
Nitrogen fixation is a chemical process by which molecular nitrogen (N 2), with a strong triple covalent bond, in the air is converted into ammonia (NH 3) or related nitrogenous compounds, typically in soil or aquatic systems but also in industry.
Full Answer
What is nitrogen fixation and why is it important?
Nitrogen fixation is a process whereby bacteria in the soil convert atmospheric nitrogen ( N2 gas) into a form that plants can use. The reason this process is so important is that animals and plants cannot use atmospheric nitrogen directly. Bacteria convert it into ammonium ( NH4+ ), which then plants can absorb.
What does it mean to have nitrogen fixed?
Nitrogen Fixation- it is a process by which the that is present in the atmosphere is converted into (Ammonia) or the other nitrogenous-based compound which is used by the plants. Atmospheric Nitrogen is non-reactive which means it cannot be obtained and used by the plants.
What two things can fix nitrogen?
Industrial processes
- Frank-Caro process. In 1898 Frank and Caro developed a way to fix nitrogen in the form of calcium cyanamide. ...
- Haber process. The most common ammonia production method is the Haber process. ...
- Homogeneous catalysis. Much research has been conducted on the discovery of catalysts for nitrogen fixation, often with the goal of reducing energy requirements.
What is nitrogen fixation and who can do this?
What is nitrogen fixation with example? some bacteria can convert N 2 into ammonia by the process termed nitrogen fixation; these bacteria are either free-living or form symbiotic associations with plants or other organisms (e.g. termites, protozoa) other bacteria bring about transformations of ammonia to nitrate, and of nitrate to N 2 or other nitrogen gases.
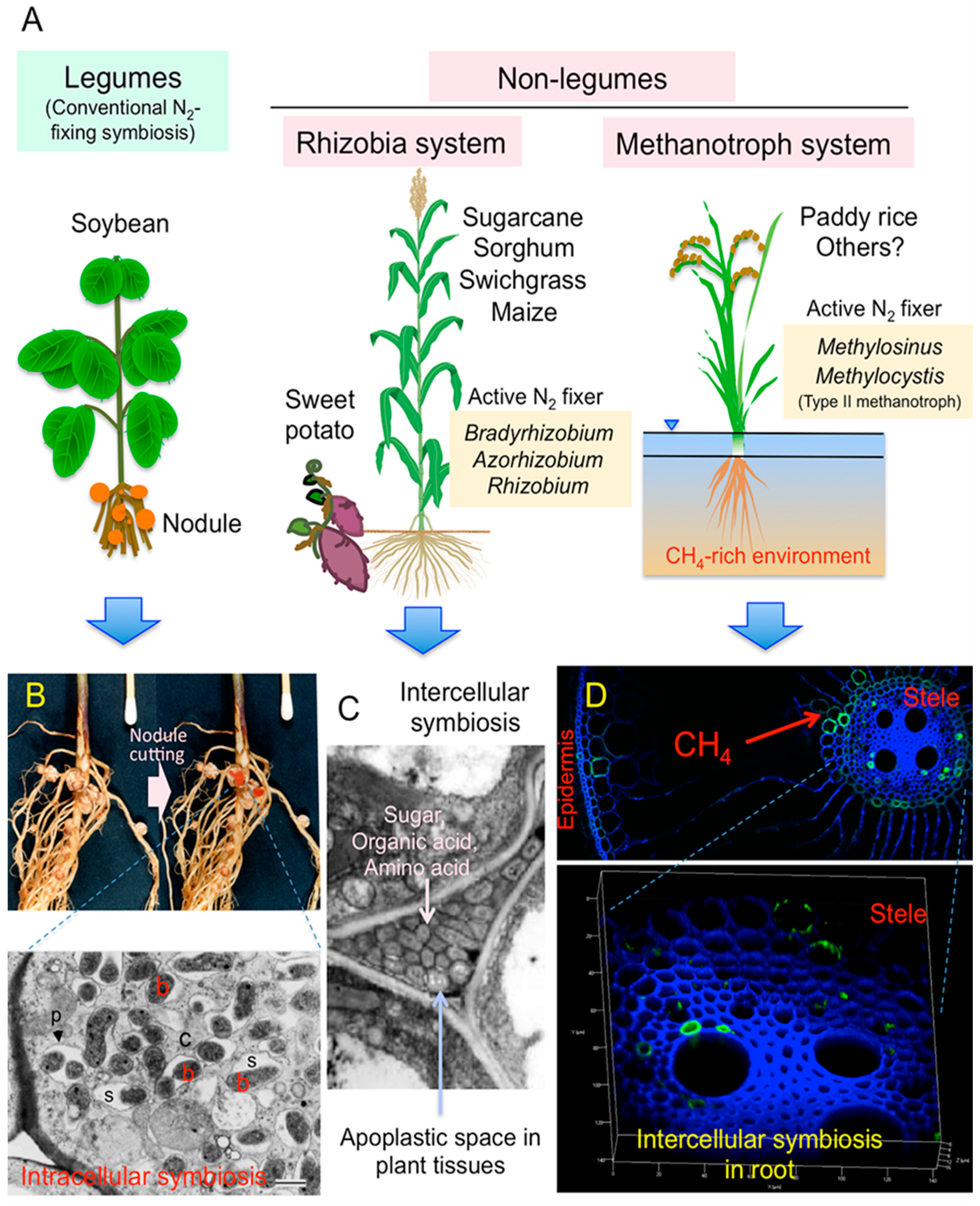
What is nitrogen-fixing short answer?
nitrogen fixation, any natural or industrial process that causes free nitrogen (N2), which is a relatively inert gas plentiful in air, to combine chemically with other elements to form more-reactive nitrogen compounds such as ammonia, nitrates, or nitrites.
What does nitrogen fixation do?
Nitrogen fixation is the process by which nitrogen is taken from its molecular form (N2) in the atmosphere and converted into nitrogen compounds useful for other biochemical processes. Fixation can occur through atmospheric (lightning), industrial, or biological processes.
