What is the rate determining step in an S n 1 reaction?
In an S N 1 reaction, the rate determining step is the loss of the leaving group to form the intermediate carbocation. The more stable the carbocation is, the easier it is to form, and the faster the S N 1 reaction will be.
What does SN1 mean in chemistry?
SN1 reaction. The SN1 reaction is a substitution reaction in organic chemistry. "SN" stands for "nucleophilic substitution", and the "1" says that the rate-determining step is unimolecular. Thus, the rate equation is often shown as having first-order dependence on electrophile and zero-order dependence on nucleophile.
What happens in Step 2 of SN1 reaction?
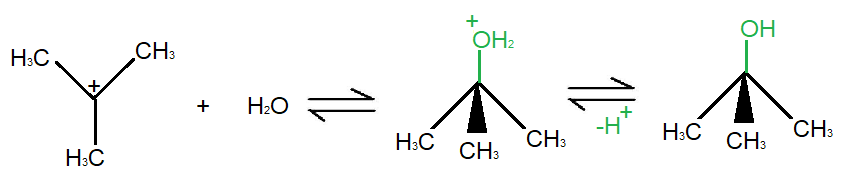
Step 2 1 In the second step of the SN1 reaction mechanism, the carbocation is attacked by the nucleophile. 2 Since water is used as a solvent, an oxonium ion intermediate is formed. 3 Since the solvent is of a neutral nature, a third step where deprotonation occurs is necessary. More ...
What is the effect of solvent on s n 1 reaction?
Effect of Solvent A solvent that can facilitate the formation of the carbocation intermediate will speed up the rate determining step of the S N 1 reaction. The preferred solvents for this type of reaction are both polar and protic.
What affects rate of reaction in SN1 reactions?
In the case of SN1 eactions, polar protic solvents speed up the rate of SN1 reactions because the polar solvent helps stabilize the transition state and carbocation intermediate. Since the carbocation is unstable, anything that can stabilize this even a little will speed up the reaction.
Does SN1 rate depend on nucleophile?
The Mechanism Of The SN1 Is Stepwise. The SN2 reaction is concerted. That is, the SN2 occurs in one step, and both the nucleophile and substrate are involved in the rate determining step. Therefore the rate is dependent on both the concentration of substrate and that of the nucleophile.
What increases the rate of SN1?
The rates of SN1 reactions are generally increased by the use of a highly polar solvent, including protic (hydrogen bonding) solvents such as water or ethanol.
On which factors SN1 and sn2 reaction rate depends?
A polar solvent stabilizes nucleophile (one of the reactants) by solvation. Thus, solvent deactivates the nucleophile by stabilizing it. Hence, aprotic solvents or solvents of low polarity will favour the SN2 mechanism. SN1 mechanism: SN1 mechanism proceeds via the formation of a carbocation intermediate.
What drives an SN1 reaction?
Greater nucleophilicity of the attacking group than the nucleophilicity of the leaving group causes SN1 reactions. The cleavage of the C-X bond keeps occuring always, both the reactant and the product remaining in the solution. The equilibrium shifts towards the final product.
What will be the effect on rate of reaction of SN1 if the concentration of nucleophile is doubled?
solution: The rate equation of an SN1 reaction is: k [R-X]. Notice that the nucleophile is not in the rate equation. Therefore, doubling the concentration of the nucleophile will have no effect on the rate of the reaction.
How does nucleophile affect SN2 reaction?
Nucleophilicity Because the nucleophile is involved in the rate-determining step of SN2 reactions, stronger nucleophiles react faster. Stronger nucleophiles are said to have increased nucleophilicity.
What is the second step of the SN1 reaction?
In the second step of the SN1 reaction mechanism, the carbocation is attacked by the nucleophile. Since water is used as a solvent, an oxonium ion intermediate is formed. Since the solvent is of a neutral nature, a third step where deprotonation occurs is necessary.
What is the S N 1 reaction?
The S N 1 reaction is often referred to as the dissociative mechanism in inorganic chemistry.
What does S N 1 stand for?
S N 1 stands for substitution nucleophilic unimolecular. Thus, the rate equation (which states that the S N 1 reaction is dependent on the electrophile but not on the nucleophile) holds in situations where the amount of the nucleophile is far greater than the amount of the carbocation intermediate. This reaction involves the formation ...
