Deamination converts nitrogen from the amino acid into ammonia, which is converted by the liver into urea in the urea cycle. This example is from Wikipedia and may be reused under a CC BY-SA license. The most common mutation is the deamination of cytosine to uracil.
What is deamination of amino acids?
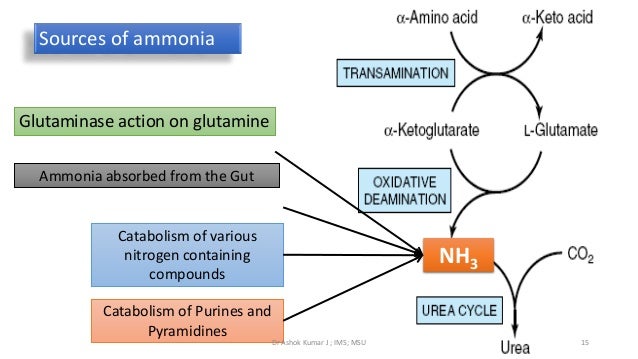
Deamination is the removal of an amino group from a molecule. Enzymes that catalyse this reaction are called deaminases . In the human body, deamination takes place primarily in the liver, however it can also occur in the kidney. In situations of excess protein intake, deamination is used to break down amino acids for energy.
What is the deamination process of ammonia?
Ammonia is toxic to the human system, and enzymes convert it to urea or uric acid by addition of carbon dioxide molecules (which is not considered a deamination process) in the urea cycle, which also takes place in the liver. Urea and uric acid can safely diffuse into the blood and then be excreted in urine. Deamination of cytosine to uracil.
What is the purpose of deamination?
Deamination. In situations of excess protein intake, deamination is used to break down amino acids for energy. The amino group is removed from the amino acid and converted to ammonia. The rest of the amino acid is made up of mostly carbon and hydrogen, and is recycled or oxidized for energy. Ammonia is toxic to the human system,...
What are the processes of nitrogen cycle?
Process of Nitrogen Cycle consists of the following steps – Nitrogen fixation, Nitrification, Assimilation, Ammonification and Denitrification. These processes take place in several stages and are explained below:

What is the deamination process?
Typically in humans, deamination occurs when an excess in protein is consumed, resulting in the removal of an amine group, which is then converted into ammonia and expelled via urination. This deamination process allows the body to convert excess amino acids into usable by-products.
Does deamination remove nitrogen?
Rather, almost all nitrogen is transported throughout the body as amino or amide groups of other molecules, particularly alanine and glutamine. In the liver, nitrogen destined for disposal is removed from amino acids in a two-step process referred to as “trans-deamination”.
What is deamination and detoxification?
Deamination is the removal of an amine group from a molecule with an amine group. Detoxification is the metabolism of a toxic compound to a non-toxic compound, or the excretion of a toxic compound.
What is deamination in excretion?
Deamination is the process by which amino acids are broken down. This process allows the system to convert excess amino acids into usable resources, such as hydrogen and carbon.
Is deamination an oxidation reaction?
Oxidative Deamination. Introduction: Deamination is also an oxidative reaction that occurs under aerobic conditions in all tissues but especially the liver.
How does deamination produce energy?
amino acid degradation …acids for energy production is deamination, the splitting off of ammonia from the amino-acid molecule. The remainder is oxidized to carbon dioxide and water, with the concomitant production of the energy-rich molecules of adenosine triphosphate (ATP; see metabolism).
What is transamination and deamination?
Definition. Transamination refers to the transfer of an amino group from one molecule to another, especially from an amino acid to a keto acid, while deamination refers to the removal of an amino group from an amino acid or other compounds.
Is deamination anabolic or catabolic?
Catabolic Processes. The main processes of catabolism include the citric acid cycle, glycolysis, oxidative deamination, the breakdown of muscle tissue and the breakdown of fat.
What is deamination in biology Igcse?
In deamination, the nitrogen-containing amino group is removed and converted into ammonia and then urea to be excreted.