Where are nitrogen-fixing?
soilMost nitrogen fixation occurs naturally, in the soil, by bacteria. In Figure 3 (above), you can see nitrogen fixation and exchange of form occurring in the soil. Some bacteria attach to plant roots and have a symbiotic (beneficial for both the plant and the bacteria) relationship with the plant [6].
What are the 3 types of nitrogen fixation?
(a) Nitrogen fixation (b) Ammonificiation (c) Nitrification (d) Denitrification.
What is an example of a nitrogen-fixing plant?
Plants that contribute to nitrogen fixation include the legume family – Fabaceae – with taxa such as clover, soybeans, alfalfa, lupins, peanuts, and rooibos.
Is nitrogen-fixing bad for the environment?
Biological nitrogen fixation can fuel CO2 sequestration by forests but can also stimulate soil emissions of nitrous oxide (N2O), a potent greenhouse gas.
What trees are nitrogen-fixing?
Black Locust, Mimosa, Alder, Redbud, Autumn Olive, Kentucky Coffee Tree, Golden Chain Tree, Acacia, Mesquite and others are examples of trees that support nitrogen in soil with the help of bacteria. These NFTs pull the element out of the atmosphere and build a storehouse of the gas through their nodule root formation.
What are the two methods of fixing nitrogen?
The two types of nitrogen fixation are: (1) Physical Nitrogen Fixation and (2) Biological Nitrogen Fixation. Apart from carbon, hydrogen and oxygen, nitrogen is the most prevalent essential macro-element in living organisms.
Who fixes the nitrogen in atmosphere?
BacteriaAnswer: Solution 6: These microbes are commonly called biological nitrogen fixers. Bacteria such as rhizobium and certain blue-green algae present in the soil can fix atmospheric nitrogen and convert into usable nitrogenous compounds, which are used by plants for the synthesis of plant proteins and other compounds.
Who fix the nitrogen in soil?
Nitrogen fixing bacteria are microbes or bacteria that fix nitrogen in soil. Many bacteria species may fix nitrogen in soil, but the most common are Azotobacter, Rhizobia, Acetobacter, and some blue green algae.
Where is nitrogen manufactured?
Nitrogen is produced commercially almost exclusively from air, most commonly by the fractional distillation of liquid air.
