
What is needed to change the phase of matter? Evaporation involves a liquid becoming a gas and sublimation is the change of a solid directly to a gas. Phase changes require either the addition of heat energy (melting, evaporation, and sublimation) or subtraction of heat energy (condensation and freezing).
What causes matter to change phases?
What makes matter change its phase? If we heat ice then it melts. If we heat liquid water then it evaporates. It changes its phase from solid to liquid to vapor or gas. Ice can change into vapor directly by sublimation. If we cool it then its phases change in the reverse direction. Pressure affects the transition points.
What are the 6 phase changes of matter?
What are the six phases of matter? There are at least six: solids, liquids, gases, ... Examples of phase changes are melting (changing from a solid to a liquid), freezing (changing from a liquid to a solid), evaporation (changing from a liquid to a gas), and condensation (changing from a gas to a liquid). ...
What causes matter to change its phase?
Examples of Sublimation:
- Dry ice
- Moth balls sublime
- Frozen foods will sublime and you will find ice crystals inside of the box or bag.
What is needed for matter to change phase?
Section Summary
- Most substances have three distinct phases: gas, liquid, and solid.
- Phase changes among the various phases of matter depend on temperature and pressure.
- The existence of the three phases with respect to pressure and temperature can be described in a phase diagram.
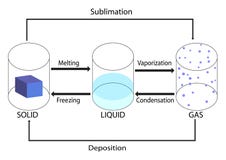
What is phase change?
A phase change is when matter changes to from one state (solid, liquid, gas, plasma) to another. (see figure 1). These changes occur when sufficient energy is supplied to the system (or a sufficient amount is lost), and also occur when the pressure on the system is changed.
What is the energy associated with these transitions called?
The energy associated with these transitions is called latent heat. Water is a substance that has many interesting properties that influence its phase changes.
How many phase changes are there in matter?
Matter exists as solids, liquids, gases, and plasma phases or states. A phase change is a transition of matter from one state to another. There are a total of eight phase changes that can occur. Let's look at what happens in each change.
How does a phase change happen?
A phase change is a transition of matter from one state to another. Phase changes occur regularly in nature and can be conducted by people. The phase in which matter exists is determined by the pressure and temperature of its surroundings. By manipulating the pressure and temperature, you can change the phase of the matter.
What happens when a gas changes directly to a liquid?
Condensation is when a gas changes directly to a liquid. Vaporization happens when a liquid changes directly to a gas. Ionization occurs when gas changes to plasma. Recombination occurs when plasma changes to gas. Learning Outcomes. Studying this lesson on the phase changes of matter could prepare you to subsequently:
What are some examples of phase changes?
For example, you have probably witnessed freezing, melting, and vaporization just by making ice, melting ice, and boiling water. Condensation often occurs on the outside of cold beverage containers.
What is the difference between plasma and gas?
A gas is any matter that does not have a fixed shape or volume. Gases take the shape of their container and can expand or contract to change their volume. Plasma is matter that exists in the form of high energy charged particles. A phase change is a transition of matter from one state to another.
What does it mean when a liquid has a definite volume?
This means that liquids retain their volume but take the shape of the container that is holding them.
What is the definition of phase change?
Definition of Phase Changes of Matter. By definition, matter is anything that has mass and takes up space. Matter exists as solids, liquids, gases, and plasma phases or states. A solid is any matter in a form that has a definite shape and volume. This means that no matter where you put a solid, its shape and volume will stay the same.
What is the process of a solid changing to a liquid called?
The process in which a solids change to a liquid is called melting. The melting point is the temperature at which a solids change to a liquid.
When a substance absorbs energy, the atoms and molecules move more rapidly?
When a substance absorbs energy the atoms and molecules move more rapidly and this increased kinetic energy pushes particles far enough, that they change form. This energy is usually heat or thermal energy. In this article, let us understand the science behind the changing states of matter.
What happens to the molecules when the temperature increases?
When the temperature or pressure increases, the interaction between the molecules increases. Similarly, when the temperature decreases, it is easier for molecules and atoms to settle into a more rigid structure. Freezing Melting Vaporization Condensation Sublimation Questions.
How does water transfer to solid ice?
Heat transfer occurs between the warmer tray and the colder air in the freezer. The warm water loses heat to the cold air in the freezer. This heat transfer occurs until no energy is available for the particles to slide past each other. This forces them to remain in fixed positions, locked in place by the force of attraction between them. This way liquid water is changed into solid ice. The process of liquid water changing to solid ice is termed as freezing. The temperature at which it occurs is known as the freezing point.
What is the process of a liquid boiling and changing to a gas called?
The bubbles rise through the water and escape from the pot as steam. The process in which a liquid boils and changes to a gas is called vaporization. The temperature at which a liquid boils is its boiling point.
How does water change to gas?
This happens as particles of liquid water gain enough energy to completely overcome the force of attraction between them and change to the gaseous state. The bubbles rise through the water and escape from the pot as steam. The process in which a liquid boils and changes to a gas is called vaporization. The temperature at which a liquid boils is its boiling point.
What happens when you fill a pot with cold tap water and heat it on a hot stovetop?
If you fill a pot with cold tap water and heat it on a hot stovetop, the water heats up. Heat energy travels from the stovetop to the pot, and the water absorbs the energy from the pot. What happens to the water next?
What are the three phases of matter?
There are liquids like water, honey, and juice. And there are gases, we breathe air and the helium in balloons. The main difference between the three phases is how fast the matter’s atoms move.
What is matter made of?
Everything is made of matter, and matter is made of atoms. Matter exists in three phases, solids, liquids, and gases. Adding or taking away energy makes matter change phase.
Why do atoms move around?
All atoms move around because they have energy. The more energy that’s in something, the faster the atoms move. Atoms in an ice cube don’t move very much – they’re frozen in place. The atoms in liquid water slip and slide around – that’s why you can pour it and spill it.
Why does water vapor move so fast?
Water vapor atoms are moving pretty fast – that’s why they float around in air (a mixture of other gasses). Changing an object’s phase of matter is just a matter (ha, ha) of adding energy to atoms or taking it away. Heating adds energy to atoms, and cooling takes energy away from atoms.
