
The ph in our bodies is close to neutral. For example, the pH of blood should be around 7.4. The only exception is the stomach, where stomach acids can even reach a pH of 1. There are three different theories that define what an acid and base is:
What foods have high pH?
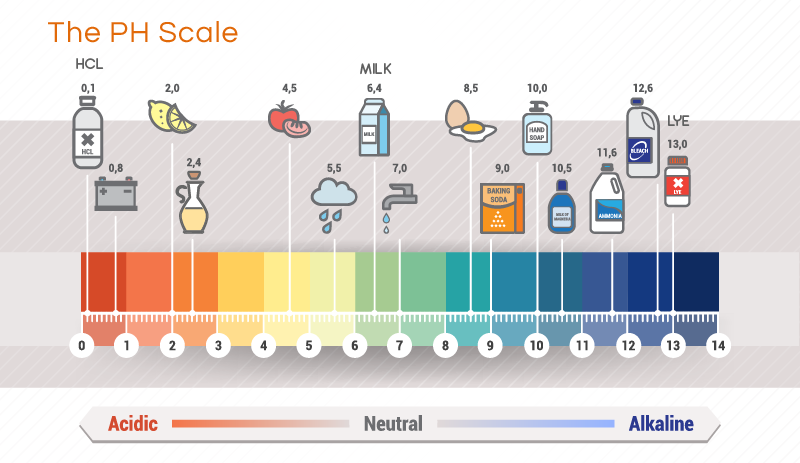
What has a pH of 1? Stomach acid Increasing pH (Decreasing Acidity) Substances 0 (most acidic) Hydrochloric acid (HCl) 1 Stomach acid 2 Lemon juice 3 Cola, beer, vinegar. Can you have a pH of 1? The pH scale is often said to range from 0 to 14, and most solutions do fall within this range, although it’s possible to get a pH below 0 or above 14.
What substance has a pH level of 1?
· Detailed Description. pH is a measure of how acidic/basic water is. The range goes from 0 - 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. pH is really a measure of the relative amount of free hydrogen and hydroxyl ions in the water. Water that has more free hydrogen ions is ...
What acid would have the pH of 1?
Typical acid rain has a pH value of 4.0. A decrease in pH values from 5.0 to 4.0 means that the acidity is 10 times greater. How pH is Measured. There are many high-tech devices that are used to measure pH in laboratories. One easy way that you can measure pH is with a strip of litmus paper. When you touch a strip of litmus paper to something ...
What chemicals are high in pH?
The pH scale, with examples of common solutions and their pH values. Download/View. For commercial use please contact us.

What had a pH of 1?
Stomach acidThe pH scaleIncreasing pH (Decreasing Acidity)Substances0 (most acidic)Hydrochloric acid (HCl)1Stomach acid2Lemon juice3Cola, beer, vinegar15 more rows•Mar 26, 2016
What is a pH of 1 considered?
Anything below 7.0 (ranging from 0.0 to 6.9) is acidic, and anything above 7.0 (from 7.1 to 14.0) is alkaline. The blood in your veins is slightly alkaline (pH = 7.4). The environment in your stomach is highly acidic (pH = 1 to 2).
Is pH less than 1 possible?
Summary of Negative pH Negative pH is possible and simple to calculated, but not something you can easily measure. Special electrodes are used to assess extremely low pH values. In addition to negative pH, it's also possible for pH to have a value of 0.
Is 1.0 A high pH?
A solution that has a pH of 1.0 has 10 times the [H +] as a solution with a pH of 2.0, which in turn has 10 times the [H +] as a solution with a pH of 3.0 and so forth....Example 12.SubstancepH1.0 M NaOH14.0*Actual values may vary depending on conditions.12 more rows
What is the pH of vinegar?
2–3Vinegar is mildly acidic with a pH of 2–3. Apple cider vinegar is slightly more alkaline than pure vinegar because it contains more alkaline nutrients.