What are the three types of nitrogen fixation?
1. Biological nitrogen fixation 2. Atmospheric nitrogen fixation 3. Industrial nitrogen fixation
What are the 7 steps of the nitrogen cycle?
1. Nitrogen fixation. 2. Assimilation 3. Ammonification 4. Nitrification 5. Denitrification 6. Dissimilatory nitrate reduction to ammonium 7. Anaer...
How nitrogen fixation takes place in the soil?
Nitrogen fixation is carried out naturally in soil by microorganisms that are termed diazotrophs. These include bacteria such as Azotobacter and Ar...
What is nitrogen fixation, and why is it important?
The process of fixing atmospheric nitrogen into the soil through various means such as lightning or by certain organisms is called nitrogen fixatio...
What is an example of nitrogen fixation?
Nitrogen fixation in the root nodules of leguminous plants or by the symbiotic association of Azolla and Anabaena in the paddy fields are two examp...
What is the role of nitrogenase in a cell?
The protein complex nitrogenase is responsible for catalyzing the reduction of nitrogen gas (N 2) to ammonia (NH 3 ). In Cyanobacteria, this enzyme system is housed in a specialized cell called the heterocyst. The production of the nitrogenase complex is genetically regulated, and the activity of the protein complex is dependent on ambient oxygen concentrations, and intra- and extracellular concentrations of ammonia and oxidized nitrogen species (nitrate and nitrite). Additionally, the combined concentrations of both ammonium and nitrate are thought to inhibit N Fix, specifically when intracellular concentrations of 2-oxoglutarate (2-OG) exceed a critical threshold. The specialized heterocyst cell is necessary for the performance of nitrogenase as a result of its sensitivity to ambient oxygen.
What is the nitrogenase complex?
The protein complex nitrogenase is responsible for catalyzing the reduction of nitrogen gas (N 2) to ammonia (NH 3 ). In Cyanobacteria, this enzyme system is housed in a specialize cell called the heterocyst. The production of the nitrogenase complex is genetically regulated, and the activity of the protein complex is dependent on ambient oxygen concentrations, and intra- and extracellular concentrations of ammonia and oxidized nitrogen species (nitrate and nitrite). Additionally, the combined concentrations of both ammonium and nitrate are thought to inhibit N Fix, specifically when intracellular concentrations of 2-oxoglutarate (2-OG) exceed a critical threshold. The specialized heterocyst cell is necessary for the performance of nitrogenase as a result of its sensitivity to ambient oxygen.
Why is nitrogen fixation important?
Nitrogen fixation is essential to life because fixed inorganic nitrogen compounds are required for the biosynthesis of all nitrogen-containing organic compounds , such as amino acids and proteins, nucleoside triphosphates and nucleic acids.
How much nitrogen does red clover fix?
For example, nitrogen fixation by red clover can range from 50 to 200 lb./acre.
How is ammonia produced?
The most common ammonia production method is the Haber process. The Haber-Bosch nitrogen reduction process for industrial fertilizer production revolutionized modern day technology. Fertilizer production is now the largest source of human-produced fixed nitrogen in the terrestrial ecosystem. Ammonia is a required precursor to fertilizers, explosives, and other products. The Haber process requires high pressures (around 200 atm) and high temperatures (at least 400 °C), which are routine conditions for industrial catalysis. This process uses natural gas as a hydrogen source and air as a nitrogen source. The ammonia product has resulted in an intensification of nitrogen fertilizer globally and is credited with supporting the expansion of the human population from around 2 billion in the early 20th century to roughly 8 billion people now.
What was the first species to use diatomic nitrogen?
In 1901 Beijerinck showed that azotobacter chroococcum was able to fix atmospheric nitrogen. This was the first species of the azotobacter genus, so-named by him. It is also the first known diazotroph, species that use diatomic nitrogen as a step in the complete nitrogen cycle .
Why is nitrogen important to life?
Importance of nitrogen. Atmospheric nitrogen is inaccessible to most organisms, because its triple covalent bond is very strong. Life takes up fixed nitrogen in various ways. Considering atom acquisition, for every 100 atoms of carbon, roughly 2 to 20 atoms of nitrogen are assimilated.
