List of Chemical Elements
| Atomic Number | Atomic Mass | Chemical Element Name | Symbol | Discovery (Year) |
| 115 | 290 | Moscovium | Mc | 2010 |
| 116 | 293 | Livermorium | Lv | 2000 |
| 117 | 294 | Tennessine | Ts | 2010 |
| 118 | 294 | Oganesson | Og | 2006 |
Where is the atomic size on a periodic table?
Atomic Size & Atomic Radius
- Table of Contents. What is Atomic Radius? What is Atomic Radius? An atomic radius is half the distance between adjacent atoms of the same element in a molecule.
- Recommended Videos
- Trends in the Periodic Table. Moving down a group or across a column or row in the modern periodic table, we can observe a lot of trends in the properties ...
What is 23 on the periodic table?
Vanadium is a silvery colored metal. It is used as an additive in steel to strengthen and protect against corrosion. Named after Scandinavian goddess, Vanadis. Vanadium was first discovered by del Rio in 1801.
What is the atomic number of the periodic table?
The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. It is the charge number of the nucleus since neutrons carry no net electrical charge. The atomic number determines the identity of an element and many of its chemical properties. The modern periodic table is ordered by increasing atomic number.
What is the atomic structure of the periodic table?
The periodic table is a table containing all elements arranged in ascending order from the one with lowest atomic number to the one with highest atomic number. There are there are 8 vertical groups (Columns) in the periodic table.

What is an atom and example?
The defining particle that identifies an atom is the number of protons it contains. So, a particle that lacks protons is not an atom. However, even one lone proton is an atom (of hydrogen). Examples of atoms include single particles of the elements of the periodic table, such as sodium, uranium, argon, and chlorine.
Where is the atom on the periodic table?
The number of protons in an atom is equal to the atomic number of the element. For example, let's use oxygen. According to the periodic table, oxygen has the atomic number eight. The atomic number is located above the element's symbol.
Is an element an atom?
An atom is the part of an element. A particular element is composed of only one type of atom. Atoms are further composed of subatomic particles called electrons, protons and neutrons. Elements can combine with each other to form molecules via chemical reaction.
How many atoms are in the periodic table?
The periodic table, also called the periodic table of elements, is an organized arrangement of the 118 known chemical elements.
What is the periodic table?
The periodic table is a tabular array of the chemical elements organized by atomic number, from the element with the lowest atomic number, hydrogen...
What do periodic table groups have in common?
The groups of the periodic table are displayed as vertical columns numbered from 1 to 18. The elements in a group have very similar chemical proper...
Where does the periodic table come from?
The arrangement of the elements in the periodic table comes from the electronic configuration of the elements. Because of the Pauli exclusion princ...
Why does the periodic table split?
The periodic table has two rows at the bottom that are usually split out from the main body of the table. These rows contain elements in the lantha...
What is the periodic table?
periodic table, in full periodic table of the elements, in chemistry, the organized array of all the chemical elements in order of increasing atomic number —i.e., the total number of protons in the atomic nucleus. When the chemical elements are thus arranged, there is a recurring pattern called the “periodic law” in their properties, ...
What is the atomic number of an element?
The atomic number of an element is the number of protons in the nucleus of an atom of that element . Hydrogen has 1 proton, and oganesson has ...
How many protons does hydrogen have?
The atomic number of an element is the number of protons in the nucleus of an atom of that element. Hydrogen has 1 proton, and oganesson has 118.
Who proposed the atomic weights of the elements?
Attempts were later made to show that the atomic weights of the elements could be expressed by an arithmetic function, and in 1862 A.-E.-B. de Chancourtois proposed a classification of the elements based on the new values of atomic weights given by Stanislao Cannizzaro’s system of 1858.
When the chemical elements are thus arranged, there is a recurring pattern called the periodic law?
When the chemical elements are thus arranged, there is a recurring pattern called the “periodic law” in their properties, in which elements in the same column (group) have similar properties. The initial discovery, which was made by Dmitry I. Mendeleyev in the mid-19th century, has been of inestimable value in the development of chemistry.
What are the elements that are related to the first seven?
Newlands proposed classifying the elements in the order of increasing atomic weights, the elements being assigned ordinal numbers from unity upward and divided into seven groups having properties closely related to the first seven of the elements then known: hydrogen, lithium, beryllium, boron, carbon, nitrogen, and oxygen . This relationship was termed the law of octaves, by analogy with the seven intervals of the musical scale.
