulfate. Iron sulfate is another form of iron that is available to plants. ...
- Blood Meal. Blood meal is made from dried and powdered blood from slaughterhouses. ...
- Compost High in Iron. ...
- Fertilizer High in Iron. ...
What plants are rich in iron?
- Tofu
- Beans (kidney, garbanzo, or white, canned)
- Tomato products (e.g., paste)
- Dried peas
- Dried beans
- Lentils
- Instant breakfast
- Corn syrup
- Maple syrup
- Molasses
What plants have iron in them?
Vegetable Plants That Contain Large Amounts of Iron
- Spinach and Collard Greens. Leafy vegetables featuring a deep, dark green color, such as spinach and collard greens, are usually high in iron.
- Beans. Various beans, including green beans, peas and lentils, contain a higher amount of iron than many other vegetables.
- Potatoes. ...
- Tomatoes. ...
What planet's soil is rich in iron oxide?
The soil on Mars contains a substance called iron oxide, which gives the planet a red color. Like Earth, Mars also has a core, mantle, and crust. Mars is lighter for its size than the other rocky planets.

What is the role of iron in soil?
In plants, iron is also required for photosynthesis and chlorophyll synthesis. The availability of iron in soils dictates the distribution of plant species in natural ecosystems and limits yield and nutritional quality of crops.
Does iron change soil pH?
Iron sulfate and aluminum sulfate lower soil pH faster than elemental sulfur since they require chemical rather than biological reactions.
Is iron good for soil?
Iron is a nutrient that all plants need to function. Many of the vital functions of the plant, like enzyme and chlorophyll production, nitrogen fixing, and development and metabolism are all dependent on iron. Without iron, the plant simply cannot function as well as it should.
How does iron help plants grow?
Plants only need a tiny amount of iron to be healthy, but that small amount is crucial. First of all, iron is involved when a plant produces chlorophyll, which gives the plant oxygen as well as its healthy green color. This is why plants with an iron deficiency, or chlorosis, show a sickly yellow color to their leaves.
Can too much iron hurt plants?
Iron toxicity in soil is not common, but some plants do secrete acids from the roots, which lowers soil pH. These plants can take up too much iron, leading to toxicity. The symptoms of iron toxicity in soil include bronzing and stippling of leaves.
What happens if there is too much iron in soil?
In plants such as rice, elevated soil iron levels cause direct cellular damage by harming fats and proteins, decreasing roots' ability to grow. Yet, some plants appear to have inherent tolerance to high iron levels; scientists wanted to understand why.
Does iron raise or lower pH?
Iron is dissolved by ground waters containing carbon dioxide. Carbon dioxide will lower the pH of groundwater making it acidic. Generally, the lower the water's pH, the higher the iron content because the iron compounds in soils and rocks are dissolved by the acid water.
How can I add iron to my soil naturally?
So, how do you add iron to garden soil? You can add chelated iron powder or blood meal directly to the soil to add iron. You can also add fertilizer or your own compost, as long as the iron content is high enough. Another option is to add chelated iron or iron sulfate in liquid form by spraying the leaves of the plant.
Is iron a good fertilizer?
Iron helps the plant move oxygen throughout the roots, leaves, and other parts of the plant, producing the green color that lets you know your plant is healthy. Many plants also rely on iron to complete the enzyme functions that keep the plant thriving.
What plants need lots of iron?
Vegetables that prefer moderately acidic soils -- usually a soil pH of 5.5 to 6.5 -- prefer more iron than crops that prefer slightly acidic conditions and include turnips, tomatoes, squash and radishes. Those that prefer extremely acidic soils, such as radishes and sweet potatoes, enjoy iron-rich soils the most.
Is iron An fertilizer?
Although not one of the main three components -- nitrogen, phosphorous and potassium -- of most fertilizers, iron is one of the trace nutrients necessary for healthy plants. Iron in fertilizer comes in two main forms: iron sulfate and chelated iron.
How often do you apply iron to plants?
Unlike soil applications which are required only once in the spring, spraying iron on the leaves requires multiple applications for most trees and shrubs. Applications to the leaves may be required four or five times, a few days apart, to get a decent green color again.
What happens when plants lack iron?
