
A reverse phase column, or reversed-phase HPLC
High-performance liquid chromatography
High-performance liquid chromatography (HPLC; formerly referred to as high-pressure liquid chromatography), is a technique in analytical chemistry used to separate, identify, and quantify each component in a mixture. It relies on pumps to pass a pressurized liquid solvent containing the …
What is reverse phase?
Reverse phase chromatography is the most commonly used LC or HPLC separation mode.It is used to separate nonpolar molecules in solution.In reverse phase the stationary phase is nonpolar and the mobile phase is polar.The name “reversed phase” is derived from the opposite technique of “normal phase” chromatography which involves the separation of molecules based upon their interaction ...
Why is reverse phase chromatography so called?
Reversed high performance liquid chromatography (HPLC) has this name because the order of the process is, as you might expect, reversed. Whereas in normal HPLC the non-polar parts of a substance are separated at the stationary phase, thus eluting the polar ones afterwards, in reverse HPLC the polar ones are subtracted first.
What is normal phase column chromatography?
Normal-phase liquid chromatography (NPLC) is a technique that uses columns packed with polar stationaryphases combined with nonpolar or moderately-polar mobile phases to separate the components of mixtures. The rate at which individual solutes migrate through NPLC columns is primarily a function of their polarity.
How to perform column chromatography?
To load the column:
- Dissolve the sample in the minimum amount of solvent (5–10 drops). ...
- Using a pipette or syringe with a thick needle, drip the sample directly onto the top of the silica. ...
- Once the entire sample has been added, allow the column to drain so that the solvent level touches the top of the silica.
- Carefully add a layer of sand (approx. ...

What do you mean by reverse phase chromatography?
Reversed-phase chromatography is the term given to chromatographic conditions in which a nonpolar stationary phase is used in conjunction with a polar mobile phase.
What is reverse phase and normal phase chromatography?
Normal phase chromatography, an adsorptive mechanism, is used for the analysis of solutes readily soluble in organic solvents, based on their polar differences such as amines, acids, metal complexes, etc.. Reversed-phase chromatography, a partition mechanism, is typically used for separations by non-polar differences.
Why reverse phase column is used in HPLC?
What are the Advantages of Reverse HPLC? By using water (or a water-based substance) as the solvent, reversed HPLC eliminates the danger of the analyte retention times being skewed due to absorption of water in the atmosphere.
What is meant by reverse phase HPLC?
Reverse-phase HPLC involves binding an organic molecule to a stationary phase, often silica derivatized with alkyl chains, in a relatively polar environment (the mobile phase), which could contain water, and then eluting the organic molecule using a gradient of a less polar organic solvent.
What is C8 and C18 column?
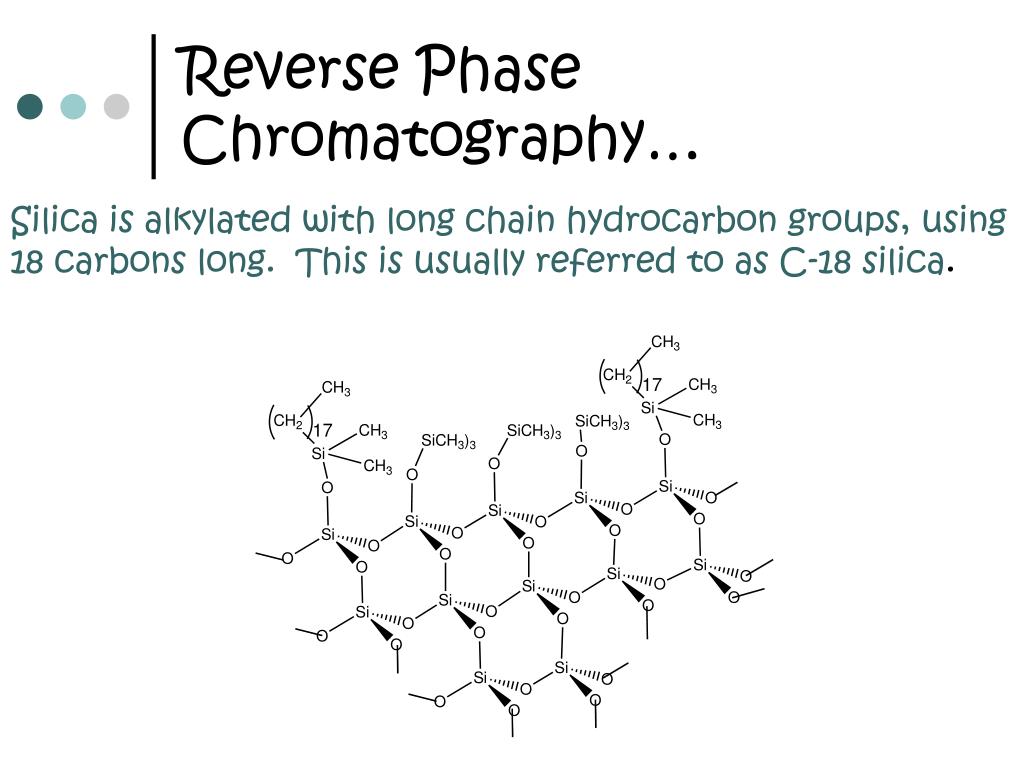
Definition. C8 column refers to a type of column used in the reverse-phase chromatography, containing octylsilane as its stationary phase, while the C18 column refers to another type of column used in the reverse-phase chromatography, containing octadecylsilance as the stationary phase.
