How are trimeric G proteins activated?
Trimeric G protein signaling is a fundamental mechanism of cellular communication in eukaryotes. The core of this mechanism consists of activation of G proteins by the guanine-nucleotide exchange factor (GEF) activity of G protein coupled receptors.
What is the active form of a trimeric G-protein?
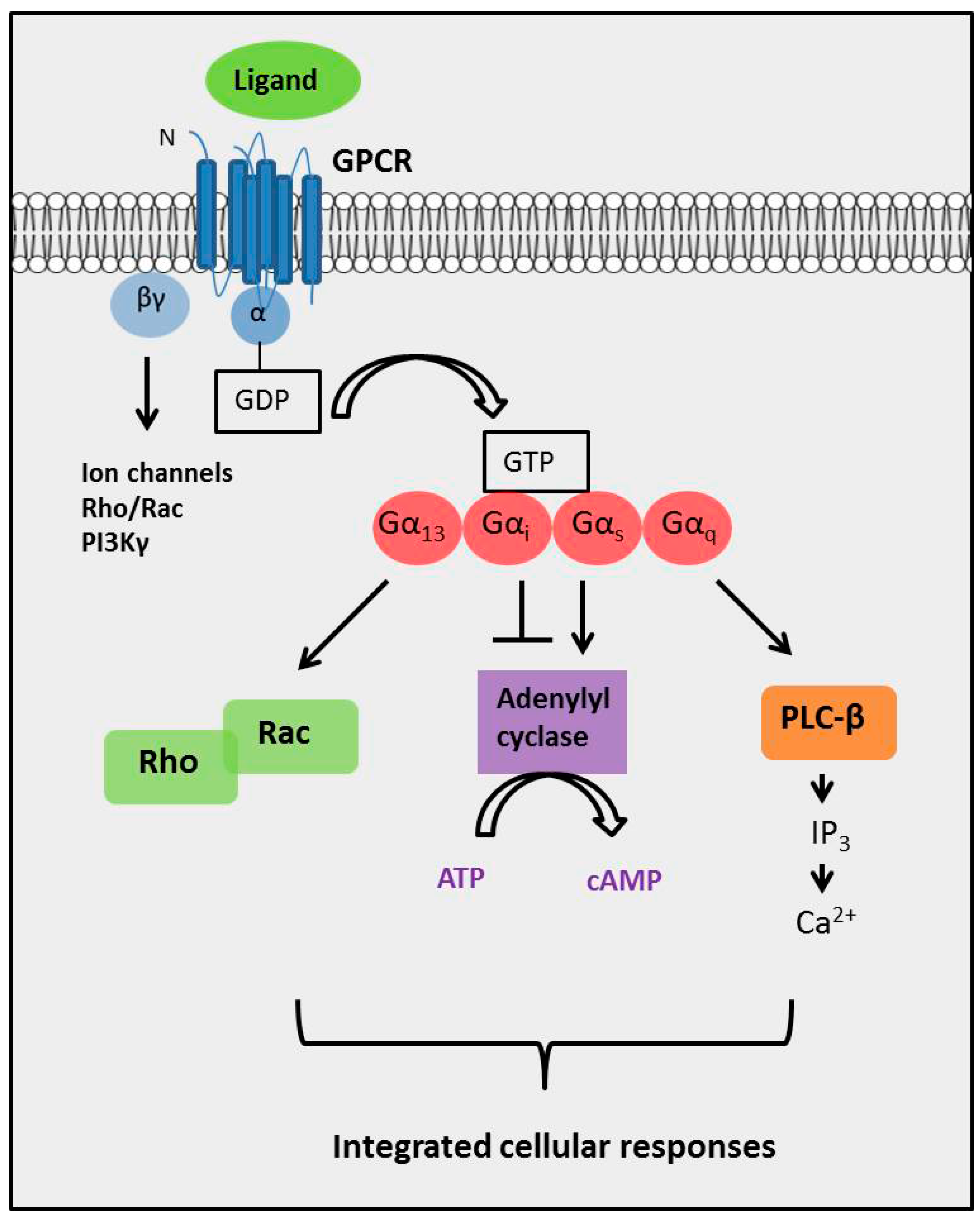
G-proteins are a form trimeric proteins, meaning they have 3 subunits, Alpha, Beta and Gamma. The trimeric protein is activated by the displacement of GDP with GTP through a conformation change in the receptor to which the G-protein is coupled, which in turn causes the release of the alpha subunit (Gsα).