The symptoms of iron deficiency appear on the youngest, newest leaves. The area between the leaf veins becomes pale yellow or white (this is called interveinal chlorosis). Usually, no noticeable physical deformity occurs, but in severe cases the youngest leaves may be entirely white and stunted.
What happens to plants without iron?
Without iron, plants would not be able to produce chlorophyll, which gives plants oxygen and its healthy green color. Without iron, the plant can go into a state of chlorosis (lack of chlorophyll, plant is less green), where the leaves will have a sickly yellow color. But iron goes above and beyond chlorophyll.
Is rusty iron good for plants?
Metal rust is not bad for plants as long as the rust does not come from a metal that has toxic materials on it such as oil, paint, or dissolving agents. Rust could even be good for the plants as it would provide iron for the plants through the soil.
Do rusty nails help plants?
They said: “Rusty nails contain iron oxide, a reddish brittle coating, which forms when iron and oxygen react together in the presence of moisture in the air. “Also known as ferric oxide, the rust in nails can provide iron to plants, which is beneficial for plants.”
What fertilizer is high in iron?
Fertilizers that are high in iron include iron sulfates, iron chelates (chelated iron), greensand, iron supplements, blood meal, compost, and manure. Some of these will affect soil pH, so keep this in mind when you apply them, and get a soil test before doing so.
How much iron does a plant need?
Iron is a micronutrient and is required by plants in small amounts. Most annual plants have a requirement for Fe on the order of 1 to 1.5 lb Fe per acre, compared with nitrogen (N) at 80 to 200 lb per acre.
Do trees like iron?
Iron is needed for the production of chlorophyll. Therefore, a lack of iron results in a loss of the green color in the leaves. In severe cases, leaf color may change from yellow to white to brown. If uncorrected, twigs and eventually branches may die leading to the death of the tree.
What makes soil a very dark color?
Organic matter tends to make the soil color darker. Humus, the final stage of organic matter breakdown, is black. Throughout the stages of organic matter breakdown, the colour imparted to the soil varies from browns to black. Sodium content influences the depth of colour of organic matter and therefore the soil.
Is iron rich soil acidic?
Choosing Plants Although iron is plentiful in acidic soils, many types of nutrients become less available in acidic soil, such as magnesium, and certain nutrient deficiencies may occur.
What is the relation between pH and the amount of active iron in soils?
The amount of iron and its availability in soil is influenced by the following: pH ‑ high pH reduces iron availability, low pH increases it. Organic matter ‑ organic matter provides iron and makes it more readily available.
What is the relationship of pH with active Fe?
Solubility of Fe decreases by approximately 1000-fold for each unit increase of soil pH in the range of 4–9 compared to approximately 100-fold decreases in activity of Mn, Cu, and Zn (Lindsay, 1979).
How do you neutralize iron in soil?
Reducing Iron Availability by Adding Bicarbonates Bicarbonates bond to iron in the soil and will prevent the iron from being available to plants. By adding bicarbonate-containing chemicals to the soil, you can reduce the availability of iron to treat iron excess. This works particularly well in soils with a neutral pH.
Why do plants need iron?
As previously stated, it’s mostly to help the plant move oxygen through its system. Plants only need a tiny amount of iron to be healthy, but that small amount is crucial. First of all, iron is involved when a plant produces chlorophyll, which gives the plant oxygen as well as its healthy green color. This is why plants with an iron deficiency, ...
What is the function of iron?
So what is iron? The function of iron is to act much like it does in the human bloodstream — helping to carry important elements through a plant’s circulatory system.
What is Iron and its Function?
So what is iron? The function of iron is to act much like it does in the human bloodstream — helping to carry important elements through a plant’s circulatory system.
What causes iron deficiency in plants?
Soil that is alkaline or has had too much lime added often causes an iron deficiency in the plants in the area. You can correct it easily by adding an iron fertilizer, or evening out the pH balance in the soil by adding garden sulfur.
Where does iron come from?
Iron for plants can come from a number of sources. Ferric oxide is a chemical present in soil that gives dirt a distinctive red color, and plants can absorb iron from this chemical. Iron is also present in decomposing plant matter, so adding compost to your soil or even allowing dead leaves to collect on the surface can help to add iron ...
How to add iron to soil?