What is the pH of soda?
around 2.5 to 3.5Most carbonated drinks, which are approximately 94% water, have pH around 2.5 to 3.5, and hence are highly acidic.
What is the lowest pH ever recorded?
The pH values recorded here are the lowest yet reported anywhere that we know of for any acid mine water. The lowest pH of -3.6 is the lowest known for any water in the environment.
Is milk an acid or base?
Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it's acid-forming or alkaline-forming.
What is the pH of battery acid?
Typically referring to the type of acid used in rechargeable lead-acid batteries, like the ones used in cars, battery acid is made of sulphuric acid (H2SO4) that has been diluted with purified water to a concentration of around 30-50%. In this context, battery acid has an acidic pH of 0.8.
What is the pH of lemon juice?
between 2 and 3Because they contain a high amount of citric acid, lemons have an acidic pH. Lemon juice has a pH between 2 and 3, which means it's 10,000–100,000 times more acidic than water (1, 2, 3). A food's pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.
What is the pH of bleach?
pH 11-13Bleach: pH 11-13 Bleach is one of the most common cleaning supplies in households and commercial settings. This particular product has a pH between 11 and 13. Its high level of alkalinity is what makes it corrosive.
What has a pH of zero?
hydrochloric acidA solution of a strong acid, such as hydrochloric acid, at concentration 1 mol dm−3 has a pH of 0.
How to measure pH?
To measure pH, a piece of pH test paper or an indicator stick is dipped into the liquid. The color of the dipped paper/stick is then matched to a color key that comes with the container of pH test paper or indicator sticks. Each color on the key represents a different pH.
What is the equation for pH?
To be more precise, pH is the negative logarithm of the hydrogen ion concentration: pH = −log [H+] The square brackets around the H + automatically mean "concentration" to a chemist. What the equation means is just what we said before: for each 1-unit change in pH, the hydrogen ion concentration changes ten-fold.
How many times more hydrogen ions are in a strongly acidic solution than a strongly acidic solution?
Here is why: a strongly acidic solution can have one hundred million million, or one hundred trillion (100,000,000,000,000) times more hydrogen ions than a strongly basic solution! The flip side, of course, is that a strongly basic solution can have 100,000,000,000,000 times more hydroxide ions than a strongly acidic solution.
Why is a solution alkaline?
Because the base "soaks up" hydrogen ions, the result is a solution with more hydroxide ions than hydrogen ions. This kind of solution is alkaline. Acidity and alkalinity are measured with a logarithmic scale called pH. Here is why: a strongly acidic solution can have one hundred million million, or one hundred trillion (100,000,000,000,000) ...
What scale do you use to measure hydrogen ion concentration?
In order to deal with these large numbers more easily, scientists use a logarithmic scale, the pH scale. Each one-unit change in the pH scale corresponds to a ten-fold change in hydrogen ion concentration.
How to measure pH in a solution?
pH test paper and indicator sticks are pieces of paper or stiffer sticks that contain pH indicators (chemicals that change color depending on how acidic or basic a solution is). To measure pH, a piece of pH test paper or an indicator stick is dipped into the liquid. The color of the dipped paper/stick is then matched to a color key that comes with the container of pH test paper or indicator sticks. Each color on the key represents a different pH. An example of a used pH indicator stick and the corresponding color key is shown below in Figure 1. pH meters are electronic devices that used to measure pH. They consist of a probe that is dipped in a solution, and a digital readout. pH meters are even more precise than pH test paper or indicator sticks. Table 2 below discusses what types of pH measuring devices are best for different science project applications, and offers a quick link to purchasing different pH test papers and indicator sticks.
What is the chemical symbol for hydrogen?
It all has to do with hydrogen ions (abbreviated with the chemical symbol H + ). In water (H 2 O), a small number of the molecules dissociate (split up). Some of the water molecules lose a hydrogen and become hydroxide ions (OH − ). The "lost" hydrogen ions join up with water molecules to form hydronium ions (H 3 O + ).