Why reverse phase is mostly used?
Reversed-phase chromatography provides better solubility for polar analytes, uses nontoxic solvents, offers a method for removal of contaminants and mobile phase additives, and gives timely sample recovery with little solvent evaporation.
What is polar and nonpolar in HPLC?
Reverse Phase HPLC The stationary phase is nonpolar, like C18 bonded silica. The mobile phase is polar, usually being water and polar organic solvent. Compounds with the most hydrophobicity elute later in the chromatogram and those with the least hydrophobicity elute earlier.
Why C18 column is non polar?
There is only one reason why C18 is unpolar: it contains very long hydrocarbon chains (octadecyl, so 18 carbon atoms). These chains are very unpolar, so the column itself is unpolar.
Why C8 and C18 column is used in HPLC?
When a short retention time is required, C8 is more preferred. Its lower hydrophobicity will cause faster retention of nonpolar compounds. Therefore, nonpolar compounds will move down the column more rapidly with C8 than with C18.
Why we use C18 column?
C18 columns are HPLC (high performance liquid chromatography) columns that use a C18 substance as the stationary phase. C18 HPLC columns are used in environmental sciences and chemical analysis, as well as industries such as pharmaceutical and environmental sciences, to analyze individual parts of chemical mixtures.
Are all C18 columns reverse phase?
A C18 column is an example of a "reverse phase" column. Reverse phase columns are often used with more polar solvents such as water, methanol or acetonitrile. The stationary phase is a nonpolar hydrocarbon, whereas the mobile phase is a polar liquid. The same approach can also be used in TLC.
Is silica polar or nonpolar?
polarSilica gel is simply very finely ground very pure sand. It should be noted that silica gel is highly polar and is capable of hydrogen bonding.
What is the difference between Hilic and normal phase?
HILIC is a variation of normal phase chromatography. The major differences between HILIC and NP are the composite of the mobile phase and the mechanism of separation. Common NP chromatography uses 100 % organic mobile phases while HILIC uses organic mobile phases that are water miscible.
What is meant by normal phase?
Definition: A separation method where the components are distributed between two phases, one of which is stationary and polar, while the other is non-polar and moves in a definite direction.
What is the difference between a reverse phase run and a normal phase run in HPLC?
The key difference between reverse phase and normal phase HPLC is that the reverse phase HPLC uses a nonpolar stationary phase and a polar mobile phase whereas the normal phase HPLC uses a polar stationary phase and a less polar mobile phase.
What is reverse and forward phase?
Reverse phase dimming usually gives you better overall control and smooth dimming down to 5% light levels, while forward phase normally dims smoothly down to 10%. Generally works better with LED light sources and all electronic low-voltage systems.
What is the purpose of adding acetonitrile to the elution buffer?
Instead of using salt gradients to elute hydrophobic species, organic modifiers such as acetonitrile or propanol are added to the elution buffer to decrease the water concentration in the mobile phase. This in turn weakens the hydrophobic attraction of the hydrophobic groups on the chromatography matrix for the protein.
What is HP-RPC in RPC?