What is the function of G-protein?
The main physiological functions of G-proteins are to relay the signals from GPCRs which function as GEFs for G-proteins. Binding with exogenous or endogenous agonists induces GPCRs into an active conformational state which, in turn, influences intracellular binding of G-proteins or arrestin proteins [23, 24].
What is a monomeric G-protein?
A monomeric G-protein (also known as small G protein or small GTPases, is the umbrella term for a family of signal transducing proteins. These comprise of: Rab, Arf, Ras, Ran and Rho, which are also vital in the growth of cells, as well as cell transport, motility, cytokinesis and cell differentiation.
Why is G proteins trimeric?
G proteins are attached to the cytosolic face of the plasma membrane, where they serve as relay proteins between the receptors and their target signalling proteins. Trimeric G proteins interact with 7TM receptors and are all heterotrimeric, having structurally different α, β and γ subunits.
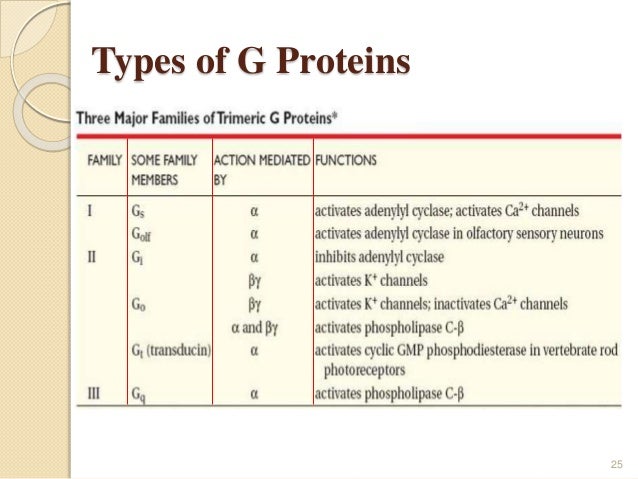
What are the types of G proteins?
Structure of G-Proteins The G-protein is heterotrimeric and is made up of three different subunits: alpha (α), beta (β) and gamma (γ). In its inactive state, GDP is bound to the α-subunit of the G-protein.
What is the 3 types of G protein?
G protein coupled receptors (GPCRs) are one of the major classes of cell surface receptors and are associated with a group of G proteins consisting of three subunits termed alpha, beta, and gamma.
Where is G protein found?
the cell membraneG protein-coupled receptors (GPCRs) are seven-transmembrane proteins that are located in the cell membrane, with their N- and C-termini located on the outer and inner surfaces, respectively. GPCRs mediate various cellular responses from the extracellular environment.
Why G protein is so called?
G proteins are so-called because they bind the guanine nucleotides GDP and GTP. They are heterotrimers (i.e., made of three different subunits) associated with the inner surface of the plasma membrane and transmembrane receptors of hormones, etc. These are called G protein-coupled receptors (GPCRs).
What is the difference between monomeric and heterotrimeric G proteins?
The key difference between heteromeric G protein and monomeric G protein is that heteromeric G protein is a large membrane-associated G protein made up of alpha (α), beta (β), and gamma (γ) subunits, while monomeric G protein is a small membrane-associated G protein made up of only an alpha subunit.
How are monomeric G proteins activated?
Activation of G proteins is enabled by accessory proteins which catalyse guanine nucleotide exchange—the sequential release of GDP and binding of GTP. For monomeric G proteins these are known as guanine nucleotide exchange factors (GEFs).
What is the role of G-proteins in the cell?
Within such cascades, G-proteinsserve a pivotal function as the molecular transducing elements that couple membrane receptors to their molecular effectors within the cell . The diversity of G-proteins and their downstream targets leads to many types of physiological responses. By directly regulating the gating of ion channels, G-proteins can influence the membrane potential of target cells.