What are the Types of Nitrogen Fixation?
Nitrogen fixation is carried by physicochemical and biological means. Only 10 % of natural nitrogen fixation takes place by physicochemical means, whereas 90 % is carried out by biological means. Thus, we can classify nitrogen fixation in following two types:
What is the process of converting nitrogen into nitrogenous compounds?
The process of conversion of atmospheric nitrogen into nitrogenous compounds by microorganisms such as bacteria, fungi and algae is known as Biological Nitrogen Fixation (BNF) or diazotrophy. Some prokaryotes such as bacteria and cyanobacteria that can fix atmospheric nitrogen are called nitrogen fixers or diazotrophs.
How is nitrogen converted into ammonia?
Nitrogen needs to be converted into different forms like ammonia, nitrites or nitrates through nitrogen fixation via various means as the molecular form of nitrogen is of no use to plants and animals. Nitrogen plays a vital role in the life of living organisms. Hence nitrogen fixation is a very necessary and crucial process. Nitrogen fixation is carried by physicochemical (only 10 % and biological 90 % means. Biological nitrogen fixation is mainly performed by a set of microorganisms such as bacteria, fungi, cyanobacteria, etc.
How does nitrogen fixation occur?
Nitrogen fixation occurs with the help of microorganisms as a part of the nitrogen cycle either by natural means or via industrial methods.
Why is nitrogen fixation important?
The process of nitrogen fixation is very important as the molecular form of nitrogen is of no use to plants and animals.
What is the name of the enzyme that reduces nitrogen to ammonia?
The atmospheric nitrogen is reduced to ammonia in the presence of a catalyst known as nitrogenase. This enzyme is found naturally in certain microorganisms like symbiotic ( Rhizobium and Frankia) and non-symbiotic or free-living ( Azospirillum, Azotobacter and BGA).
What did Beijerinck discover?
Later, Beijerinck discovered that there were certain bacteria that were responsible for fixing nitrogen. He named them ‘rhizobia’. He also discovered the symbiotic relationship between the root nodules of leguminous plants and the bacteria.
How does grazing affect nitrogen fixation?
Rates of nitrogen fixation in reef environments are variable and can be affected by the presences of grazers. Grazing by Acanther plancii , the crown of thorns starfish on corals resulted in high rates of nitrogen fixation on the coral skeletons after the outbreak of this starfish ( Larkum, 1988 ). Similarly, the sea urchin Diadema antillarum was shown to significantly increase nitrogen fixation when grazing on “algal turf” in reef environments, as compared to areas where no sea urchins were present. The “diminutive” tightly cropped “algal turf” assemblage contained a significant proportion of cyanobacteria in addition to benthic diatoms and dinoflagellates ( Williams and Carpenter, 1997 ). Grazers also aid in nitrogen cycling by excreting nitrogenous waste which enhances algal turf, while simultaneously cropping it ( Williams and Carpenter, 1997 see below). Fish grazing may also be important in maintaining rates of nitrogen fixation on reefs, by keeping other benthic algae in check. Some diazotrophic cyanobacteria are less palatable than other algae, as they often have chemical deterrents, and also tend to be less nutritionally complete, lacking some essential fatty acids, and hence macroalgae are often preferred by grazers ( Capper et al., 2005; O'Neil, 1999 ). Therefore, cyanobacteria may have less competition for growing space and expand into areas where the more palatable species have been removed by grazers ( Wilkinson and Sammarco, 1983 ).
What are symbiotics with sponges?
Invertebrate diazotrophic symbioses have been reported from reef systems including reef sponges that acquire fixed nitrogen via associated cyanobacteria ( Wilkinson and Fay, 1979 ). Symbiotic cyanobacteria and bacteria are found in almost all marine sponges ( Mohamed et al., 2006; Thacker, 2005) and may form mutualistic associations with hosts especially if the symbiont provides fixed C or N. The cyanobacterium Oscillatoria spongieliae has been found within several species of sponges including the reef sponge Dysidea herbacea on the Great Barrier Reef (GBR) ( Flowers et al., 1998; Ridley et al., 2005 ). Synechococcus has also been described from several species of sponge, which are genetically distinct from free living planktonic species of Synechococcus ( Thacker, 2005 ). In some instances metabolic products (e.g., fixed carbon) are translocated from symbiont to host ( Arillo et al., 1993 ). However, in most cases the relationship between host and symbiont are not well defined ( Ridley et al., 2005 ).