How does the S N1 reaction work?
Then the carbocation is attacked by the nucleophile. Finally, the deprotonation of the protonated nucleophile takes place to give the required product. The rate determining step of this reaction depends purely on the electrophilicity of the leaving group and is not impacted at all by the nucleophile.
What are some examples of solvents used in S N 1 reactions?
Examples of solvents used in S N 1 reactions include water and alcohols . These solvents also act as nucleophiles.
What is the carbocation intermediate formed in step 1 of the S N 1 reaction mechanism?
The carbocation intermediate formed in step 1 of the S N 1 reaction mechanism is an sp2 hybridized carbon. Its molecular geometry is trigonal planar, therefore allowing for two different points of nucleophilic attack, left and right.
What happens to the carbocation reaction when it takes place at a stereocenter?
If the reaction takes place at a stereocenter and if neither avenue for nucleophilic attack is preferred, the carbocation is then attacked equally from both sides , yielding an equal ratio of left and right-handed enantiomers as shown below.
What does SN1 mean in chemistry?
The SN1 reaction is a substitution reaction in organic chemistry, the name of which refers to the Hughes-Ingold symbol of the mechanism. "S N " stands for " nucleophilic substitution ", and the "1" says that the rate-determining step is unimolecular. Thus, the rate equation is often shown as having first-order dependence on electrophile ...
Why does the S N 1 mechanism dominate?
The S N 1 mechanism tends to dominate when the central carbon atom is surrounded by bulky groups because such groups sterically hinder the S N 2 reaction. Additionally, bulky substituents on the central carbon increase the rate of carbocation formation because of the relief of steric strain that occurs.
How is carbocation intermediate formed?
The carbocation intermediate is formed in the reaction's rate determining step is an sp2 hybridized carbon with trigonal planar molecular geometry. This allows two different ways for the nucleophilic attack, one on either side of the planar molecule. If neither way is preferentially favored, these two ways occur equally, yielding a racemic mixture of enantiomers if the reaction takes place at a stereocenter. This is illustrated below in the S N 1 reaction of S-3-chloro-3-methylhexane with an iodide ion, which yields a racemic mixture of 3-iodo-3-methylhexane:
What solvents act as nucleophiles?
Typical polar protic solvents include water and alcohols, which will also act as nucleophiles and the process is known as solvolysis. The Y scale correlates solvolysis reaction rates of any solvent ( k) with that of a standard solvent (80% v/v ethanol / water) ( k0) through.
Is the rate equation steady state?
Instead, the rate equation may be more accurately described using steady-state kinetics. The reaction involves a carbocation intermediate and is commonly seen in reactions of secondary or tertiary alkyl halides under strongly basic conditions or, under strongly acidic conditions, with secondary or tertiary alcohols.
Can an alkene be formed in a cold reaction?
Even if the reaction is performed cold, some alkene may be formed. If an attempt is made to perform an S N 1 reaction using a strongly basic nucleophile such as hydroxide or methoxide ion, the alkene will again be formed, this time via an E2 elimination. This will be especially true if the reaction is heated.
Is the rate law of the S N 1 reaction first order?
Although the rate law of the S N 1 reaction is often regarded as being first order in al kyl halide and zero order in nucleophile, this is a simplification that holds true only under certain conditions. While it too is an approximation, the rate law derived from the steady state approximation (SSA) provides more insight into the kinetic behavior of the S N 1 reaction. Consider the following reaction scheme for the mechanism shown above:
What is SN1 reaction?
SN1 Reaction is a Nucleophilic Substitution reaction (A class of reactions wherein the electron-rich nucleophile attacks a positively charged electrophile to replace a leaving group) in which the rate-determining step involves 1 component. S N 1 reactions are unimolecular and proceed through an intermediate carbocation. The first step, being slower, determines the rate.
What does S N 1 mean?
The S N 1 reaction is a substitution reaction, the name of which refers to the Hughes-Ingold symbol of the mechanism. “S N ” stands for “ substitution nucleophilic “, and the “1” says that the rate-determining step is unimolecular.
What is the first step in an organic reaction?
This type of mechanism involves two steps. 1. The first step is the ionization of alkyl halide in the presence ...
Which solvents are polar enough to dissolve the substrate and nucleophile?