What is the structure of an atom?
Atomic structure is the positively charged nucleus and the negatively charged electrons circling around it, within an atom. The atom itself is the fundamental building block of all matter in the universe. From cars to cats and airplanes, everything is made up of atoms. The atom is made up of three core components which are called subatomic ...
Which part of an atom contains the protons and neutrons?
In the centre is the nucleus. This is an area of the atom that contains the protons (positive subatomic particles) and the neutrons (neutral subatomic particles). Overall the nucleus has a positive charge due to the presence of the proton, it also contains most of the mass of the atom as the proton and neutron both have a mass of one.
What holds protons and neutrons together?
Whilst inside the nucleus the proton and neutron are held together by a strong nuclear force, this is like a very strong glue that keeps protons and neutrons stuck together. The force between protons and electrons is the electromagnetic attraction, as they have opposite charges that attract each other.
How does adding more protons to the nucleus change the identity of the element?
As you add more protons to the nucleus this changes the identity of the element meaning each atom has a different number of protons. When the number of neutrons is changed this can change the mass too but it does not change the identity.
How many protons does lithium have?
For example Lithium contains 3 protons and has 3 electrons orbiting the nucleus in shells. This means the nucleus has a 3+ charge and the electrons contribute a -3 charge. The 3+ charge of the nucleus and the -3 charge of the 3 electrons cancel out to give an overall charge of 0 meaning the element is neutral. . .
How many particles are in an atom?
The atom contains 3 particles that give it identity. The nucleus contains two particles, one is the proton. The proton is a positively charged particle with a mass of one. The other particle is the neutron.
What are electrons in the solar system?
These are electrons, negatively charged particles that orbit the nucleus. As electrons are negatively charged there is an attraction to the nucleus but instead of combining they orbit the nucleus, much like the planets of the solar system orbit the sun.
Why Arrange Elements in a Table?
Seeing chemical elements arranged in the modern periodic table is as familiar as seeing a map of the world, but it was not always so obvious.
Does the Modern Periodic Table Change? If So, How and Who Does That?
The periodic table as we know it today is managed by the International Union of Pure and Applied Chemistry, or IUPAC (eye-you-pack).
What is an atom?
Anne Marie Helmenstine, Ph.D. Updated May 06, 2019. An atom is the basic unit of an element. An atom is a form of matter which may not be further broken down using any chemical means. A typical atom consists of protons, neutrons, and electrons.
What are some examples of substances that are not atoms?
Examples of chemical species that are not typically considered atoms includes particles that are components of atoms: protons, neutrons, and electrons. Molecules and compounds consist of atoms but are not themselves atoms. Examples of molecules and compounds include salt ...
Is hydrogen an atom?
Would you consider a single unit of hydrogen to be an example of an atom? Keep in mind, most hydrogen "atoms" do not have a proton, neutron, and electron. Given that the number of protons determines the identity of an element, many scientists consider a single proton to be an atom of the element hydrogen .
What is the atomic number of an atom?
Atomic number. The single most important characteristic of an atom is its atomic number (usually denoted by the letter Z ), which is defined as the number of units of positive charge (protons) in the nucleus. For example, if an atom has a Z of 6, it is carbon, while a Z of 92 corresponds to uranium. A neutral atom has an equal number of protons and ...
How many atoms are in a row?
All atoms are roughly the same size, whether they have 3 or 90 electrons. Approximately 50 million atoms of solid matter lined up in a row would measure 1 cm (0.4 inch). A convenient unit of length for measuring atomic sizes is the angstrom (Å), defined as 10 −10 metre. The radius of an atom measures 1–2 Å.
What is the difference between a neutral and a neutral atom?
For example, if an atom has a Z of 6, it is carbon, while a Z of 92 corresponds to uranium. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance.
What is matter made of?
Most matter consists of an agglomeration of molecules, which can be separated relatively easily. Molecules, in turn, are composed of atoms joined by chemical bonds that are more difficult to break. Each individual atom consists of smaller particles—namely, electrons and nuclei. These particles are electrically charged, and the electric forces on the charge are responsible for holding the atom together. Attempts to separate these smaller constituent particles require ever-increasing amounts of energy and result in the creation of new subatomic particles, many of which are charged.
