What is the first group on the periodic table?
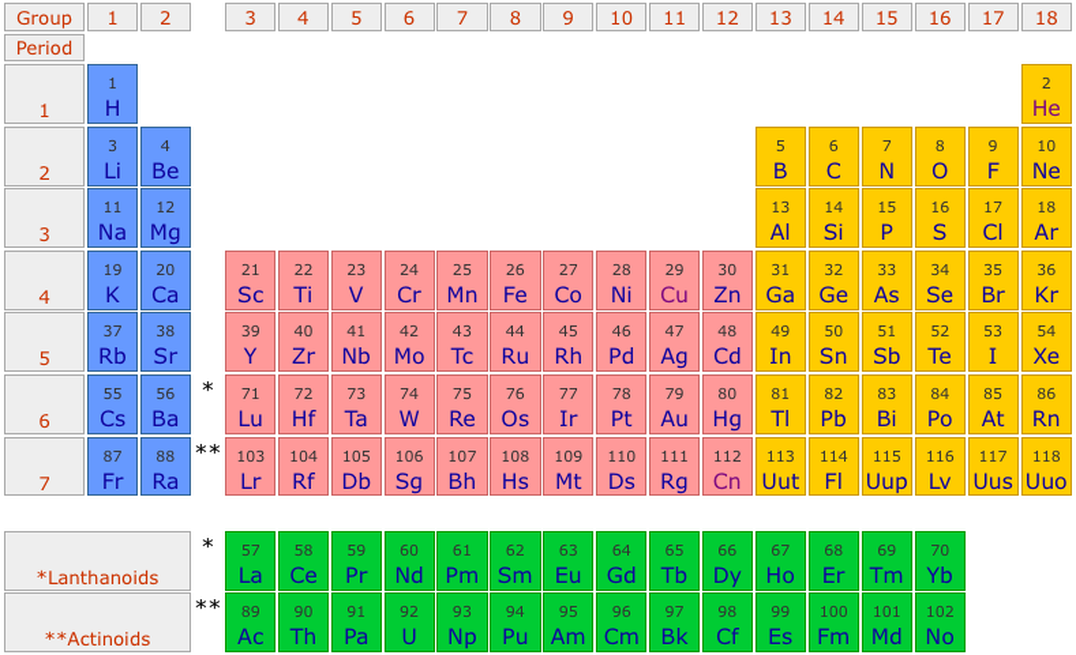
Alkali metals group is the very first group (group 1) on the periodic table. For detailed information on Alkali metals, read the Ultimate guide on Alkali metals of periodic table.
What is the periodic table?
Explanation of the periodic table. Encyclopædia Britannica, Inc. See all videos for this article. Periodic table, in full periodic table of the elements, in chemistry, the organized array of all the chemical elements in order of increasing atomic number —i.e., the total number of protons in the atomic nucleus.
Which group is the last group on the periodic table?
Noble gases group is the last group (group 18) on the periodic table. The elements included in Noble gases group are;
What group are the elements in the bottom row of periodic table?
The elements in the two bottom rows of the periodic table are also included in these groups. They are placed in the two separate rows at the bottom because they show few different properties. Actually, the elements in the bottom rows are the extension of group 3 only. So they are included in group 3.
What is 1s 2s 2p 3s 3p?
1s 2s 2p 3s 3p represents the electron orbital energy levels.
What is s in periodic?
Sulfur - Element information, properties and uses | Periodic Table.
What period is s in the periodic table?
Sulfur is the 16th element on the periodic table. It is located in period 3 and group 16. It is directly below oxygen (O) and between phosphorus (P) and chlorine (Cl).
What is the s sublevel on the periodic table?
Since all of the valence electrons in Group 1A and Group 2A elements exist in s orbitals, the first two columns of the Periodic Table (columns 1A and 2A) are known as the "s sublevel block".
Why is it called the s-block?
The s-block and p-block elements are so called because their valence electrons are in an s orbital or p orbital respectively. They are also called Typical Elements to distinguish them from the transition and inner transition series.
What does s-block stand for?
In chemistry, s-block refers to a group of elements on the periodic table that all have their valence electrons occupying the same electron orbital, the s-orbital. S-block elements include hydrogen and helium, and alkali metals and alkaline earth metals.
Is silicon a metal?
silicon (Si), a nonmetallic chemical element in the carbon family (Group 14 [IVa] of the periodic table).
Is sulfur a metal?
The non-metallic chemical element sulfur, 3216S , referred to in Genesis as brimstone and identified as element by Lavoisier, is the tenth most abundant element in the universe and the fifth most common element on Earth.
What type of element is sulfur?
nonmetallic chemical elementsulfur (S), also spelled sulphur, nonmetallic chemical element belonging to the oxygen group (Group 16 [VIa] of the periodic table), one of the most reactive of the elements. Pure sulfur is a tasteless, odourless, brittle solid that is pale yellow in colour, a poor conductor of electricity, and insoluble in water.
What is 1s and 2s in chemistry?