So, how do you add iron to garden soil? You can add chelated iron powder or blood meal directly to the soil to add iron. You can also add fertilizer or your own compost, as long as the iron content is high enough. Another option is to add chelated iron or iron sulfate in liquid form by spraying the leaves of the plant.
Why is Iron Important for Plants?
Iron serves several important functions in plants. First of all, iron is involved in the production of chlorophyll through photosynthesis. Remember: chlorophyll is the compound that makes plants green.
What happens if the pH of the soil is too high?
If the pH of your soil is too high, then your plants can end up with an iron deficiency – even if there is plenty of iron in the soil! How can this happen?
What causes iron deficiency in plants?
Extreme weather conditions can also cause iron deficiency in plants. High temperatures or intense sunlight can disrupt a plant’s uptake of iron.
What can block a plant from absorbing another nutrient?
Too much of one nutrient can block a plant from absorbing another. For example, excessive amounts of phosphorus can prevent iron uptake in a plant. This can occur if you use a garden fertilizer with high phosphorus content.
How to find iron deficiency in soil?
The best way to find out is to use a soil test kit, which you can buy at a garden center or online.
Why is my garden soil not iron?
If none of the above conditions apply to your garden, then your soil may contain insufficient iron for your plants. This can occur for a couple of different reasons. One possible explanation is that there was never enough iron in the soil to begin with.
What is ironite used for?
Phosphorus, a key plant nutrient, is also "fixed" in alkaline soils in a form that plants cannot use. Ironite is thus used to adjust the soil's pH to make it less alkaline and to allow the plant to more easily absorb iron and phosphorus.
Why is fertilizer important for gardeners?
Fertilizers can foster healthy plant growth, increase disease resistance in plants, stave off insect infestations and offer many other important benefits .
Where is ironite made?
Ironite is a material commercially sold as a fertilizer. It is made from waste products from the Iron King large-scale mine located in Arizona and other large-scale mining operations. Controversy has surrounded Ironite for many years because it contains arsenic, lead and other heavy metals. Though producers claim that the heavy metals are in a form that do not pose a threat to human health or the environment at large, concerns over the leaching of heavy metals from Ironite use has led to a ban on Ironite fertilizer in Canada and prolonged research by the Environmental Protection Agency (EPA).
Does ironite fertilizer affect oats?
Indeed, the oat crops treated with Ironite had higher yields , possibly due to the use of the Ironite fertilizer.
Is ironite fertilizer banned in Canada?
Though producers claim that the heavy metals are in a form that do not pose a threat to human health or the environment at large, concerns over the leaching of heavy metals from Ironite use has led to a ban on Ironite fertilizer in Canada and prolonged research by the Environmental Protection Agency (EPA). Advertisement.
Is ironite soluble in humans?
The EPA has disputed claims by Ironite manufacturers that arsenic and lead in Ironite exist in forms that are not hazardous to humans and the environment, citing research that demonstrates that heavy metals in Ironite actually are in a form that is soluble in humans and animals. Advertisement.
Why is iron rich soil important?
Iron-rich soils are so named for their high content of iron mineral deposits, and their nutrient composition impacts their ability to be cultivated to support plant life.
What is iron rich soil?
Iron-rich soil is often the result of high levels of iron deposits beneath the top layers of soil. A majority of iron deposits are formed in sedimentary rock beds beneath the Earth's surface, called banded iron formations. Another source of iron-rich soil is magma flow from erupted volcanoes.
What is the source of iron in soil?
Another source of iron-rich soil is magma flow from erupted volcanoes. The liquid magma contains high levels of magnetite phenocrysts that harden over time into magnetite iron. The high iron levels mix with other nutrients and organic materials in the soil to create an iron-rich top soil or clay. Advertisement.
What is the most distinctive characteristic of an iron-rich soil?
Identification. The most distinctive characteristic of an iron-rich soil is a ruddy orange or red color , though not all red soils are rich in iron. When the iron deposits in the soil oxidize, they turn a distinctive rust color that tints the soil red.
Why is iron important for plants?
Iron is an essential plant nutrient as it serves as one of the main catalysts for a plant's production of chlorophyll, but high levels of iron in soil reduce the levels of other necessary soil elements. When iron levels are too high, organic matter that sustains plant life is reduced and the soils is unsuitable for most crops.