How many protons are in a nucleus?
The fact that nuclei can have anywhere from 1 to nearly 300 protons and neutrons accounts for their wide variation in mass. The lightest nucleus, that of hydrogen, is 1,836 times more massive than an electron, while heavy nuclei are nearly 500,000 times more massive.
Which part of an atom contains the most mass?
The nucleus is the positively charged centre of an atom and contains most of its mass. It is composed of protons, which have a positive charge, and neutrons, which have no charge. Protons, neutrons, and the electrons surrounding them are long-lived particles present in all ordinary, naturally occurring atoms.
Why do physicists use complementary pictures of the atom?
Because of the nature of quantum mechanics, no single image has been entirely satisfactory at visualizing the atom’s various characteristics , which thus forces physicists to use complementary pictures of the atom to explain different properties .
What is the periodic table?
The periodic table, also known as the periodic table of elements, is a tabular display of the chemical elements, which are arranged by atomic number, electron configuration, and recurring chemical properties. The structure of the table shows periodic trends. The seven rows of the table, called periods, generally have metals on ...
What are the columns of periodic table called?
The seven rows of the table, called periods, generally have metals on the left and nonmetals on the right. The columns, called groups , contain elements with similar chemical behaviours.
What is the energy of ionization?
The first ionization energy is the energy it takes to remove one electron from an atom, the second ionization energy is the energy it takes to remove a second electron from the atom, and so on. For a given atom, successive ionization energies increase with the degree of ionization. For magnesium as an example, the first ionization energy is 738 kJ/mol and the second is 1450 kJ/mol. Electrons in the closer orbitals experience greater forces of electrostatic attraction; thus, their removal requires increasingly more energy. Ionization energy becomes greater up and to the right of the periodic table.
What is the atomic number plotted against?
Atomic number plotted against atomic radius, excluding the noble gases. Atomic radii vary in a predictable and explainable manner across the periodic table. For instance, the radii generally decrease along each period of the table, from the alkali metals to the noble gases; and increase down each group.
What is the electron configuration of a neutral atom?
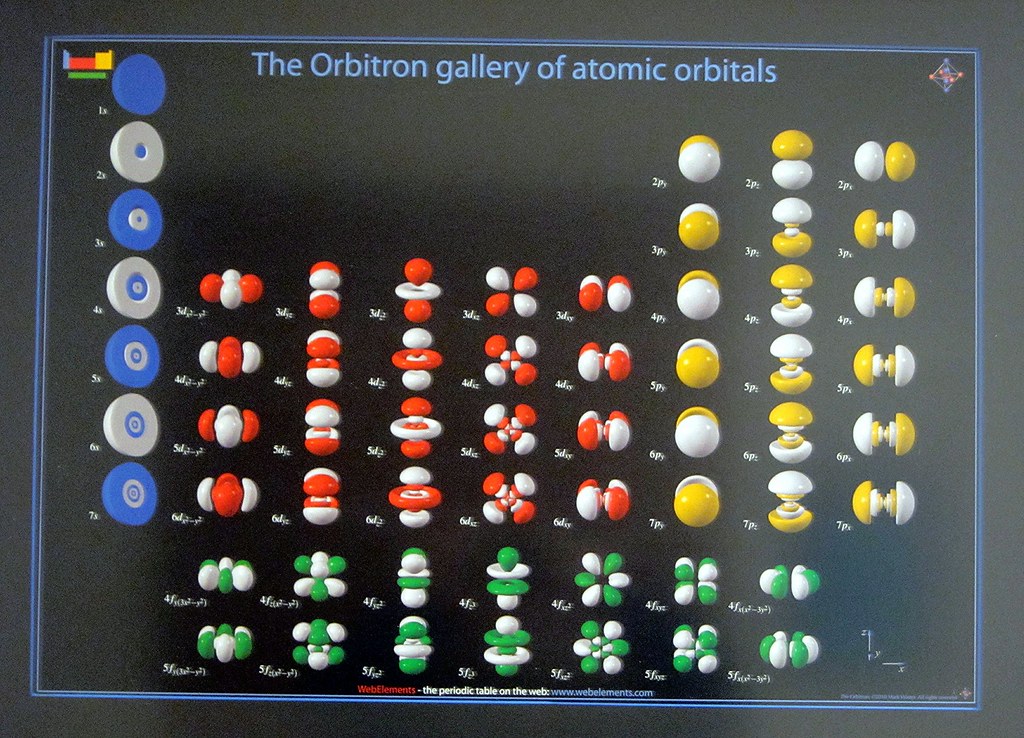
The electron configuration or organisation of electrons orbiting neutral atoms shows a recurring pattern or periodicity. The electrons occupy a series of electron shells (numbered 1, 2, and so on). Each shell consists of one or more subshells (named s, p, d, f and g). As atomic number increases, electrons progressively fill these shells and subshells more or less according to the Madelung rule or energy ordering rule, as shown in the diagram. The electron configuration for neon, for example, is 1s 2 2s 2 2p 6. With an atomic number of ten, neon has two electrons in the first shell, and eight electrons in the second shell; there are two electrons in the s subshell and six in the p subshell. In periodic table terms, the first time an electron occupies a new shell corresponds to the start of each new period, these positions being occupied by hydrogen and the alkali metals.
