Why is lead used in a storage battery?
Various Uses of Lead:
- Lead Acid Battery. The principal consumption of Lead is for the Lead-Acid storage battery in which grid or plate is made of Lead or Lead with other metal more ...
- Rolled Extrusions. Lead Sheet is used in the building industry for flashings or weathering to prevent water penetration & for roofing and cladding.
- Lead Pipes. ...
- Pigments. ...
- Cable Sheathing. ...
What are the advantages of lead storage batteries?
Lead batteries connect, power and protect our everyday energy needs. With over 160 years of history, the benefits of lead batteries stand the test of time. Lead batteries are not a product of the past; they power innovative, safe energy storage solutions across the globe and are a critical piece in us building a more sustainable future.
How is a lead storage battery recharged?
In the CCCV method, lead-acid batteries are charged in three stages:
- DC Charging
- Charging
- Trickle Charging
What type of a battery is the lead storage battery?
lignosulfonateWater-soluble anionic polyelectrolyte polymers; they are byproducts from the production of wood pulp using sulfite pulping. A lead storage battery, also known as a lead-acid battery, is the oldest type of rechargeable battery and one of the most common energy storage devices.
See more

How does lead storage battery work?
Ans: Lead and lead dioxide, the active materials on the plate of the battery, react to lead sulfate in the electrolyte with sulphuric acid. The lead sulfate first forms in a finely divided, amorphous state, and when the battery recharges easily returns to lead, lead dioxide, and sulphuric acid.
What is lead storage battery in chemistry class 12?
A lead storage battery is the most important type of secondary cell having a lead anode a grid of lead packed with PbO2 as a cathode. A 38% solution of sulphuric acid is used as electrolyte. (Density 1.294gmL−1) The battery holds 3.5L of the acid.
What do you mean by lead storage cell?
Solution : Lead - acid storage battery. It is the most frequently used battery in automobiles. As the voltage of individual cell in only 2 V, therefore 3 or 6 such cells are joined in series to get a 6V or 12V battery. The cell anode generally conists of a number of plates joined in parallel.
What are storage batteries in chemistry?
Flow batteries store energy by means of a reversible chemical reaction. The energy is stored in two liquid electrolyte solutions. The energy capacity and the rated power of the system are independent of one another.
What type of battery is lead storage?
Therefore, a lead storage battery is a secondary type of battery. Note: Primary batteries are used only for once. They become dead after one use. Example of the primary battery is leclanche cell and mercury cell.
Is lead storage battery and lead-acid battery the same?
A lead storage battery, also known as a lead-acid battery, is the oldest type of rechargeable battery and one of the most common energy storage devices. These batteries were invented in 1859 by French physicist Gaston Planté, and they are still used in a variety of applications.
Is lead storage battery a secondary cell?
Lead storage cell is a secondary voltaic cell. It can be recharged by reversing the direction of current flow and thereby regenerating the original reactants. To reverse the direction of current flow, a potential slightly greater than that generated by cell is applied across the electrodes.
What is a battery lead?
A lead acid battery consists of a negative electrode made of spongy or porous lead. The lead is porous to facilitate the formation and dissolution of lead. The positive electrode consists of lead oxide. Both electrodes are immersed in a electrolytic solution of sulfuric acid and water.
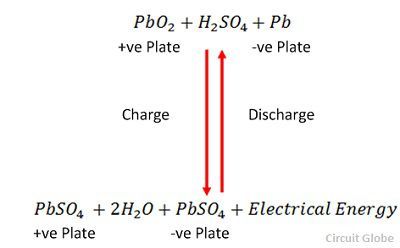
What happens when lead storage battery is charged?
During the charging of lead storage batteries, it acts as an electrolytic cell and during the discharging of the lead storage battery, it acts as a galvanic cell. During the charging of the lead storage battery, the reactions get reversed and the cathode becomes anode and anode becomes cathode.
What is the purpose of the storage battery?