What happens when iron levels are too high?
When iron levels are too high, organic matter that sustains plant life is reduced and the soils is unsuitable for most crops. Though some plants and trees like the Joshua tree, fruit-bearing trees, huckleberry and black-eyed Susan can withstand high levels of iron, treating iron-rich soil with a fertilizer with high organic content combats ...
Why is soil orange?
Because other factors like high levels of sediment or low levels of rainfall can make a soil appear red or orange, soil testing is the only definitive way to determine whether or not red soil is iron-rich. Advertisement.
What is the role of iron in soil?
Iron is a necessary component for the formation of chlorophyll, so it impacts a plant’s ability to harness the energy of the sun.
What is the best soil amendment?
A popular choice as a soil amendment is powdered or granular chelated iron. Correcting other soil imbalances, like pH and the other aforementioned nutrients, can also go a long way toward preventing iron chlorosis. FOLLOW. MORE ARTICLES.
Is red soil iron rich?
Soils can have a lot of iron, but plants growing in them can still be iron-deficient. Red soils, for example, are typically iron -rich, but often it is in the insoluble form of the mineral. Iron is one of the most common micronutrients that plants can be deficient in, which is probably why the older Irish lady suggested burying a piece ...
Can you add iron to soil without a soil test?
Even without a comprehensive soil test, a diagnosis of iron deficiency is often right on the money. Adding iron to the soil can be as simple as using scrap pieces of iron, but sharp, rusted pieces of metal in the garden carry their own risks. A popular choice as a soil amendment is powdered or granular chelated iron.
How to deal with too much iron in soil?
To deal with too much iron in your garden’s soil, you’ll want to raise the pH to a more neutral level, aerate the water, and make sure all other nutrients are available in sufficient quantities.
What is iron used for?
Iron is used as a catalyst to synthesize chlorophyll, which is required for photosynthesis. Without chlorophyll, plants would not be able to produce enough energy to grow. Iron is also used for a plant’s protein synthesis and for nitrogen fixation and reduction.
How many micronutrients are needed for plants?
The seven micronutrients needed by plants need to be balanced. Generally, toxicity or excess of one nutrient leads to a deficiency in others. Almost all micronutrient deficiencies are caused by another micronutrient’s excess.
How to treat iron toxicity?
All micronutrient deficiencies and imbalances, including iron toxicity, can be treated in approximately the same way: 1 Make sure all minerals are present in water. Don’t use reverse osmosis water, which has all of the nutrients removed. You can add trace mineral drops to add these micronutrients back into the water if they are not present. 2 Test the pH of water and soil. Make sure it is in the 6-7 range, or a range suitable for your plant. Test the micronutrient content of the soil and the plant to make sure all the micronutrients are present and being absorbed. 3 Flush the soil with water to remove all nutrients that have built up and to break any nutrient lockout. Wait for the soil to dry before watering the plant again.
How to tell if a plant has iron poisoning?
The plants in your garden may be suffering from iron toxicity if leaves show signs of bronzing (that is, getting darker), if brown spots form on the leaves, or if the plant’s growth becomes stunted. Bronzing is the most common symptom.
Why are minerals called micronutrients?
These minerals are called micronutrients because they are only needed in small quantities by the plant. Other nutrients such as nitrogen, phosphorus, or potassium are called macronutrients and are needed in much greater amounts.
What are the micronutrients that plants need to grow?
Iron is one of seven micronutrients that plants need to grow. Other micronutrients include copper, cobalt, boron, molybdenum, manganese, and zinc. These nutrients are found in varying small amounts in soil all over the planet and in trace amounts in water sources.
Why is Iron Important for Your Lawn?
Iron supplements for lawns became especially popular with homeowners trying to rival the lovely green grass of golf courses.
How to apply iron to lawn?
Apply granular iron supplements directly on the lawn soil with a broadcast or drop spreader. This type of iron supplement for lawns will take longer to have an effect since the structure of its molecules needs to be broken down, which happens over time.
What is the best product to use to improve lawn health?
Products like spray-on Ironite and LawnStar Chelated Liquid Iron (the liquid Iron product I use, here’s the Amazon link) act quickly and can be useful to improve your lawn’s health and appearance.