What are the effects of GTP-linked receptors?
For both these receptortypes, the coupling between receptor activationand their subsequent effects are the GTP-binding proteins. There are two general classes of GTP-binding protein (Figure 8.5). Heterotrimeric G-proteinsare composed of three distinct subunits (α, β, and γ). There are many different α, β, and γ subunits, allowing a bewildering number of G-protein permutations. Regardless of the specific composition of the heterotrimeric G-protein, its α subunit binds to guanine nucleotides, either GTP or GDP. Binding of GDP allows the α subunit to bind to the β and γ subunits to form an inactive trimer. Binding of an extracellular signal to a G-protein-coupled receptor allows the G-protein to bind to the receptor and causes GDP to be replaced with GTP (Figure 8.5A). When GTP is bound to the G-protein, the α subunit dissociates from the βγ complex and activates the G-protein. Following activation, both the GTP-bound α subunit and the free βγ complex can bind to downstream effector molecules and mediate a variety of responses in the target cell.
Why are heterotrimeric G proteins activated?
They are activated in response to a conformational change in the GPCR, exchanging GDP for GTP, and dissociating in order to activate other proteins in a particular signal transduction pathway. The specific mechanisms, however, differ between protein types.
What are the subunits of G proteins?
Phosducin - transducin beta-gamma complex. Beta and gamma subunits of G-protein are shown by blue and red, respectively. Guanosine diphosphate. Guanosine triphosphate. G proteins, also known as guanine nucleotide-binding proteins, are a family of proteins that act as molecular switches inside cells, and are involved in transmitting signals ...
What are the two classes of G proteins?
There are two classes of G proteins. The first function as monomeric small GTPases (small G-proteins), while the second function as heterotrimeric G protein complexes. The latter class of complexes is made up of alpha (α), beta (β) and gamma (γ) subunits. In addition, the beta and gamma subunits can form a stable dimeric complex referred to as ...
How does GTP hydrolyze GDP?
The G α subunit will eventually hydrolyze the attached GTP to GDP by its inherent enzymatic activity, allowing it to re-associate with G βγ and starting a new cycle. A group of proteins called Regulator of G protein signalling (RGSs), act as GTPase-activating proteins (GAPs), are specific for G α subunits. These proteins accelerate the hydrolysis of GTP to GDP, thus terminating the transduced signal. In some cases, the effector itself may possess intrinsic GAP activity, which then can help deactivate the pathway. This is true in the case of phospholipase C -beta, which possesses GAP activity within its C-terminal region. This is an alternate form of regulation for the G α subunit. Such G α GAPs do not have catalytic residues (specific amino acid sequences) to activate the G α protein. They work instead by lowering the required activation energy for the reaction to take place.
How many GPCRs are there in the human genome?
The human genome encodes roughly 800 G protein-coupled receptors, which detect photons of light, hormones, growth factors, drugs, and other endogenous ligands. Approximately 150 of the GPCRs found in the human genome still have unknown functions.
How many G proteins are there in eukaryotes?
All eukaryotes use G proteins for signaling and have evolved a large diversity of G proteins. For instance, humans encode 18 different G α proteins, 5 G β proteins, and 12 G γ proteins.
What is the role of G protein in cell function?
The G protein activates a cascade of further signaling events that finally results in a change in cell function. G protein-coupled receptor and G proteins working together transmit signals from many hormones, neurotransmitters, and other signaling factors.
What are the G proteins?
G proteins are membrane-bound heterotrimeric GTPases, consisting of an α, β, and γ subunit, and function as the main signal transducers of the cell.
What is the role of the G protein in a cell?
These proteins are involved during signal transduction and signal amplifications through their interaction with protein coupled receptors , thus, modulating the events of the downstream effectors (Jones and Assmann, 2004 ).
What is the GPR motif?