HP-RPC separates compounds according to their relative nonpolarity or hydrophobicity. In RPC, the polarity of the stationary and mobile phase is to the reverse of that used in NPC. HP-RPC is performed on porous or nonporous stationary phases with immobilized nonpolar polymers (i.e., n -alkylsilicas) or nonpolymer polymers (i.e., microparticulate polystyrenes). The most commonly accepted retention mechanism in RPC is based on the solvophobic theory, which describes the hydrophobic interaction between the nonpolar surface regions of the analytes and the nonpolar ligands/surfaces of the stationary phase. 15,16
What is surface initiated radical polymerization?
Surface-initiated atom transfer radical polymerization (ATRP) is an attractive method as it allows the preparation of dense PIPAAm brushes on the surface [3]. The grafted amounts of PIPAAm on the silica-bead surface exceed that of the PIPAAm hydrogel-modified silica bead prepared by conventional radical polymerization (type D) by nearly one order of magnitude because the surface-initiated ATRP forms dense and thick PIPAAm brushes on the surface. In temperature-dependent chromatographic analyses, dense PIPAAm brush-grafted silica beads resulted in relatively longer steroid retention times than those prepared by conventional radical polymerization methods. This result suggests that densely grafted PIPAAm brushes produce strong hydrophobic interactions with steroids. Furthermore, surface-initiated ATRP facilitates variation of the density and/or molecular weight of PIPAAm brushes prepared by changing the density of ATRP initiator and ATRP polymerization time.
What is the mechanism of protein separation?
Separation of proteins by reversed-phase chromatography (RPC) depends on the reversible adsorption/desorption of the proteins, which have varying degrees of hydrophobicity, to a hydrophobic stationary matrix. The nature of the hydrophobic binding interaction is thought to be the result of a favorable entropy effect in areas adjacent to the hydrophobic regions where there is a higher degree of organized water structure, although the actual mechanism has yet to be fully elucidated. The matrices are generally silica-based or synthetic organic polymers and contain covalently bound alkyl chains of different lengths such as n -octadecyl (C18), n -butyl (C4), or phenyl groups. The strength of the hydrophobic interaction increases as the alkyl chain size increases.
Why do hydrophobic forces increase entropy?
The hydrophobic forces are due to structuring of water around the hydrophobic residue resulting in a decrease in total system entropy. In order to maximize the system entropy, the hydrophobic residues orient away from the water resulting in an increase in bulk water entropy and hence an increase in system entropy.
Why do you dilution detergents?
Dilution of contaminating detergents may be required to prevent interference with protein binding to the sorbent. Desalting may require the resin to be washed, for example with 5% methanol, 0.1% TFA, prior to elution while selective elution may be achieved by experimenting with alternative ion-pairing agents.
What is reverse phase chromatography?
Reversed phase (RP) chromatography is a popular technique for the separation, desalting, and concentration of proteins, in part because the sample is concentrated in a small volume of volatile solvent that can be removed by evaporation.
What is HIC chromatography?
It is regarded as a type of reversed-phase chromatography and is based on the separation of molecules as per their hydrophobicity. HIC acquires an advantage of the hydrophobic collaboration of large molecules with a moderately hydrophobic stationary phase. For example: butyl-bonded [C4], rather than octadecyl-bonded [C18], silica.
What is normal phase chromatography?
Normal phase chromatography is established on the partition equilibrium between the polar stationary phase and the non-polar mobile phase.
What is the difference between a stationary and a mobile phase?
The stationary phase is acidic or basic having a negative or positive charge whereas the mobile phase is constituted by a polar liquid such as the salt solution in water. The separation occurs because of the attractive ionic force between molecules and the charged stationary phase.
What is HPLC used for?
HPLC discovers its application in numerous fields such as foods and beverages, forensics, clinical research, environmental laboratories, and most importantly in the pharmaceutical sector.
What is the basis of IEC?
The basis of IEC is the electrostatic interaction between the ion exchanger and the ionic solutes.
Why does it take longer to elute a sample?
If the charge on the sample is stronger, the stronger it is attracted to the ionic surface and hence, it takes longer to elute.
What are some examples of mobile phase?
Examples of mobile phase are blends of water or buffer (aqueous solvents) and organic solvents such methanol, acetonitrile, or tetrahydrofuran ( THF).
