
What are 4 examples of reversible reactions?
What are 4 examples of reversible reactions?
- Ammonium chloride is a white strong. It breaks down when heated, forming ammonia and hydrogen chloride.
- Ammonium chloride ⇌ ammonia + hydrogen chloride.
- The image ⇌ has two half arrowheads, one pointing in every course. It’s utilized in equations that mannequin reversible reactions:
What are some examples of reversible reactions?
- Frictionless movement
- Restrained compression or expansion
- Energy transfer as heat due to infinitesimal temperature nonuniformity
- Electric current flow through a zero resistance
- Restrained chemical reaction
- Mixing of two samples of the same substance at the same state.
What are some easy examples of chemical reactions?
Some examples of simple synthesis reactions include:
- hydrogen + oxygen ---> water
- iron + oxygen ---> rust
- potassium and chlorine gas ---> chloride
- lime + carbon dioxide ---> calcium carbonate (used to strengthen masonry)
- water + carbon dioxide + light ---> glucose and oxygen (photosynthesis)
- sodium + chloride ---> sodium chloride (table salt)
What are examples of irreversible chemical reactions?
- 2Na + H2O ——→ 2NaOH + H2
- NaOH + HCl ——→ NaCl + H2O
- CaCO3 + 2HNO3 ——→ Ca (NO3)2 + CO2+ H2O
- MnO2 + 4HCl → MnCl2 + Cl2 + 2H2O
- BaCl2 + H2SO4 ——→ BaSO4 + 2HCl
- CH4 + 2O2 ——→ CO2 + 2H2O (combustion)
What are the 4 examples of reversible reactions?
Examples of Reversible ReactionReaction between hydrogen (H2) and iodine (I2) to produce hydrogen iodide (HI). ... Nitrogen (N2) reacting with hydrogen (H2) to produce ammonia (NH3). ... Sulfur dioxide (SO2) reacts with oxygen (O2) to make sulfur trioxide (SO3)More items...
What is a reversible chemical reaction?
A reversible reaction is defined as a chemical reaction where the reactants and the products react together to give the reactants back.
Are there any reversible chemical reactions?
In principle, all chemical reactions are reversible reactions . This means that the products can be changed back into the original reactants .
What are some everyday examples of reversible reactions?
Examples of reversible changesMelting: Melting is when solid converts into a liquid after heating. Example of melting is turning of ice into water.Freezing: Freezing is when a liquid converts into a solid. Example of freezing is turning of water into ice.Boiling: Boiling is when a liquid converts into a gas.
Which one of the following is an example of reversible change?
Melting, boiling, evaporation, freezing, condensation, and disintegration are examples of reversible transformations. Melting wax, freezing ice, and boiling water that evaporates as steam and condenses back to water are a few examples. Hence option C is correct.
Why are some chemical reactions reversible?
Because at the level of atoms and molecules every collision and change can happen in both the directions. This is called "principle of microscopic reversibility". If a bond can be broken, the same bond can be formed from the fragments; If a torsion is possible, the opposite torsion is equally possible, and so on.
Could any chemical change be reversible if yes give examples?
In most circumstances, new material is not created when a reversible chemical reaction occurs. Examples of reversible chemical changes include spring elongation, paper folding, and so on.
Is boiling reversible?
This is a reversible change. Similarly, if you boil water, it evaporates and becomes water vapor. When you cool this vapor down, it turns back to water. A substance can return to its original state.
Is combustion a reversible reaction?
Combustion reactions are generally irreversible. Combustion occurs whenever a fuel burns. In this type of reaction, the fuel may combine with oxygen (in the air) and produces carbon dioxide and water vapor.
Is freezing reversible?
Freezing is a reversible change. For example you can freeze juice to make ice lollies. The lollies can be changed back into juice by heating.
What is reversible reaction and irreversible reaction with example?
Irreversible chemical reactions can occur in only one direction. The reactants can change to the products, but the products cannot change back to the reactants. Reversible chemical reactions can occur in both directions. The reactants can change to the products, and the products can also change back to the reactants.
What are reversible and irreversible reaction explain with examples?
Reversible reaction : A reaction which takes place in both forward and backward direction is called reversible reaction. Irreversible reaction: A reaction in which entire amount of reactant is changed into product and no reaction from product side occurs in called irreversible reaction.
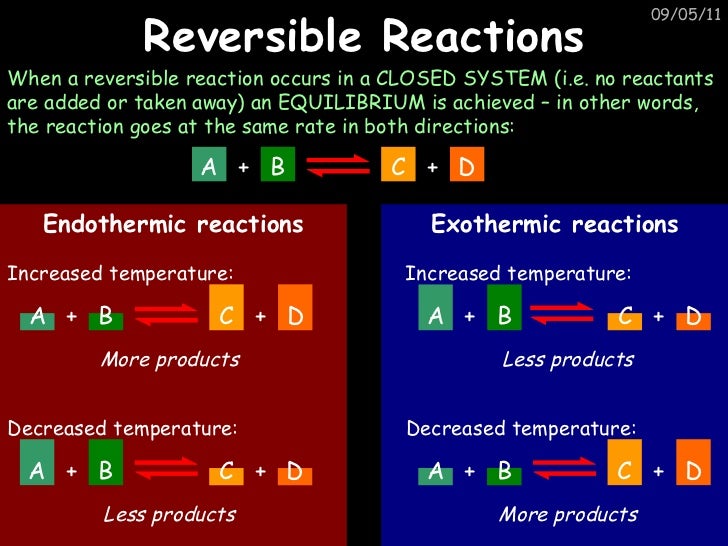
What is a reversible reaction?
Updated August 19, 2019. A reversible reaction is a chemical reaction where the reactants form products that, in turn, react together to give the reactants back. Reversible reactions will reach an equilibrium point where the concentrations of the reactants and products will no longer change.
When did Berthollet propose the idea of a reversible reaction?
At one time, scientists believed all chemical reactions were irreversible reactions. In 1803 , Berthollet proposed the idea of a reversible reaction after observing the formation of sodium carbonate crystals on the edge of a salt lake in Egypt.
Do two chemical reactions occur at the same time?
Two chemical reactions occur simultaneously: Reversible reactions do not necessarily occur at the same rate in both directions, but they do lead to an equilibrium condition. If dynamic equilibrium occurs, the product of one reaction is forming at the same rate as it is used up for the reverse reaction.
What is a Reversible Process?
Reactions that do not reach to the absolute completion are reversible.
Give one Example of Reversible Chemical Change
The reaction of ammonia and hydrogen chloride is one such example of reversible chemical change.
What should be the equilibrium position for a reversible reaction?
The equilibrium of a reversible chemical reaction is affected by several factors.