In contrast to primary cells, which are discharged once and then discarded, storage batteries can be supplied with direct current (DC) of the correct polarity and recharged to or near their original energy content and power capability—i.e., they can repeatedly store electrical energy.…
What are storage batteries?
Definition of storage battery : a cell or connected group of cells that converts chemical energy into electrical energy by reversible chemical reactions and that may be recharged by passing a current through it in the direction opposite to that of its discharge. — called also storage cell.
What does battery storage mean?
Battery storage, or battery energy storage systems (BESS), are devices that enable energy from renewables, like solar and wind, to be stored and then released when customers need power most.
Where is lead storage battery used?
They are used in back-up power supplies for alarm and smaller computer systems (particularly in uninterruptible power supplies; UPS) and for electric scooters, electric wheelchairs, electrified bicycles, marine applications, battery electric vehicles or micro hybrid vehicles, and motorcycles.
Is lead storage battery a secondary cell?
Lead storage cell is a secondary voltaic cell. It can be recharged by reversing the direction of current flow and thereby regenerating the original reactants. To reverse the direction of current flow, a potential slightly greater than that generated by cell is applied across the electrodes.
What's in a lead acid battery?
The lead-acid battery consists of (in the charged state) electrodes of lead metal (Pb) and lead oxide (PbO2) in an electrolyte of about 37% sulfuric acid (H2SO4). In the discharged state both electrodes turn into lead sulfate (PbSO4) and the electrolyte loses its dissolved sulfuric acid and becomes primarily water.
What is lead acid battery used for?
The lead-acid battery represents the oldest rechargeable battery technology. Lead acid batteries can be found in a wide variety of applications including small scale power storage such as UPS systems, starting lighting and ignition power sources for automobiles, along with large, grid-scale power systems.
Lead Storage Batteries Definition
The battery that uses lead peroxide and sponge lead to convert chemical energy to electrical energy is called lead storage battery
Overview of Lead Storage Batteries
Lead storage batteries were invented in the year 1859 by a French physicist Gaston Plante. Lead batteries are rechargeable batteries. The electrodes are made up of lead grids. The opening of the anodic grid is filled with porous lead and the cathodic opening is filled with lead dioxide.
Construction of Lead Batteries
A lead-acid battery consists of various parts, which include a container, plate, active materials, separators, and battery terminals. The two main parts of the lead-acid battery are the plates and the container. The container is a store of chemical energy while the plates convert this stored energy into electrical energy.
Working Principle of Lead Battery
The sulfuric acid and hydrogen acid are formed when each hydrogen ion takes one electron from the cathode, and each sulfate ions takes the two negative ions from the anode.
Charging of Lead Acid Batteries
The sulfuric acid that exists between the lead discharge batteries decomposes over some time and hence it needs to be replaced. Sometimes the plate that exists in those batteries changes itself on its own; due to this reason the charging of these batteries becomes less.
What is a Lead-acid Battery?
The Lead-acid battery is one of the oldest types of rechargeable batteries. These batteries were invented in the year 1859 by the French physicist Gaston Plante.
Why is it important to check a battery periodically?
In cases where the sulphuric acid in the battery (or some other component of the battery) has undergone decomposition, the charging process may become inefficient. Therefore, it is advisable to check the battery periodically.
Why do car batteries need to be charged?
When car batteries spend considerable durations of time in their discharged states, the lead sulfate build-up may become extremely difficult to remove. This is the reason why lead-acid batteries must be charged as soon as possible (to prevent building up of lead sulfate). Charging of the lead batteries is usually done by providing an external current source.
How does a lead battery charge?
Charging of the lead batteries is usually done by providing an external current source. A plug is inserted which is linked to the lead-acid battery and chemical reaction proceeds in the opposite direction. In cases where the sulphuric acid in the battery (or some other component of the battery) has undergone decomposition, ...
What happens when a lead battery decomposes?
Sometimes, the plates change their structure by themselves. Eventually, the battery becomes less efficient and should be charged or changed.
How is lead sulfate formed?