What is deamination GCSE?
The liver is involved in the process of deamination. This is the removal of the nitrogen-containing part of amino acids, to form urea, followed by the release of energy from the remainder of the amino acid.
What happens to an amino acid during deamination?
Oxidative deamination provides a reaction in which the amino group [as the ammonium (NH4+) ion] is removed from a molecule, not simply transferred from one molecule to another. Most of the NH4+ ion is converted to urea and excreted from the body.
What is deamination in zoology?
Deamination is the process by which an amino group will be removed from the molecule. Enzymes that are involved in the catalysis of this reaction are called deaminases. In humans, the deamination will occur in the liver and kidney.
Why is nitrogen important for life?
Nitrogen constitutes many cellular components and is essential in many biological processes. For instance, the amino acids contain nitrogen and for...
Why do plants need nitrogen?
Plants need nitrogen as this element is an important component of chlorophyll. Consequently, chlorophyll is vital for the process of photosynthesis...
List the different steps that explain the Nitrogen Cycle process.
Nitrogen Fixation Assimilation Ammonification Nitrification Denitrification
What is Ammonification?
Ammonification occurs during the decomposition of organic matter, where ammonifying bacteria convert organic nitrogen into inorganic components lik...
What is Nitrification?
Nitrification is a process that converts ammonia into nitrate by bacteria. Initially, the ammonia is converted to nitrite (NO 2 − ) by the bacteria...
What is Denitrification?
Denitrification is the process of converting the nitrate back into molecular nitrogen by bacterias such as Pseudomonas, Thiobacillus, Bacillus sub...
What is the function of nitrifying bacteria?
Nitrifying bacteria are a small group of aerobic bacteria, which are mainly involved in the conversion of ammonia into nitrates.
Which part of the plant is involved in nitrogen fixation?
The process of nitrogen fixation is carried out naturally in the soil within nodules in the plant’s root systems.
What is the nitrogen cycle?
“Nitrogen Cycle is a biogeochemical process which transforms the inert nitrogen present in the atmosphere to a more usable form for living organisms.”. Furthermore, nitrogen is a key nutrient element for plants. However, the abundant nitrogen in the atmosphere cannot be used directly by plants or animals.
Which bacterial species are responsible for the final stage of the nitrogen cycle?
Denitrification is carried out by the denitrifying bacterial species- Clostridium and Pseudomonas, which will process nitrate to gain oxygen and gives out free nitrogen gas as a byproduct.
How is ammonia converted into nitrate?
In this process, the ammonia is converted into nitrate by the presence of bacteria in the soil. Nitrites are formed by the oxidation of Ammonia with the help of Nitrosomonas bacterium species. Later, the produced nitrites are converted into nitrates by Nitrobacter. This conversion is very important as ammonia gas is toxic for plants.
How does nitrogen fixation occur?
Nitrogen fixation can occur either by atmospheric fixation- which involves lightening or industrial fixation by manufacturing ammonia under high temperature and pressure condition. This can also be fixed through man-made processes, primarily industrial processes that create ammonia and nitrogen-rich fertilisers.
How is nitrogen made available to plants?
This nitrogen is made available to plants by symbiotic bacteria which can convert the inert nitrogen into a usable form – such as nitrites and nitrates. Nitrogen undergoes various types of transformation to maintain a balance in the ecosystem. Furthermore, this process extends to various biomes, with the marine nitrogen cycle being one ...
How is ammonia produced?
Ammonia is produced by the direct combination of nitrogen and hydrogen and later, it is converted into various fertilisers such as urea. Biological nitrogen fixation: We already know that nitrogen is not usable directly from the air for plants and animals.
What is nitrogen fixation?
Nitrogen fixation. It is the initial step of the nitrogen cycle. Here, Atmospheric nitrogen (N2) which is primarily available in an inert form, is converted into the usable form -ammonia (NH3). During the process of Nitrogen fixation, the inert form of nitrogen gas is deposited into soils from the atmosphere and surface waters, ...