How does nitrogen fixation affect soil?
Consequently, disturbance can result in large decreases in soil nitrogen through a combination of reduced biological nitrogen input and elevated gaseous loss of nitrogen and soil loss. Short-term reductions in nitrogen fixation range up to 100%. Long-term studies show a 42% decrease in soil nitrogen 25 years following disturbance.
Why is NFIX lower than other N cycles?
NFIX rates in sediments are often lower than rates of the other N cycle processes because NFIX is inhibited by the high concentrations of bioavailable dissolved inorganic nitrogen (DIN) that are often present in sediments ( Fig. 19.1, arrow 1; Capone, 1988; Seitzinger and Gardner, 1987 ).
What is the effect of nitrogen fixation on the atmosphere?
NO y is almost completely emitted to the atmosphere, where it can increase concentrations of gases such as ozone via photochemical reactions. In the lower atmosphere, the troposphere, nitrogen oxides can form a variety of nitrate containing organic compounds (e.g., peroxyacytyl nitrate, PAN) and contribute to formation of excessive levels of ozone after reactions with volatile organic compounds (VOCs); both PAN and ozone have ecotoxicological effects. In addition, nitrogen oxides can form the acidic HNO 2 and HNO 3 which can have ecotoxicological effects when deposited on aquatic and terrestrial ecosystems.
What is the fixation rate of speckled alder?
fixation rates of speckled alder ( Alnus rugosa = A. incana ssp. rugosa) have been measured at 85 to 167 kg N 2 ha −1 yr −1. Some nonsymbiotic, aerobic and anaerobic bacteria, and blue-green algae are also significant nitrogen fixers in wetlands.
How does grazing help with nitrogen cycling?
Grazers also aid in nitrogen cycling by excreting nitrogenous waste which enhances algal turf, while simultaneously cropping it ( Williams and Carpenter, 1997 see below). Fish grazing may also be important in maintaining rates of nitrogen fixation on reefs, by keeping other benthic algae in check.
What is the nitrogen fixation method?
There are two key methods of natural nitrogen fixation: Lightning provides energy to react water (H 2 O) and nitrogen gas (N 2) to form nitrates (NO 3) and ammonia (NH 3 ). Rain and snow carry these compounds to the surface, where plants use them. Microorganisms that fix nitrogen are known collectively as diazotrophs.
How do diazotrophs get nitrogen?
Diazotrophs convert nitrogen from the atmosphere into ammonia, which can be converted into nitrates or ammonium compounds. Plants and fungi use the compounds as nutrients. Animals obtain nitrogen by eating plants or animals that eat plants. There are multiple synthetic methods for fixing nitrogen:
What is fixed nitrogen?
Here is a look at what fixed nitrogen is and an explanation of different fixation processes. Fixed nitrogen is nitrogen gas, N 2, that has been converted to ammonia (NH 3, an ammonium ion (NH 4, nitrate (NO 3, or another nitrogen oxide ...
What is the process of forming cyanamide?
The cyanamide process forms calcium cyanamide (CaCN 2, also known as Nitrolime) from calcium carbide that is heated in a pure nitrogen atmosphere. Calcium cyanamide then is used as a plant fertilizer. Lord Rayleigh devised the electric arc process in 1895, making it the first synthetic method of fixing nitrogen.
What are the organisms that fix nitrogen?
Bacteria. Microorganisms that fix nitrogen are known collectively as diazotrophs. Diazotrophs account for about 90% of natural nitrogen fixation. Some diazotrophs are free-living bacteria or blue-green algae, while other diazotrophs exist in symbiosis with protozoa, termites, or plants.
Why is nitrogen unavailable in the atmosphere?
Living organisms need nitrogen to form nucleic acids, proteins, and other molecules. However, the nitrogen gas, N 2, in the atmosphere is unavailable for use by most organisms because of the difficulty breaking the triple bond between nitrogen atoms.