1s will be filled first, with the maximum of 2 electrons. 2s will be filled next, with the maximum of 2 electrons.
What is 1s 2s?
1s vs 2s Orbital 1s orbital is the closest orbital to the nucleus. 2s orbital is the second closest orbital to the nucleus. Energy Level. Energy of 1s orbital is lower than that of 2s orbital. 2s has comparatively higher energy.
How many s orbitals are there?
one orbitalThe s sublevel has just one orbital, so can contain 2 electrons max. The p sublevel has 3 orbitals, so can contain 6 electrons max. The d sublevel has 5 orbitals, so can contain 10 electrons max. And the 4 sublevel has 7 orbitals, so can contain 14 electrons max.
What does S stand for in organic chemistry?
A counterclockwise direction is an S (sinister, Latin for left) configuration. A clockwise direction is an R (rectus, Latin for right) configuration.
Is sulphur a metal?
The non-metallic chemical element sulfur, 3216S , referred to in Genesis as brimstone and identified as element by Lavoisier, is the tenth most abundant element in the universe and the fifth most common element on Earth.
What is the symbol of sulphur?
SSulfur / Symbol
What is the group number of sulfur?
Group 6A (or VIA) of the periodic table are the chalcogens: the nonmetals oxygen (O), sulfur (S), and selenium (Se), the metalloid tellurium (Te), and the metal polonium (Po).
What is the first group of elements in the periodic table?
Group 1: Alkali metals group. Alkali metals group is the very first group (group 1) on the periodic table. The elements included in the Alkali metals group are; Lithium (Li)
Why are the elements in the bottom two rows of the periodic table included in group 3?
The elements in the two bottom rows of the periodic table are also included in these groups. They are placed in the two separate rows at the bottom because they show few different properties. Actually, the elements in the bottom rows are the extension of group 3 only. So they are included in group 3. But as these elements have few different ...
How many groups are there in the periodic table?
Groups are the vertical columns on the periodic table. There are total 18 vertical columns on periodic table. Hence there are 18 groups. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.
What is the oxygen group on the periodic table?
Oxygen group is the group 16 on the periodic table.
Which group is alkaline earth metals?
Alkaline earth metals are the group 2 elements on the periodic table.
Can you find every detail of an interactive periodic table?
You can effortlessly find every single detail about the elements from this single Interactive Periodic table.
Do elements of the same group have the same number of valence electrons?
In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons.
What is the periodic table?
periodic table, in full periodic table of the elements, in chemistry, the organized array of all the chemical elements in order of increasing atomic number —i.e., the total number of protons in the atomic nucleus. When the chemical elements are thus arranged, there is a recurring pattern called the “periodic law” in their properties, ...
When the chemical elements are thus arranged, there is a recurring pattern called the periodic law?
When the chemical elements are thus arranged, there is a recurring pattern called the “periodic law” in their properties, in which elements in the same column (group) have similar properties. The initial discovery, which was made by Dmitry I. Mendeleyev in the mid-19th century, has been of inestimable value in the development of chemistry.
What are the rows of lanthanoid and actinoid?
These rows contain elements in the lanthanoid and actinoid series, usually from 57 to 71 ( lanthanum to lutetium) and 89 to 103 ( actinium to lawrencium ), respectively. There is no scientific reason for this. It is merely done to make the table more compact.
What is the atomic number of an element?
The atomic number of an element is the number of protons in the nucleus of an atom of that element . Hydrogen has 1 proton, and oganesson has ...
What elements are triads?
Döbereiner in 1817 showed that the combining weight, meaning atomic weight, of strontium lies midway between those of calcium and barium, and some years later he showed that other such “ triads ” exist (chlorine, bromine, and iodine [halogens] and lithium, sodium, and potassium [alkali metals]). J.-B.-A. Dumas, L. Gmelin, E. Lenssen, Max von Pettenkofer, and J.P. Cooke expanded Döbereiner’s suggestions between 1827 and 1858 by showing that similar relationships extended further than the triads of elements, fluorine being added to the halogens and magnesium to the alkaline-earth metals, while oxygen, sulfur, selenium, and tellurium were classed as one family and nitrogen, phosphorus, arsenic, antimony, and bismuth as another family of elements.
Why do the elements in the periodic table have different orbits?
The arrangement of the elements in the periodic table comes from the electronic configuration of the elements. Because of the Pauli exclusion principle, no more than two electrons can fill the same orbital. The first row of the periodic table consists of just two elements, hydrogen and helium. As atoms have more electrons, they have more orbits available to fill, and thus the rows contain more elements farther down in the table.
What are the elements that are related to the first seven?
Newlands proposed classifying the elements in the order of increasing atomic weights, the elements being assigned ordinal numbers from unity upward and divided into seven groups having properties closely related to the first seven of the elements then known: hydrogen, lithium, beryllium, boron, carbon, nitrogen, and oxygen . This relationship was termed the law of octaves, by analogy with the seven intervals of the musical scale.