What is RPLC used for?
RPLC utilizes a nonpolar stationary phase and a polar mobile phase. In the past several decades, RPLC with a C18 stationary phase has been widely used in peptides separation because of the relatively high hydrophobicity of peptides ( Zhang et al., 2013 ). Meanwhile, large-scale glycopeptide separations have relied almost exclusively on C18-RPLC ( Anonsen, Vik, Egge-Jacobsen, & Koomey, 2012; Halim, Nilsson, Ruetschi, Hesse, & Larson, 2012; Halim, Ruetschi, Larson, & Nilsson, 2013; Yin et al., 2013 ). Since RPLC is the standard separation system employed by proteomics laboratories, its compatibility with other lines of proteomic research permits the wide use of RPLC in glycopeptide separation ( Thaysen-Andersen & Packer, 2014 ). The retention of glycopeptides in RPLC is directly related to the hydrophobicity of the molecules. In a glycopeptide molecule, the glycan moiety contributes hydrophilicity, while the peptide backbone contributes hydrophobicity. Therefore, the hydrophobicity of a glycopeptide is generally lower than its peptide backbone. As a result, those hydrophilic glycopeptides containing relatively larger glycan moieties and smaller peptide moieties might not be retained by C18. Moreover, the hydrophobicity variation of glycan isomers is very limited. Therefore, RPLC offers poor glycan isomer differentiation ( Hua et al., 2012 ). This limits the use of RPLC in the glycoproteomics analysis.
What is RPC in chemistry?
RPC has become the predominant branch of analytical chromatography (HPLC) in the life sciences and in biotechnology. Preparative applications of RPC are the exception, although some have been reported. Most commonly, bonded high performance stationary phases prepared by covalently binding hydrophobic ligands such as C4 -, C 8 -, and C 18-alkyl chains or aromatic functions to the surface of a rigid siliceous or polymeric support are used. Due to the pronounced hydrophobic character of the stationary phase, proteins and peptides bind tightly from a neat aqueous mobile phase and require hydro-organic eluents for release. The separation of peptides and proteins in RPC is therefore typically carried out by gradient elution with increasing concentration of an organic modifier such as acetonitrile, methanol, tetrahydrofuran, and isopropanol. In addition, the mobile phase usually contains low levels of trifluoroacetic or phosphoric acid. The role of the acids is to protonate the residual silanol groups at the surface of the siliceous support and the carboxyl groups of the eluates as well as to form ion pairs with the charged amino groups of the substances to be separated. Ion-pairing agents such as perchlorate can be used at neutral pH.
What software is used to do a chromatography separation?
The most important of them are DryLab, 1 PREOPT-W, 2 OSIRIS, 3 MICHROM, 4 and ChromSword. 5 However, in most laboratories where RPLC is used daily, computer-assisted separation optimization is not used to improve separations. The lack of popularity of readily available multivariate optimization software may due to the fact that many chromatographers are uncomfortable with software tools, which are still rather expensive and not very user-friendly.
What is reverse phase chromatography?
Reversed-phase chromatography is the most common HPLC separation technique and is used for separating compounds that have hydrophobic moieties and do not have a dominant polar character (although polarity of a compound does not exclude the use of RP-HPLC).
What is the most widely used method for purification and analysis of peptides?
Reversed-phase chromatography has become the most widely-used method for purification and analysis of peptides owing to its powerful resolving capability, reproducibility and recovery [1,2] even at ultramicroanalytical levels. The purification factor achievable with reversed-phase chromatography ranges from 2 to 200. In comparison the purification factor of size exclusion ranges from 2 to 20, of ion exchange chromatography from 2 to 40 and of hydrophobic interaction chromatography from 2 to 30 [3 ]. The major advantage of reversed-phase chromatography, apart from its resolving capability, is the availability and use of volatile mobile phases (e.g. aqueous trifluoracetic acid–acetonitrile systems) which do away with the need for sample desalting.
What is RPC in preparative work?
The use of RPC in preparative work commonly requires the refolding of the product into its native configuration after separation. This has been shown to be possible for a number of peptides and smaller proteins, which are of interest to the pharmaceutical industry such as h-insulin and h-growth hormone (hGH).
How does double bonding affect retention?