The S N 2 reaction is favored by polar aprotic solvents – these are solvents such as acetone, DMSO, acetonitrile, or DMF that are polar enough to dissolve the substrate and nucleophile but do not participate in hydrogen bonding with the nucleophile.
What happens when you start with an enantiomer?
If we start with an enantiomerically pure product, (that is, one enantiomer ), these reactions tend to result in a mixture of products where the stereochemistry is the same as the starting material (retention) or opposite (inversion). In other words, some degree of racemization will take place.
Rate Law for Sɴ1 Reaction Definition
Rate Law for Sɴ1 Reaction Definition S N 1 { {\rm {S}}_ {\rm {N}}}1 SN 1 is a very common reaction in organic chemistry. Its is a nucleophilic substitution reaction. The term S refers to substitution, the term N refers to nucleophilic and the number 1 indicates that the rate determining step of this reaction is unimolecular.
Overview of Rate Law For Sn1 Reaction
The S N 1 { {\rm {S}}_ {\rm {N}}}1 SN 1 reaction has a multiple step mechanism in which there is formation of a carbocation. The rate of this nucleophilic substitution reaction depends on the stability of the carbocation. Solvolysis reactions follow S N 1 { {\rm {S}}_ {\rm {N}}}1 SN 1 mechanism.
Rate Law for Sɴ1 Reaction
Rate law of the unimolecular nucleophilic substitution reaction is only sensitive to the concentration of the substrate. It does not depend on the concentration of the nucleophile. Therefore, if the concentration of the substrate is doubled then the rate of the reaction increases by a factor of two.
Unimolecular Nucleophilic Substitution Reaction Mechanism
The carbon-halogen bond in an alkyl halide is a polar bond. The breaking of this bond facilitates the removal of halide group. When the halide ions leave the tertiary alkyl halide, a carbocation is formed. This carbocation is very reactive and readily accepts any nucleophile. So, even a weak nucleophile is capable of attacking this intermediate.
Effect of Solvent on Sɴ1 Reaction
Any solvent that is capable of forming a carbocation quickly is responsible for speeding up the rate determining step of unimolecular nucleophilic substitution reaction. The ideal solvent for unimolecular nucleophilic substitution reaction are polar protic solvents.
Table of Contents
What Is An SN1 reaction?
- The SN1 reaction is a nucleophilic substitution reaction where the rate-determining step is unimolecular. It is a type of organic substitution reaction. SN1 stands for substitution nucleophilic unimolecular. Thus, the rate equation (which states that the SN1 reaction is dependent on the electrophile but not on the nucleophile) holds in situations w...
SN1 Reaction Mechanism
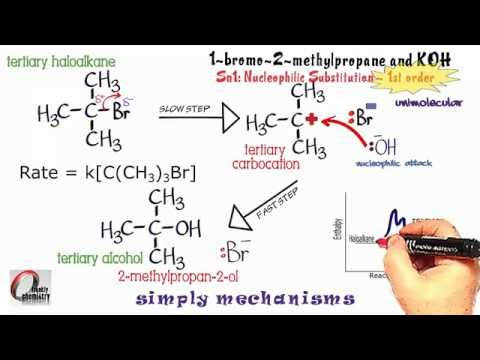
- Taking the hydrolysis of tertiary butyl bromide as an example, the mechanism of the SN1 reaction can be understood via the following steps.
Stereochemistry of SN1 Reaction
- The carbocation intermediate formed in step 1 of the SN1 reaction mechanism is an sp2 hybridized carbon. Its molecular geometry is trigonal planar, therefore allowing for two different points of nucleophilic attack, left and right. If the reaction takes place at a stereocenter and if neither avenue for the nucleophilic attack is preferred, the carbocation is then attacked equally f…
Overview
The SN1 reaction is a substitution reaction in organic chemistry, the name of which refers to the Hughes-Ingold symbol of the mechanism. "SN" stands for "nucleophilic substitution", and the "1" says that the rate-determining step is unimolecular. Thus, the rate equation is often shown as having first-order dependence on the substrate and zero-order dependence on the nucleophile. This relationship holds for situations where the amount of nucleophile is much greater than that …
Mechanism
An example of a reaction taking place with an SN1 reaction mechanism is the hydrolysis of tert-butyl bromide forming tert-butanol:
This SN1 reaction takes place in three steps:
• Formation of a tert-butyl carbocation by separation of a leaving group (a bromide anion) from the carbon atom: this step is slow.