How does an electric arc work?
An electric arc reacts oxygen and nitrogen in air to form nitrogen oxides. The oxide-laden air is bubbled through water to form nitric acid. Helmenstine, Anne Marie, Ph.D.
What are some examples of symbiotic nitrogen fixers?
Examples of symbiotic nitrogen-fixing bacteria include Rhizobium, which is associated with plants in the pea family, and various Azospirillum species, which are associated with cereal grasses. Free-living nitrogen-fixers include the cyanobacteria Anabaena and Nostoc and genera such as Azotobacter, Beijerinckia, and Clostridium.
Why are legumes good for you?
Because of these bacteria, legumes have the nitrogen necessary to make lots of proteins, which, in turn, is why beans are such a good source of dietary protein for humans and other animals.
What are the two types of bacteria that fix nitrogen?
An overview of nitrogen fixation. Two kinds of nitrogen-fixing bacteria are recognized. The first kind, the free-living (nonsymbiotic) bacteria, includes the cyanobacteria (or blue-green algae) Anabaena and Nostoc and genera such as Azotobacter, Beijerinckia, and Clostridium.
What is the function of nitrogen-fixing bacteria in plants?
The symbiotic nitrogen-fixing bacteria invade the root hairs of host plants, where they multiply and stimulate formation of root nodules, enlargements of plant cells and bacteria in intimate association. Within the nodules the bacteria convert free nitrogen to ammonia, which the host plant utilizes for its development.
What is the purpose of inoculating seeds?
To ensure sufficient nodule formation and optimum growth of legumes (e.g., alfalfa, beans, clovers, peas, soybeans), seeds are usually inoculated with commercial cultures of appropriate Rhizobium species, especially in soils poor or lacking in the required bacterium.
What family is the pea family?
…pea family (Fabaceae) host symbiotic nitrogen-fixing bacteria, and many plant roots also form intricate associations with mycorrhizal soil fungi; a number of non-photosynthetic mycoheterotrophic plants, such as Indian pipe, rely exclusively on these fungi for nutrition.…
What are the most important hosts for nitrogen-fixing bacteria?
Symbiotic, or mutualistic, species live in root nodules of certain plants. Plants of the pea family, known as legumes, are some of the most important hosts for nitrogen-fixing bacteria, but a number of other plants can also harbour these helpful bacteria.
What Do "Cover Crops" Have to Do With Nitrogen-Fixation?
The nitrogen-fixing plants that people speak of most often are cover crops of the pea, or "legume" family, because these plants are easy to work with (they can simply be rototilled under for you to release the valuable nitrogen). Certain trees and shrubs can also fix nitrogen, but they are seldom mentioned in this connection because they are not as easy to work with (you cannot turn them under with a rototiller).
How to tell if you have a nitrogen deficiency?
That’s why one of the important warning signs of nitrogen deficiency is yellowing, pale green leaves....". By exploiting the process of nitrogen fixation, you can obtain this plant nutrient for your soil without resorting to chemical fertilizers. For best results, inoculate your legume seeds with Rhizobium bacteria.
Why do you value nitrogen in plants?
Landscapers, gardeners, and farmers value nitrogen-fixing plants for their ability to contribute an essential plant nutrient (namely, nitrogen) to the soil. Nitrogen is one of "the big three," being the "N" in NPK, the three letters that form a virtual stamp of approval for a complete fertilizer.
Why is nitrogen important in fertilizer?
The Importance of Nitrogen in Fertilization. Landscapers, gardeners , and farmers value nitrogen-fixing plants for their ability to contribute an essential plant nutrient (namely, nitrogen) to the soil. Nitrogen is one of "the big three," being the "N" in NPK, the three letters that form a virtual stamp of approval for a complete fertilizer.
What is the name of the process that plants use to fix nitrogen?
It is an example of a symbiotic relationship (between plant and bacteria), and the name for the process is "nitrogen fixation.".
Which family of plants are nitrogen-fixing?
Plants in the legume family are known to be nitrogen-fixing. Rodale's Organic Life, in discussing the importance of this element to plant growth, observes that "when leaves contain sufficient nitrogen, photosynthesis occurs at high rates.