In NARP-LC, the elution order is based on increasing PN, thus components will elute in ascending order of chain length, while a double bond in any of the FA chain lengths reduces the retention time, roughly by the equivalent of two carbon atoms. The number and position of double bonds also affect retention, as well as their chain length. As a consequence, TAGs with identical equivalent carbon number tend to coelute, and are thus called critical pairs. However, under optimized conditions, by employing efficient stationary phases, nowadays commercially available, it is possible to separate a great number of TAGs with same PN. The separation of compounds with the same PN group is feasible, such as OOO, OOP, OPP, and PPP (O: oleic acid, P: palmitic acid), as well as the critical pair LLL/OLL n (L n: linolenic acid), having the same PN, CN, and DBN. Also TAGs differing in the position (s) of double bonds have been resolved, such as LnLnγLn–LnLnLn, which have the same PN, CN, and DBN but differently located along the same hydrocarbon chain, i.e., Δ6 or Δ9 [129].
What is the most commonly used material for reversed phase separation of biomolecules?
Alkyl bonded silica phases are the most commonly used materials for the reversed-phase separation of biomolecules. The shorter C4 matrices are generally recommended for large hydrophobic peptides and most proteins.
What is mobile phase?
The mobile phase is generally a binary mixture of water and a miscible polar organic solvent. Typical examples are methanol, acetonitrile or THF. Retention increases as the amount of the polar solvent (water) in the mobile phase increases.. A comparison list of the most important RP materials can requested through Contact Form.
What is the most common stationary phase?
Silica based Octadecyl (C18) material is the most common stationary phase. Octyl (C8) and butyl (C4) are popular but not the best choice in biochromatography. The designations for the reversed phase materials refers to the length of the hydrocarbon chain. . The range covers anything from the very short length C1 to very large C 30 alkyl chain ...
Why is C18 polar?
Traditional C18 (ODS) phases are hydrophobic and have a high polarity due to the lower purity silicas on which they are based. Use of the new high purity silicas reduces silanol activity and improves reproducibility.
Where are shorter alkyl chains found?
Shorter alkyl chain phases are found at the lower hydrophobicity area of the graph. Alternative bonded phases (including phenyl and cyano) based on high purity silicas are best considered to effect changes in polarity.
Is the stationary phase polar or nonpolar?
In Reverse Phase Liquid Chromatography the stationary phase is non-polar and the mobile phase is polar. With Silica based materials the non-polar surface is altered by means of attaching silanes with a alkyl hydrocarbons tethers.
What solvent is used to dissolve a standard molecule?
Assuming it has a UV spectrum (not all molecules do!) dissolve the standard (target molecule) in an organic solvent like Methanol, Acetonitrile, Isopropyl Alcohol. Assuming this is reverse phase chromatography (C18 column) inject 10 uL into the HPLC and note the retention time (RT). The main peak should elute at the injection front.
How to sharpen a peak?
Sharpen the peak by adjusting the pH of the mobile phase to >pKa of the sidegroups of the target molecule. These sidegroups are usually amino or carboxyl groups and may be protonated (thus pH control). By manipulating the pH you assure yourself you only have 1 form (protonated versus anion).
What pH range does a C18 column need?
Note; that a REGULAR C18 column can only tolerate a pH range of 2-7. Going above 7 ensures that the column WILL be destroyed by cleaving the silanol groups on the stationary phase.
How long does it take for a RT to elute?
Optimize the RT so it elutes at 5-10 minutes for a 5 cm column (at 1.0 mL/minute flow) OR 10-15 minutes for a 25 cm column (at 0.5 mL/minute flow).
Is there an analytical paper for nucleotides and beta cyclodextrins?
In your case this may also work out, but there are analytical papers for Nucleotides and beta cyclodextrins around, I'm sure.
Can organic compounds be purified by a silica column?
There are many organic compounds which cannot be easily purified by normal silica column chromatography. These compounds need to be purified by reverse phase column chromatography however most of the scientists do not know the standard procedure for Reverse Phase column chromatography.
Is 5% methanol saturated with ammonia?
I've used 5% methanol saturated with ammonia in dichloromethane for very polar alkaloids but above 5% methanol and you start to get silica coming through. Also ammonia is gross.
What is reverse phase HPLC?