The G-protein regulatory (GPR) motif serves as a docking site for Gαi–GDP free of Gβγ. The GPR–Gα complex may function at the cell cortex and/or at intracellular sites. GPR proteins include the Group II Activators of G-protein signaling identified in a functional screen for receptor-independent activators of G-protein signaling (GPSM1-3, RGS12) each of which contain 1–4 GPR motifs. GPR motifs are also found in PCP2/L7 (GPSM4), Rap1-Gap1 Transcript Variant 1, and RGS14. While the biochemistry of the interaction of GPR proteins with purified Gα is generally understood, the dynamics of this signaling complex and its regulation within the cell remains undefined. Major questions in the field revolve around the factors that regulate the subcellular location of GPR proteins and their interaction with Gαi and other binding partners in the cell. As an initial approach to this question, we established a platform to monitor the GPR–Gαi complex in intact cells using bioluminescence resonance energy transfer.
Why are G proteins inactive?
Figure 9.4. (A) G proteins are held in an inactive state because of very high affinity binding of GDP to their α subunits. When activated by agonist, membrane-bound seven helical receptors ( Fig. 9.4 right, glowing magenta) interact with heterotrimeric G proteins ( α, amber; β, teal; γ, burgundy) and stimulate dissociation of GDP. This permits GTP to bind to and activate α, which then dissociates from the high-affinity dimer of β and γ subunits. (B) Both activated (GTP-bound) α (lime) and βγ are capable of interacting with downstream effectors. Figure 9.4 shows the interaction of GTP- αs with adenylate cyclase (catalytic domains are mustard and ash). Adenylate cyclase then catalyzes the synthesis of the second messenger cyclic AMP (cAMP) from ATP. (C) Signaling is terminated when α hydrolyzes its bound GTP to GDP. In some signaling systems, GTP hydrolysis is stimulated by GTPase-activating proteins or GAPs (cranberry) that bind to α and stablize the transition state for GTP hydrolysis. (D) Hydrolysis of GTP permits GDP- α to dissociate from its effector and associate again with βγ. The heterotrimeric G protein is then ready for another signaling cycle if an activated receptor is present. This figure is based on the original work of Mark Wall and John Tesmer.
What is a GPER?
GPER is a seven transmembrane-domain G protein-coupled receptor. GPER was first identified and cloned by Carmechi et al. in 1997. 81 Using a differential cDNA library screening technique, the investigators identified GPER as a differentially expressed gene in a screen of genes from an ER-positive breast cancer cell line (MCF-7) versus an ER-negative breast cancer cell line (MDA-MB-361).
Where is GPER expressed?
Interestingly, sexual dimorphisms for GPER expression and/or function have been described in the brain 110 and in the pancreatic islets, where it is expressed at a much higher level in women than in men. 95 Accordingly, a role for GPER in the regulation of obesity-associated metabolic functions has been proposed.
What are G-protein coupled receptors?
G-protein coupled receptors that contain a very large amino terminal domain are made with a cleaved signal sequence. A similar situation exists with all the subunits of the acetylcholine receptor which each contain large extracellular N-glycosylated domains. Transport proteins such as the plasma membrane Na+ /H + and Na + /Ca 2 + exchangers are made with cleaved signal sequences, however these signal sequences are not required for the functional expression of these membrane proteins. The presence of an amino-terminal signal sequence likely promotes the targeting and insertion of these proteins into the ER, but other TM segments within the protein can also act as signal sequences.
What is trimeric G protein?
trimeric - has receptor, activated by the receptor ligand-binding , has three subunits that dissociate when activated, always bound to the membrane. Put the following steps of trimeric G-protein activation in order. Note: (GPCR = G-protein coupled receptor) A. G-protein dissociates into alpha and beta-gamma subunits.
What is the G-protein pathway?
1. signal molecule activates G-protein coupled receptor, which activates trimeric G-proteins and causes dissociation of the alpha subunit. 2. the alpha subunit activates adenylyl cyclase, which makes cAMP from ATP.
What is the GPCR?
Note: (GPCR = G-protein coupled receptor) True or False: trimeric G-proteins are inactivated when phosphorylated by GRK, which triggers arrestin and clathrins to bind. False: GRK, arrestin and clathrin is how the G-protein coupled RECEPTOR is inactivated.