Is a nitrogen fixer invasive?
That is, not every plant listed is a desirable plant to grow, even though it meets the definition, scientifically, of a nitrogen-fixing plant. Some nitrogen fixers are listed as invasive plants in North America; such cases are indicated in brackets:
What bacteria are in soybean nodules?
The specific and compatible rhizobia nodulating soybean is B. japonicum ( Cooper, 2007; Long, 1989; Rolfe, 1988 ). Soybean association with rhizobia , including B. japonicum and B. elkanii, provide about 50–60% of soybean nitrogen requirement supplied by the bacteria in nodules ( Salvagiotti et al., 2008 ). Rhizobia are the bacteria, which include Rhizobium, Bradyrhizobium, Sinorhizobium, etc., surviving and reproducing in the soil, and fixing atmospheric N inside the nodules produced in the roots of their specific legume (reviewed by Denison and Kiers, 2004 ).
What is the function of nodules on soybean roots?
(2015). The main function of nodules on soybean roots is to fix the atmospheric N by the process of symbiotic nitrogen fixation, supplying nitrogen for plant growth and seed production. Sugiyama et al. (2015) reported changes in the rhizospheric bacteria and especially Bradyrhizobium during soybean growth, suggesting that the symbiosis of host plant with rhizobia may be selective.
What are the bacteria that reproduce in the soil?
Rhizobia are the bacteria, which include Rhizobium, Bradyrhizobium, Sinorhizobium, etc., surviving and reproducing in the soil, and fixing atmospheric N inside the nodules produced in the roots of their specific legume (reviewed by Denison and Kiers, 2004 ).
Where are cbb3 oxidases found?
Cytochrome cbb3 oxidases were first identified in the nitrogen-fixing bacterium Bradyrhizobium japonicum, but have since been found in other environmental bacteria which can grow in microaerobic environments such as Paracoccus denitrificans and the phototroph Rhodobacter sphaeroides. They have also been found in the pathogens Campylobacter jejuni, Helicobacter pylori, Neisseria meningitidis and in Pseudomonas spp. Where measured these oxidases have been shown to have a high affinity for oxygen.
What are the symbiotic relationships between nitrogen fixing bacteria and plant roots?
There are many different symbiotic associations between nitrogen fixing bacteria and plant roots. The most significant of these for agriculture is the Fabaceae– Rhizobium spp./ Bradyrhizobium sp. root nodule symbioses. The bacteria persist in a dormant or saprophytic state in the soil before infecting a suitable root via the root hair. Legume roots exude various flavonoid and isoflavonoid molecules that induce expression of nod (nodulation) genes by such rhizobial bacteria. This results in the formation by the bacterium of lipo-oligosaccharide Nod factors, the precise structure of which determines the host range and specificity of Rhizobium spp.
What does a host plant provide to a rhizobial bacteria?
The host plant provides the bacteria with carbohydrates. In return, rhizobial bacteria fix nitrogen from the atmosphere into NH 4+, via nitrogenase. The NH 4+ is converted into amides or ureides, which are passed to the plant xylem.
What is a rhizobia?
Rhizobia are nitrogen-fixing bacteria classified and characterized by different systems. Beijerinck was able to isolate and cultivate a microorganism, named Bacillus radiocicola, from the nodules of legumes in 1888. However, Frank (1889) renamed it Rhizobium leguminosarum ( Fred et al., 1932 ), which was retained in Bergey’s Manual of Determinative Bacteriology ( Holt et al., 1994 ).
How Do Plants Fix Nitrogen?
Nitrogen fixing plants don’t pull nitrogen from the air on their own. They actually need help from a common bacteria called Rhizobium . The bacteria infects legume plants such as peas and beans and uses the plant to help it draw nitrogen from the air. The bacteria converts this nitrogen gas and stores it in the roots of the plant.
Why do plants grow greener?
Your garden will grow greener and more lush thanks to plants that fix nitrogen and their beneficial symbiotic relationship with bacteria.
What happens when legumes die?