What is the result of deamination of adenine?
Deamination of adenine results in the formation of hypoxanthine. Hypoxanthine, in a manner analogous to the imine tautomer of adenine, selectively base pairs with cytosine instead of thymine. This results in a post-replicative transition mutation, where the original A-T base pair transforms into a G-C base pair.
What is the purpose of deamination?
In situations of excess protein intake, deamination is used to break down amino acids for energy. The amino group is removed from the amino acid and converted to ammonia. The rest of the amino acid is made up of mostly carbon and hydrogen, and is recycled or oxidized for energy.
What is the result of cytosine methylation?
A known result of cytosine methylation is the increase of C-to-T transition mutations through the process of deamination. Cytosine deamination has been seen needed for many regulatory functions in the genome; many regulatory functions are related to the loss in CPG sites due to the presence of transposable elements. TEs have been proposed to accelerate the mechanism of enhancer creation by providing extra DNA that is compatible with the host transcription factors that eventually have an impact on C-to-T mutations.
What is the name of the reaction that deaminates cytosine?
Cytosine. Deamination of cytosine to uracil. Spontaneous deamination is the hydrolysis reaction of cytosine into uracil, releasing ammonia in the process. This can occur in vitro through the use of bisulfite, which deaminates cytosine, but not 5-methylcytosine.
Where does deamination take place?
Enzymes that catalyse this reaction are called deaminases . In the human body, deamination takes place primarily in the liver, however it can also occur in the kidney.
Is ammonia toxic to humans?
Ammonia is toxic to the human system , and enzymes convert it to urea or uric acid by addition of carbon dioxide molecules (which is not considered a deamination process) in the urea cycle, which also takes place in the liver. Urea and uric acid can safely diffuse into the blood and then be excreted in urine.
How does the nitrogen cycle affect the environment?
The nitrogen cycle causes many environmental issues such as global warming and pollution itself. The global increase in temperature is mainly due to the release of greenhouse gases. Greenhouse gases causes the blanket of air surrounding the earth to thicken, thus warming up the earth. The byproducts of the nitrogen cycle produce greenhouse effects, specifically in agricultural fields. Farmers input high amounts of ammonium nitrate to fertilize their soil to grow their crops. Consequently this increases the production of pollutants: nitrous oxide (N 2 O) and nitric oxide (NO), through denitrification. CO 2, another greenhouse gas, would also be produced from aerobic respiring microbes.
What are the chemical transformations of nitrogen?
In soil, most of the chemical transformations of nitrogen are due to microbial activities, such as nitrogen fixation, nitrification, immobilization, and denitrification. Learning how organisms behave in soil is critical for humans to understand the complex nitrogen cycle.
What is the role of dinitrogen in the nitrogen cycle?
Dinitrogen is an essential element for the nitrogen cycle. The symbiotic microbes fix the dinitrogen (N 2) into volatile ammonia, NH 3, by the process of nitrogen fixation. The ammonia is further protonated to ammonium, NH 4+, which is uptaken by plants to manufacture amino acids for growth. When plants decompose, the organic molecules present in the plant residues become a source of nitrogen and other nutrients to soil microbes. The microbes conduct mineralization (ammonification) to utilize the organic molecules as electron donors, acquiring energy and producing ammonium. In contrast, the transformation of organic nitrogen to ammonium is reversible through immobilization (assimilation) when the carbon to nitrogen ratio (C/N ratio) is high. Ammonium can also undergo nitrification to produce NO 3- in an aerobic environments. The NO 3- can be utilized by plants or by other organisms in anaerobic environments as an electron acceptor, can leach out by dissolving in water, or be reduced to dinitrogen gas via denitrification processes in anaerobic conditions. During the denitrification and nitrification processes nitrous oxide, N 2 O, is produced and escapes into atmosphere.
Why is denitrification important?
Denitrification is important in the continuation of the nitrogen cycle, as it is the step where gaseous dinitrogen is released back into the atmosphere. The natural cycle of denitrification comprises a series of enzymes such as bacterial NORs enzymes reduce nitrate to dinitrogen.