What is the pH scale?
The pH scale measures how acidic an object is. Objects that are not very acidic are called basic. The scale has values ranging from zero (the most acidic) to 14 (the most basic). As you can see from the pH scale above, pure water has a pH value of 7.
How to measure pH in lab?
There are many high-tech devices that are used to measure pH in laboratories. One easy way that you can measure pH is with a strip of litmus paper. When you touch a strip of litmus paper to something, the paper changes color depending on whether the substance is acidic or basic.
Is rain acidic or basic?
This value is considered neutral—neither acidic or basic. Normal, clean rain has a pH value of between 5.0 and 5.5, which is slightly acidic. However, when rain combines with sulfur dioxide or nitrogen oxides—produced from power plants and automobiles—the rain becomes much more acidic. Typical acid rain has a pH value of 4.0.
What is the pH of a chemical?
Key Takeaways: pH of Common Chemicals 1 pH is a measure of how acidic or basic an aqueous solution is. pH usually ranges from 0 (acidic) to 14 (basic). A pH value around 7 is considered neutral. 2 pH is measured using pH paper or a pH meter. 3 Most fruits, vegetables, and body fluids are acidic. While pure water is neutral, natural water may be either acidic or basic. Cleaners tend to be basic.
What is the pH of a substance?
Substances with a pH greater than 7 up to 14 are considered bases. Chemicals with a pH lower than 7 down to 0 are considered acids. The closer the pH is to 0 or 14, the greater its acidity or basicity, respectively. Here's a list of the approximate pH of some common chemicals.
What is the pH of soil?
Soil pH ranges from 3 to 10. Most plants prefer a pH between 5.5 and 7.5. Stomach acid contains hydrochloric acid and other substances and has a pH value of 1.2. While pure water free of undissolved gases is neutral, not much else is. However, buffer solutions may be prepared to maintain a pH near 7. Dissolving table salt (sodium chloride) in water does not change its pH.
What is the pH of an aqueous solution?
pH is a measure of how acidic or basic an aqueous solution is. pH usually ranges from 0 (acidic) to 14 (basic). A pH value around 7 is considered neutral.
Why is distilled water acidic?
Distilled water tends to be slightly acidic because of dissolved carbon dioxide and other gases. Pure water is nearly neutral, but rain water tends to be slightly acidic. Natural water rich in minerals tends to be alkaline or basic.
What is a pH test kit?
pH test kits are available to test liquids. Usually these are designed for a particular application, like aquaria or swimming pools. pH test kits are fairly accurate, but may be affected by other chemicals in a sample.
Is milk acidic or neutral?
Milk is often considered to be neutral, since it's only slightly acidic. Milk becomes more acidic over time. The pH of urine and saliva is slightly acidic, around a pH of 6. Human skin, hair, and nails tends to have a pH around 5. 0 - Hydrochloric Acid (HCl) 1.0 - Battery Acid (H 2 SO 4 sulfuric acid) and stomach acid.
What Is An Acid Or A Base?
What Is Ph?
- Acidity and alkalinity are measured with a logarithmic scale called pH. Here is why: a strongly acidic solution can have one hundred million million, or one hundred trillion (100,000,000,000,000) times more hydrogen ions than a strongly basic solution! The flip side, of course, is that a strongly basic solution can have 100,000,000,000,000 times more hydroxide ions than a strongly acidic s…
How Do You Measure Ph?
- The pH of a liquid or solution is often an important piece of information in science. Measuring pH can be done simply and quickly using pH test paper, pH indicator sticks, or a pH meter. pH test paper and indicator sticks are pieces of paper or stiffer sticks that contain pH indicators(chemicals that change color depending on how acidic or basic a solution is). To mea…
Bibliography
- For more information about acids, bases, and the pH scale, try this reference: 1. Khan Academy. (2009, September 7). Arrhenius acids and bases. Retrieved July 15, 2021.