Lead sulfate is formed at both the electrodes. Two electrons are also transferred in the complete reaction. The lead acid battery is packed in a thick rubber or plastic case to prevent leakage of the corrosive sulphuric acid.
What is the process of recharging a secondary battery?
The chemical process of extracting current from a secondary battery (forward reaction) is called discharging.
What is the lead grid in a battery?
T he electrodes of the cells in a lead storage battery consist of lead grids. The openings of the anodic grid is filled with spongy (porous) lead. The openings of the cathodic grid is filled with lead dioxide { PbO2 }. Dilute sulfuric acid { H2SO4 } serves as the electrolyte. When the battery is delivering a current, i.e. discharging, the lead at the anode is oxidized:
How does a lead storage cell recharge?
T he lead storage cell can be recharged by passing a current in the reverse direction. The half-reactions are the exact reverse of those that occur when the cell is operating as a voltaic cell.
What is float charging?
Float charging is most commonly used for backup and emergency powerapplications where the discharge of the battery is infrequent. During float chargingthe charger, battery, and load are connected in parallel. The charger operates offthe normal power supply which provides current to the load during operation. Inthe event of normal power supply failure, the battery provides backup power untilthe normal power supply is restored. Since most equipment requires alternatingcurrent, a rectifier circuit is usually added between the battery and the load. Floatchargers are typically constant-voltage chargers that operate at a low voltage. Operating the charger at a low voltage, usually less than about 2.4 V per cell, keepsthe charging current low and thus minimizes the damaging effects of high-currentovercharging.
What is a constant voltage charger?
Constant-voltage (often called constant-potential) chargers maintain nearly thesame voltage input to the battery throughout the charging process, regardless of thebattery's state of charge . Constant-voltage chargers provide a high initial current tothe battery because of the greater potential difference between the battery andcharger. A constant-voltage charger may return as much as 70% of the previousdischarge in the first 30 minutes. This proves useful in many battery applicationsinvolving multiple discharge scenarios. As the battery charges its voltage increasesquickly. This reduces the potential that has been driving the current, with acorresponding rapid decrease in charge current as depicted in Figure 16. As aresult, even though the battery reaches partial charge quickly, obtaining a fullcharge requires prolonged charging.
What is battery maintenance?
Proper maintenance will prolong the life of a battery and will aid in assuring that it is capable ofsatisfying its design requirements. A good battery maintenance program will serve as avaluable aid in determining the need for battery replacement. Battery maintenance shouldalways be performed by trained personnel knowledgeable of batteries and the safetyprecautions involved.
What is a stationary battery?
Theyare used for applications where power is necessary only on a standby or emergencybasis. Stationary batteries are infrequently discharged. Stationary batteries remain on acontinuous float charge so that they can be used on demand. The largest types ofstationary batteries are those used for electrical load leveling. Load-leveling batteriesstore electrical energy for times of peak power demand and are taken off-line during
What is SLI battery?
SLI batteries are used by most people every day and are produced in greater numbers thanany other type of lead-acid storage battery. These are used to start automobiles and mostother kinds of internal combustion engines. They are not suitable for deep dischargeapplications, but excel for uses needing a high current for a brief time. They are usuallycharged in a "partial float" manner, meaning that the battery only receives a float chargewhile the vehicle is running. A cutaway view of a typical SLI battery is shown in Figure 12. SLI batteries are usually of the flat pasted plate design.
What is the grid structure of a battery made of?
Other metals in small quantities are alloyed with leadfor added strength and improved electrical properties. The most commonly alloyedmetals are antimony, calcium, tin, and selenium.
What is the name of the electrode for a lead acid battery?
The simplest method for the construction of lead-acid battery electrodes is the planté plate, named after the inventor of the lead-acid battery. A planté plate is merely a flat platecomposed of pure lead. Since the capacity of a lead-acid battery is proportional to the surfacearea of the electrodes that is exposed to the electrolyte, various schemes are employed toincrease the surface area of the electrodes per unit volume or weight. Planté plates aregrooved or perforated to increase their surface area. A typical planté plate is shown in Figure
