
Full Answer
Is the reaction between furan and maleic anhydride thermodynamically stable?
Nov 10, 2021 · What happens when furan reacts with maleic anhydride? Furan and maleic anhydride were converted to phthalic anhydride in two reaction steps: Diels–Alder cycloaddition followed by dehydration. It is shown that this reaction is resistant to thermal runaway because of its reversibility and exothermicity.
What is the reaction between diene and maleic anhydride?
Jun 23, 2020 · What happen when furan is treated with maleic anhydride? Furan reacts as a diene with powerful dienophiles like maleic anhydride , maleimide and benzyne to give Diels–Alder adducts. The kinetically favoured products are the endo adducts but the exo adducts are thermodynamically preferred (Scheme 44).
What is furan-dienophile-anhydride reaction?
Aug 07, 2021 · Answer to Question #224160 in Organic Chemistry for Prince. 1)When furan is treated with the mixture of hcn and hcl? 2)When furan treated with maleic anhydride? 1)Pyrroles and furans also undergo the Gattermann aldehyde synthesis: with HCl and HCN, furan gives furfuraldehyde. 2)Furan reacts as a diene with powerful dienophiles like maleic anhydride, …
Is the endo product for cyclopentadiene + maleic anhydride kinetically favored at room temperature?
What happens when furan reacts with maleic anhydride?
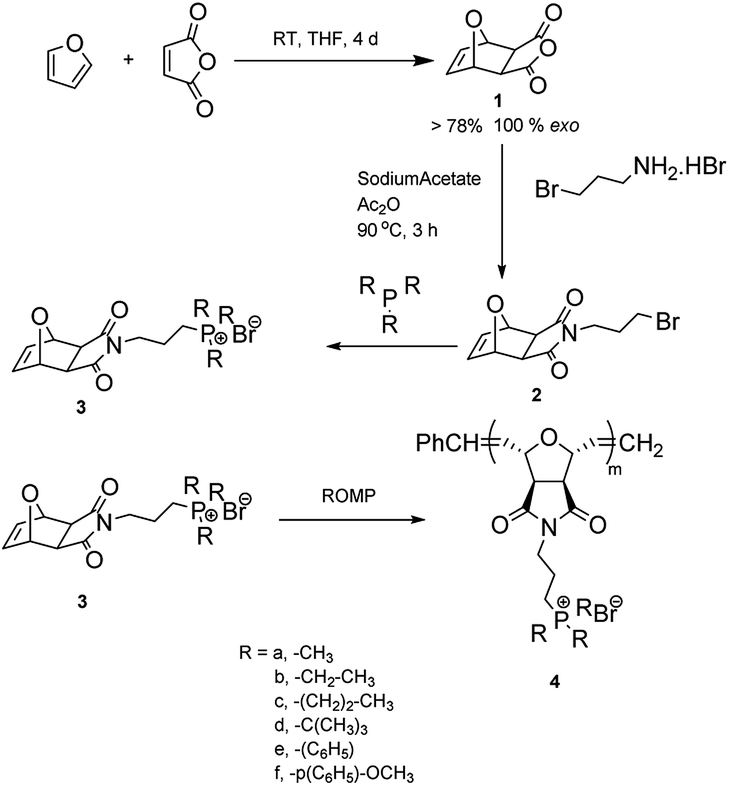
16 Furan and maleic anhydride can be used to produce phthalic anhydride (4) in a two-step process: Diels–Alder cyclo- addition to produce 3 followed by dehydration. A related product that can be prepared following this approach is the synthesis of p-xylene from dimethylfuran and ethylene.Oct 14, 2013
When furan and maleic acid react with each other which reaction take place?
DIELS-ALDER MECHANISM It is a concerted cyclization reaction which generates an adduct of the starting diene (furan) and dienophile (maleic anhydride) that contains two new C−C bonds.Apr 28, 2016
Does anthracene react with maleic anhydride?
Explanation: The reaction takes place in two steps. The maleic anhydride and anthracene initially react in a fast step to form a charge transfer complex. The maleic anhydride and anthracene react to form the product.Dec 1, 2016
Does cyclopentadiene react with maleic anhydride?
In this experiment you will react cyclopentadiene (the diene) with maleic anhydride (the dienophile) to produce the bicyclic compound, endo-bicyclo[2.2. 1]hept-5-ene-2,3-dicarboxylic anhydride. The endo-adduct is formed exclusively.
What is a furan ring?
Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen atom. Chemical compounds containing such rings are also referred to as furans. Furan is a colorless, flammable, highly volatile liquid with a boiling point close to room temperature.Aug 13, 2018
Why is the exo product thermodynamically more stable?
The exo product is most stable as the smaller one-atom bridge eclipses the anhydride ring causing less steric hindrance, and is the thermodynamic product. Under reversible conditions, the exo product is formed.
What happens in a Diels-Alder reaction?
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. Since the reaction involves the formation of a cyclic product via a cyclic transition state, it is also referred to as a "cycloaddition".
Why is xylene Dimethylbenzene used in the Diels-Alder reaction?
Xylene (dimethylbenzene) is used as a high-boiling solvent so that the reaction will work fast enough to complete conveniently. In terms of activation, notice that maleic anhydride is a highly reactive dienophile, due to the presence of two electron-withdrawing carbonyl substituents.
Does the Diels-Alder reaction have Stereoselectivity?
Stereoselectivity: The Diels-Alder reaction is stereospecific with respect to both the diene and the dienophile. Addition is syn on both components (bonds form from same species at the same time)
Why is cyclopentadiene so reactive?
Diels–Alder reactions Cyclopentadiene is a highly reactive diene in the Diels–Alder reaction because minimal distortion of the diene is required to achieve the envelope geometry of the transition state compared to other dienes.
What is Diels-Alder reaction explain with suitable examples?
In organic chemistry, the Diels–Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene derivative. It is the prototypical example of a pericyclic reaction with a concerted mechanism.
Why is Diels-Alder reaction useful?
These reactions proceed under mild conditions, give high yields, and form only inoffensive by-products. The Diels-Alder cycloaddition is one of the click reactions that do not require any metal catalyst; it is one of the most useful reactions in synthetic organic chemistry and material design.