What are metals and nonmetals?
In chronological order, this section discusses metals and nonmetals (and metalloids); categories of elements; groups and periods; and periodic table blocks. While the recognition of metals as solid, fusible and generally malleable substances dates from antiquity, Antoine Lavoisier may have the first to formally distinguish between metals and nonmetals ('non-métalliques') in 1789 with the publication of his 'revolutionary' Elementary Treatise on Chemistry. In 1811, Berzelius referred to nonmetallic elements as metalloids, in reference to their ability to form oxyanions. In 1825, in a revised German edition of his Textbook of Chemistry, he subdivided the metalloids into three classes. These were: constantly gaseous 'gazolyta' (hydrogen, nitrogen, oxygen); real metalloids (sulfur, phosphorus, carbon, boron, silicon); and salt-forming 'halogenia' (fluorine, chlorine, bromine, iodine). Only recently, since the mid-20th century, has the term metalloid been widely used to refer to elements with intermediate or borderline properties between metals and nonmetals. Mendeleev published his periodic table in 1869, along with references to groups of families of elements, and rows or periods of his periodic table. At the same time, Hinrichs wrote that simple lines could be drawn on a periodic table in order to delimit properties of interest, such as elements having metallic lustre (in contrast to those not having such lustre). Charles Janet, in 1928, appears to have been the first to refer to the periodic table's blocks.
How many electrons are in neon?
The electron configuration for neon, for example, is 1s 2 2s 2 2p 6. With an atomic number of ten, neon has two electrons in the first shell, and eight electrons in the second shell; there are two electrons in the s subshell and six in the p subshell. In periodic table terms, the first time an electron occupies a new shell corresponds to ...
What is the periodic table?
The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. In the periodic table, the vertical columns are called ‘groups’ and the horizontal rows are called ‘periods’. The modern periodic table is based on the modern periodic law put forward by the English physicist Henry Moseley, ...
What is the atomic number of an element?
The atomic number of an element is equal to the total number of protons in the nucleus of the atoms of that element. The atomic number can provide insight into the electronic configuration of the element. For example, carbon has an electron configuration of [He] 2s 2 2p 2, since its atomic number is 6.
What is the number of protons in the nucleus called?
The number of protons in the nucleus is called the atomic number. The atomic number of each element is unique.
Why is the atomic number of each element unique?
While the atomic number always stays the same some elements have atoms with different atomic mass numbers. This is because some elements have a different number of neutrons in the nucleus.
How can periodic trends be observed?
Periodic trends in the properties of the elements can be observed down the groups and across the periods of the modern periodic table. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus.
Why is the atomic number important?
This number is very important, because it is unique to a given element’s atoms. An element’s atoms all have the same number of protons and each element has a different number of protons in its atoms. Test your knowledge on periodic table elements.
Who created the modern periodic table?
The modern periodic table is based on the modern periodic law put forward by the English physicist Henry Moseley, which states that “the properties of elements are periodic functions of their atomic numbers”.
Periods
Periods are the rows of the periodic table that from left to right increase in order of mass. As you go along periods the number of protons and neutrons increases and the number of electron shells remains the same.
Groups
Groups are the columns of the periodic table with elements that are grouped together with similar properties.