When legumes and other nitrogen fixing plants and the bacteria work together to store the nitrogen, they are creating a green warehouse in your garden. While they are growing, they release very little nitrogen into the soil, but when they are done growing and they die, their decomposition releases the stored nitrogen and increases ...
What is the nitrogen nodule on a plant?
The bacteria converts this nitrogen gas and stores it in the roots of the plant. When the plant stores the nitrogen in the roots, it produces a lump on the root called a nitrogen nodule. This is harmless to the plant but very ...
How to get more nitrogen in your garden?
Try planting a winter cover crop of legumes, such as clover or winter peas. In the spring, you can simply till under the plants into your garden beds. As these plants decompose, they will raise the total nitrogen in the soil and will make nitrogen available for plants that are unable to get nitrogen from the air.
Why is nitrogen important for plants?
Nitrogen for plants is vital to the success of a garden. Without sufficient nitrogen, plants will fail and be unable to grow. Nitrogen is abundant in the world, but most of the nitrogen in the world is a gas and many plants cannot use nitrogen as a gas. Most plants must rely on the addition of nitrogen to the soil in order to be able to use it.

Overview
Nitrogen fixation is a chemical process by which molecular nitrogen (N 2), with a strong triple covalent bond, in the air is converted into ammonia (NH 3) or related nitrogenous compounds, typically in soil or aquatic systems but also in industry. Atmospheric nitrogen is molecular dinitrogen, a relatively nonreactive molecule that is metabolically useless to all but a few microorganisms. Biological nitrogen fixation or diazotroph is an important microbials mediated …
History
Biological nitrogen fixation was discovered by Jean-Baptiste Boussingault in 1838. Later, in 1880, the process by which it happens was discovered by German agronomist Hermann Hellriegel and Hermann Wilfarth [de] and was fully described by Dutch microbiologist Martinus Beijerinck.
"The protracted investigations of the relation of plants to the acquisition of nitr…
Biological
Biological nitrogen fixation (BNF) occurs when atmospheric nitrogen is converted to ammonia by a nitrogenase enzyme. The overall reaction for BNF is:
The process is coupled to the hydrolysis of 16 equivalents of ATP and is accompanied by the co-formation of one equivalent of H 2. The conversion of N 2 into ammonia occurs at a metal cluster called FeMoco, an abbreviation for th…
Industrial processes
A method for nitrogen fixation was first described by Henry Cavendish in 1784 using electric arcs reacting nitrogen and oxygen in air. This method was implemented in the Birkeland–Eyde process of 1903. The fixation of nitrogen by lightning is a very similar natural occurring process.
The possibility that atmospheric nitrogen reacts with certain chemicals was fir…
Lightning
Nitrogen can be fixed by lightning converting nitrogen gas (N 2) and oxygen gas (O 2) in the atmosphere into NOx (nitrogen oxides). The N 2 molecule is highly stable and nonreactive due to the triple bond between the nitrogen atoms. Lightning produces enough energy and heat to break this bond allowing nitrogen atoms to react with oxygen, forming NO x. These compounds cannot be used …
See also
• Birkeland–Eyde process: an industrial fertilizer production process
• George Washington Carver: an American botanist
• Denitrification: an organic process of nitrogen release
• Heterocyst
External links
• Hirsch AM (2009). "A Brief History of the Discovery of Nitrogen-fixing Organisms" (PDF). University of California, Los Angeles.
• "Marine Nitrogen Fixation laboratory". University of Southern California.
• "Travis P. Hignett Collection of Fixed Nitrogen Research Laboratory Photographs // Science History Institute Digital Collections". digital.sciencehistory.org. Retrieved 16 August 2019. Science History Institute Digital Collections (Photographs depicting numerous stages of …
• Hirsch AM (2009). "A Brief History of the Discovery of Nitrogen-fixing Organisms" (PDF). University of California, Los Angeles.
• "Marine Nitrogen Fixation laboratory". University of Southern California.
• "Travis P. Hignett Collection of Fixed Nitrogen Research Laboratory Photographs // Science History Institute Digital Collections". digital.sciencehistory.org. Retrieved 16 August 2019. Science History Institute Digital Collections (Photographs depicting numerous stages of the nitrogen fixa…