What are the effects of human input on the nitrogen cycle?
Anthropogenic Influences on the Soil Nitrogen Cycle. One major influence of human input is reduced biological nitrogen fixation. With excessive ammonium input, plants no longer need the symbiotic microbes to provide ammonium. As a result, the degree of symbiosis will be diminished.
What happens to the organic molecules in plants when they decompose?
When plants decompose, the organic molecules present in the plant residues become a source of nitrogen and other nutrients to soil microbes. The microbes conduct mineralization (ammonification) to utilize the organic molecules as electron donors, acquiring energy and producing ammonium.
How to reduce nitrogen in the biosphere?
Some strategies to reduce mobile nitrogen in the biosphere are bioremediation, reduction of emissions, and sequestration. Bioremediation can reduce the amount of nitrate from the environment by converting it into atmospheric nitrogen and nitric oxide. However, this has its own concerns because nitric oxide production must be limited to lessen effects of global warming. Reduction of emissions reduces the amount of nitrogen entering the biosphere, thus lessening the amount of mobile nitrogen available. Sequestration of nitrogen prevents nitrogen from becoming mobile and can be increased by selecting for crops with higher nitrogen uptake efficiency that are also able to form symbiotic relationships with nitrogen sequestering soil microorganisms. There has been renewed interest in studying these relationships in the attempt to increase nitrogen sequestration in soil.
What are the steps of the nitrogen cycle?
The nitrogen cycle is composed of four steps: nitrogen fixation, nitrification, ammonification , and denitrification . During the first step of the nitrogen cycle, nitrogen fixation , you and your boyfriend (N 2) are pulled out of the air by bacteria in the soil.
Why do you have to go through the nitrogen cycle?
The problem is, you like to hang really close to your nitrogen boyfriend in the air, joining together as N 2. You have a very strong bond to your nitrogen boyfriend, and the two of you are hard to break apart, which makes you unable to be used by living organisms. That is why you must go through the nitrogen cycle in order to be useful.
How does ammonium turn into nitrate?
The bacteria turns you both into ammonium by mixing you with hydrogen atoms. Ammonium composed of one nitrogen atom (blue) and four hydrogen atoms (white) In the next step, nitrification, is the transition of ammonia to nitrite and then nitrate through oxidation.
What happens to nitrogen during nitrogen fixation?
During nitrogen fixation, nitrogen gas from the air is converted into ammonium. During nitrification, ammonium is converted into nitrates, which are taken up by plants. The nitrates are used by the plants and animals, then returned to ammonium using ammonification.
What happens after an organism dies?
Regardless of what state you are in now, after the organism dies, you must be returned to the soil to be reused in the nitrogen cycle. This is exactly why it is called the nitrogen cycle - it is a set of repeated events.
What happens to the plant during ammonification?
During ammonification, the plant or animal that you are a part of dies. You are left to be converted back into ammonium by decomposers (bacteria and fungi that break down dead organisms). You are returned back into the soil and can then reenter the cycle.
Where is nitrogen found in a plant?
In a plant or animal, you, the nitrogen atom, may be found in a variety of different forms. You may be found in amino acids, which are the building blocks of proteins. You might also be found in DNA or in chlorophyll, the compound used by plants to capture the energy from the sun.
Glutamate Dehydrogenase
reversible — one of only three reactions in humans that can “fix” NH4+, i.e., covalently link it to carbon by the reverse reductive amination of α-ketoglutarate
Glutamine Synthetase
Glutamine Synthetase Glutamine is produced from glutamate by the addition of nitrogen to the glutamate γ carboxyl group by an ATP-dependent reaction catalyzed by Glutamine Synthetase — one of only three reactions in humans that can “fix” NH4+, i.e., covalently link it to carbon.

Overview
Deamination is the removal of an amino group from a molecule. Enzymes that catalyse this reaction are called deaminases.