Reverse Phase HPLC: The reverse phase chromatography works on the principle of hydrophobic interactions so the more nonpolar the analyte has, the longer it will be retained. It this mobile phase is polar and the stationary phase is nonpolar in nature.
What are the advantages of HPLC?
The Advantages of HPLC are as follows. 1 The high-performance liquid chromatography provides a simple, automated, and highly accurate method of identifying certain chemical components in a sample. 2 Provides a quantitative and qualitative analysis that is simple and accurate. 3 It can be upgrading to mass spectroscopy. 4 Compared to other chromatographic techniques such as column chromatography, TLC, and paper chromatography, HPLC is fast, effective and delivers high resolution. 5 The gradient elution is readily adaptable in HPLC.
What is HPLC based on?
The HPLC principle is based on the distribution of the component between a stationary phase ( HPLC column) and a mobile phase ( solvent ). Depending on the chemical structure of the molecules they are retarded as passing the stationary phase. The intermolecular interactions among ...
What is size exclusion in HPLC?
Size-Exclusion HPLC: Size Exclusion Chromatography ( SEC) is a chromatographic process that separates molecules based solely on their size , in this technique molecules are separated by the column packing material on the basis of their exclusion from por es.
What is ion exchange chromatography?
Ion exchange chromatography is the most popular method for the purification of proteins and other charged molecules.
What happens to the retention time of analytes when the pH of the mobile phase changes?
The retention time of the analyte: When the pH of the mobile phase changes a dramatic change occurs in the retention time of acidic and basic analytes. When the ionization of these molecules is changed it is often the result of different interactions amongst the column and the molecules.
How does pH affect HPLC?
The pH of the mobile phase in reversed-phase HPLC is an important factor, it can affect the peak shape as well as the retention time of the molecule as it affects the ionization state of the molecule , and therefore the chemistry of the interactions in the HPLC column.
Reversed Phase Liquid Chromatography
Elution in Reverse Phase Liquid Chromatography
- The partition-mechanism in Reverse Phase Liquid Chromatography, is typically used for separations by non-polar differences In Reversed Phase Liquid Chromatography the most polar compounds elute first with the most non-polar compounds eluting last. The mobile phase is generally a binary mixture of water and a miscible polar organic solvent. Typical examples are m…
Characteristics in Reversed Phase Liquid Chromatography
- Hydrophobicity The strength of hydrophobic interaction can be measured by the retention of neutral (non-polar) molecules. Percentage of carbon in the material is also a simplistic but useful guide to the retention characteristics of the column. In Figure 1 the loose correlation is demonstrated. An increase of retention is observed when alkyl chain length (i.e. carbon load) is i…
High Purity Base Deactivated Phases
- About the year 2000 new alkyl bonded silicas have been introduced. The cumulative metal ion impurity level within these base silicas has been reduced to <10ppm. As a result the number of isolated silanol groups and hence the polarity of the silica surface is also reduced. When coupled with the use of more effective and reproducible bonding processes, a new generation of reverse…
Hydrophobicity vs. Polarity in Reverse Phase Liquid Chromatography A Comparison
- Traditional C18 (ODS) phases are hydrophobic and have a high polarity due to the lower purity silicas on which they are based. Use of the new high purity silicas reduces silanol activity and improves reproducibility. Employing a polar embedded functionality may also result in a reduced polarity material. Shorter alkyl chain phases are found at the lower hydrophobicity area of the gr…
Wide Pore (300Å) Reversed-Phase Materials
- Introduction Indeed, for a sample molecule to freely access the interior of the pores of the packing material, its diameter must be smaller than the average pore diameter. For high molecular weight solutes, the use of lower pore size materials of 60-120Å may result in frictional drag within the pore leading to restricted diffusion and reduced column efficiency. The use of larger pore silica-…
Bonded Phases
- Alkyl bonded silica phases are the most commonly used materials for the reversed-phase separation of biomolecules. The shorter C4 matrices are generally recommended for large hydrophobic peptides and most proteins. Peptide maps, natural and synthetic peptides and small hydrophilic proteins are best chromatographed on C8 columns. C18 columns are often chosen f…
Column Dimensions
- Wide pore silica phases are available in a range of column dimensions from rapid analysis to preparative and process scale. Increased column capacity favours these wide pore materials for preparative separations of samples with molecular weight >5000 Da.