What is organic iron fertilizer?
Organic Iron Fertilizer Products. Organic iron fertilizer is comprised of organic iron sources, such as those found in Greensand and Milorganite. These break down more slowly but are healthier for your lawn in the long term. The iron in them also has natural chelation, which makes it easier for your grass to absorb.
Why is my lawn green?
Iron application can help make your lawn greener and healthier. If you’ve noticed that your lawn’s color is a lighter green or even bordering on yellow, it’s possible that your soil has an iron deficiency. Properly adding iron to your lawn requires careful application and timing. Knowing when to apply iron to lawn areas will get you the best results to keep a green lawn all summer. That said, applying Iron at the wrong time can do more harm than good.
Why is my lawn yellow?
In fact, it’s usually only beneficial to apply extra iron if you have a soil test indicating iron deficiency. Don’t automatically assume an iron deficiency is the cause of your lawn yellowing or being light in color. First, ensure that it’s sufficiently watered and has all other major nutrients.
What is liquid iron spray?
Using liquid iron in a spray form facilitates foliar feeding. This kind of feeding tends to have a more efficient and expedient effect on your lawn (you’ll see results quickly).
What is the role of organic matter in soil?
Organic matter plays an indirect, but crucial role in the removal of iron and manganese pigments in wet soils. All bacteria, including those that reduce iron and manganese, must have a food source. Therefore, anaerobic bacteria thrive in concentrations of organic matter, particularly in dead roots.
Why do soil scientists describe soil color?
Soil color can indicate the composition of the soil and give clues to the conditions of the soil. Varying horizontal bands of color in the soil often identify a specific soil horizon. They can tell us the development and distribution of color in soil results from chemical and biological weathering, especially redox reactions. As the primary minerals in soil parent material weather.
How to measure soil color?
Another way soil scientist measure soil color is through the use of spectroscopically (proximal soil sensing) or color obtained from soil profile photographs. These are optical sensors that are used to determine the soil’s ability to reflect light in different parts of the electromagnetic spectrum. Proximal optical sensors are fundamentally the same as remote sensing systems. The advantage of proximal sensors is that they can be applied at the surface and below ground. In soil survey, optical reflectance is applicable to point data documentation. It can be used for on-the-go measurements during different soil survey practices. In addition, both near and mid infrared diffuse reflectance spectroscopy are being used in the laboratory for rapid determination of some soil properties. Optical sensing systems cover the ultraviolet (100–400 nm), visible (400–750 nm), near infrared (750–2,500 nm), or mid infrared (2,500–25,000 nm) wavelengths or a combination of these wavelengths. Typically, instruments used for soil measurements include their own light source (e.g., a light bulb or light-emitting diode). Photodiodes or array detectors are used to estimate the intensity of reflected light and relate this measure to the light reflected from a given set of standards. Both source and reflected light can be transmitted through the air, via fiber optics, or when feasible, through a contact window fabricated from highly resistive material, such as sapphire or quartz. These proximal soil sensors allow rapid and inexpensive collection of precise, quantitative, high-resolution data, which can be used to better understand soil spatial and temporal variability.
Why is my soil yellow?
Yellow or red soil indicates the presence of oxidized ferric iron oxides. The red color might be mainly due to ferric oxides occurring as thin coatings on the soil particles while the iron oxide occurs as hematite or as hydrous ferric oxide, the color is red and when it occurs in the hydrate form as limonite the soil gets a yellow color.
What does soil color tell us?
Soil Color tells us stories on how the soil formed. The first impression we have when looking at bare earth or soil is of color. Bright colors especially, catch our eye and soil color can vary with ecosystems. The stories soil colors tell us vary with the ecosystems in which the soils formed and are influenced by parent material ...
Why is my soil brown?
Brown soils might be brown from decaying plant material. The darker color often indicates an increase in decomposed organic matter known as humus. Soil has living organisms and dead organic matter, which decomposes into black humus.
What is the color of grassland soil?
In grassland (prairie) soils the dark color permeates through the surface layers bringing with it nutrients and high fertility Deeper in the soil, the organic pigment coats surfaces of soil, making them darker than the color inside. Humus color decreases with depth and iron pigments become more apparent.