In the human body, deamination takes place primarily in the liver, however it can also occur in the kidney. In situations of excess protein intake, deamination is used to break down amino acids for energy. The amino group is removed from the amino acid and converted to ammonia. The rest o…
Deamination reactions in DNA
Spontaneous deamination is the hydrolysis reaction of cytosine into uracil, releasing ammonia in the process. This can occur in vitro through the use of bisulfite, which deaminates cytosine, but not 5-methylcytosine. This property has allowed researchers to sequence methylated DNA to distinguish non-methylated cytosine (shown up as uracil) and methylated cytosine (unaltered).
Additional proteins performing this function
• APOBEC1
• APOBEC3A-H, APOBEC3G - affects HIV
• Activation-induced cytidine deaminase (AICDA)
• Cytidine deaminase (CDA)
See also
• Adenosine monophosphate deaminase deficiency type 1
• Hofmann elimination
Nitrogen Cycle Processes
- Natural Soil Nitrogen Cycle
Dinitrogen is an essential element for the nitrogen cycle. The symbiotic microbes fix the dinitrogen (N2) into volatile ammonia, NH3, by the process of nitrogen fixation. The ammonia is further protonated to ammonium, NH4+, which is uptaken by plants to manufacture amino acid… - Anthropogenic Influences on the Soil Nitrogen Cycle
One major influence of human input is reduced biological nitrogen fixation. With excessive ammonium input, plants no longer need the symbiotic microbes to provide ammonium. As a result, the degree of symbiosis will be diminished. Furthermore, the excessive ammonium provid…
Nitrogen Fixation
- Prokaryotes(bacteria) are responsible for the nitrogen fixation inside the soil. They mostly establish associative relationships with leguminous plants and other plant species. Yet, there are free-living bacteria like Azotobacter. All of the prokaryotes that are capable of nitrogen fixation have nitrogenase, which can fix nitrogen into ammonia. Many prokaryotes share the same enzy…
Nitrogen Ammonification
- Net nitrogen mineralization is commonly defined as the sum of concurrent ammonium production and consumption processes. However, in recent years, it has been shown that mineralization also is driven by depolymerization of nitrogen containing polymers available to use both by plants and microorganisms, and thus it is more correct to call the process ammonification or gross nitroge…
Nitrogen Immobilization
- Immobilization is the conversion of ammonium and nitrate to organic nitrogen, primarily as a result of the assimilation of ammonium by the microbial biomass.The process requires energy for the conversion of nitrate to ammonium and subsequent incorporation of ammonium into amino acids. Microorganisms assimilate ammonium by two secondary pathways - the end result of bot…
Nitrification
- Nitrification is a two-step process of ammonium (NH4) to nitrate (NO3) by soil bacteria. Ammonium is initially oxidized to nitrite by chemoautotrophs in the following manner:NH3 + O2 --> NO2- + H+ + H2O Chemoautotrophs are able to use carbon dioxide as a carbon source for the oxidation of ammonium. The most common bacteria that oxidizes ammonium is Nitrosomonas…
Denitrification
- Denitrification is important in the continuation of the nitrogen cycle, as it is the step where gaseous dinitrogen is released back into the atmosphere. The natural cycle of denitrification comprises a series of enzymes such as bacterial NORs enzymes reduce nitrate to dinitrogen. As a result, reduction of nitrates to gaseous dinitrogen follows a series of respiratory biochemical rea…
Environmental Impacts
- The nitrogen cycle causes many environmental issues such as global warming and pollution itself. The global increase in temperature is mainly due to the release of greenhouse gases. Greenhouse gases causes the blanket of air surrounding the earth to thicken, thus warming up the earth. The byproducts of the nitrogen cycle produce greenhouse effects, specifically in agricultu…
Current Research
- Although the importance of soil microbes in the nitrogen cycle is well understood, current research is focused on the particular niches that different organisms occupy. Many of these experiments have identified some distinctive roles originally unknown to scientists, and are being studied for possible applications in agriculture and bioremediation practices. One study highligh…