Rate law
Although the rate law of the SN1 reaction is often regarded as being first order in alkyl halide and zero order in nucleophile, this is a simplification that holds true only under certain conditions. While it, too, is an approximation, the rate law derived from the steady state approximation (SSA) provides more insight into the kinetic behavior of the SN1 reaction. Consider the following reaction sche…
Scope
The SN1 mechanism tends to dominate when the central carbon atom is surrounded by bulky groups because such groups sterically hinder the SN2 reaction. Additionally, bulky substituents on the central carbon increase the rate of carbocation formation because of the relief of steric strain that occurs. The resultant carbocation is also stabilized by both inductive stabilization and hyper…
Stereochemistry
The carbocation intermediate formed in the reaction's rate determining step (RDS) is an sp hybridized carbon with trigonal planar molecular geometry. This allows two different ways for the nucleophilic attack, one on either side of the planar molecule. If neither approach is favored, then these two ways occur equally, yielding a racemic mixture of enantiomers if the reaction takes place a…
Side reactions
Two common side reactions are elimination reactions and carbocation rearrangement. If the reaction is performed under warm or hot conditions (which favor an increase in entropy), E1 elimination is likely to predominate, leading to formation of an alkene. At lower temperatures, SN1 and E1 reactions are competitive reactions and it becomes difficult to favor one over the other. Even if the reaction is performed cold, some alkene may be formed. If an attempt is made to per…
Solvent effects
Since the SN1 reaction involves formation of an unstable carbocation intermediate in the rate-determining step (RDS), anything that can facilitate this process will speed up the reaction. The normal solvents of choice are both polar (to stabilize ionic intermediates in general) and protic solvents (to solvate the leaving group in particular). Typical polar protic solvents include water and alcohols, which will also act as nucleophiles, and the process is known as solvolysis.
See also
• Arrow pushing
• Nucleophilic acyl substitution
• Neighbouring group participation
• SN2 reaction
What Is SN1 reaction?
- The SN1 reaction is a substitution reaction, the name of which refers to the Hughes-Ingold symbol of the mechanism. “SN” stands for “substitution nucleophilic“, and the “1” says that the rate-determining step is unimolecular. The organic reaction involves a carbocation intermediate and is commonly seen in reactions of secondary or tertiary alkyl halidesunder strongly basic condition…
SN1 Reaction Mechanism
- To understand the mechanism of SN1 reactions, let us take the example of hydrolysis of tertiary butyl bromide 1. Formation of a tert-butyl carbocation by separation of a leaving group (a bromide anion) from the carbon atom. This step is slow. 1. Nucleophilic attack: the carbocation reacts with the nucleophile. If the nucleophile is a neutral molecule (i.e. a solvent) a third step is required to …
Characteristics of SN1 Reaction
- This is a two-step reaction process.
- Unimolecular and follows first-order kinetics
- Rate of the reaction depends on the concentration of the substrate (alkyl halide)
- Has a racemization stereochemistry, i.e., both retention and inversion products are formed
Stereochemistry of SN1 Reaction
- If we start with an enantiomerically pure product, (that is, one enantiomer), these reactions tend to result in a mixture of products where the stereochemistry is the same as the starting material (retention) or opposite (inversion). In other words, some degree of racemization will take place. However, an excess of one stereoisomer can be observed, as the leaving group can remain in pr…
Factors Affecting SN1 Reaction Mechanism
- The reaction is favoured by a highly substituted alkyl halide and ideally the one which will not lead to rearrangement
- A non-basic nucleophile is preferred (to reduce the elimination E1side reaction)
- A good leaving group is preferred (like Iodine or Bromine)
- Polar protic solvents are most effective and suitable ones for the reaction.
Examples
- Question 1. Rank the following by increasing reactivity in an SN1 reaction. Solution. By considering the stability of the intermediate carbocation. A < D < B < C (most reactive) Question 2. 3-bromo-1-pentene and 1-bromo-2-pentene undergo SN1 reaction at almost the same rate, but one is a secondary halide while the other is a primary halide. Explain why? Solution.They both ha…