
How to calculate solubility?
Method 2 Method 2 of 2: Calculating Solubility from the K sp
- Look up the product solubility constant (Ksp). This constant is different for each compound, so you'll need to look it up on a chart in your textbook.
- Write the chemical equation. First, determine how the compound splits apart into ions when it dissolves.
- Modify the equation to use variables. ...
- Account for common ions, if present. ...
- Solve the equation. ...
How to find solubility equation?
Write the chemical equation.
- For example, a molecule of PbI 2 splits into the ions Pb 2+, I -, and a second I -. ...
- Write the equation 7.1×10 –9 = [Pb 2+ ] [I -] 2
- The equation is the product solubility constant, which can be found for the 2 ions in a solubility chart. ...
How do you calculate the solubility of a substance?
How do you calculate the solubility of a solute? Solubility indicates the maximum amount of a substance that can be dissolved in a solvent at a given temperature. Such a solution is called saturated. Divide the mass of the compound by the mass of the solvent and then multiply by 100 g to calculate the solubility in g/100g .
What are the characteristics of solubility?
Solubility is the ability of a solid, liquid, or gaseous chemical substance (referred to as the solute) to dissolve in solvent (usually a liquid) and form a solution. The solubility of a substance fundamentally depends on the solvent used, as well as temperature and pressure. The solubility of a substance in a particular solvent is measured by ...
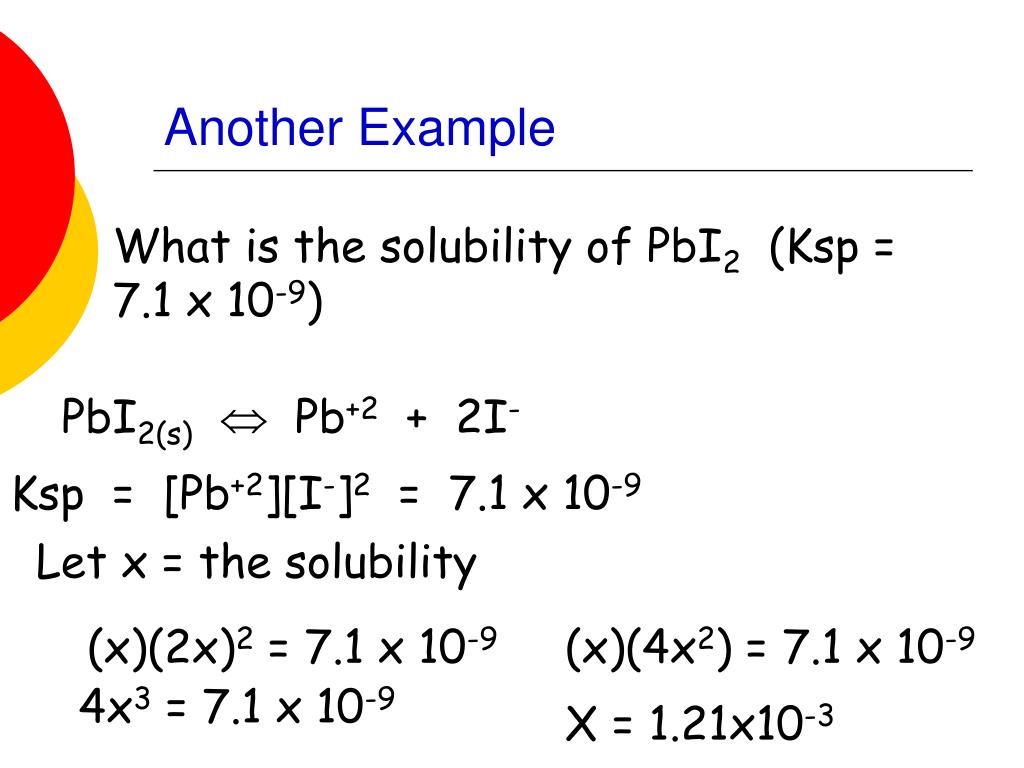
Which expression is used for solubility?
Answer and Explanation: The correct expression used for solubility is a. grams per 100 grams of solvent. Solubility is usually expressed in terms of the maximum amount of the solute that dissolves in the given amount of a solvent.
How do you write an expression for a solubility product?
0:431:41ALEKS: Writing a solubility product (Ksp) expression - YouTubeYouTubeStart of suggested clipEnd of suggested clipFor each one of the ions. And then number two like being able to recognize polyatomic ions gettingMoreFor each one of the ions. And then number two like being able to recognize polyatomic ions getting the stoichiometric coefficients corresponding with the subscripts. Correctly.
How do you express solubility of matter?
Solubility is often expressed as the mass of solute per volume (g/L) or mass of solute per mass of solvent (g/g), or as the moles of solute per volume (mol/L). Even for very soluble substances, however, there is usually a limit to how much solute can dissolve in a given quantity of solvent.
What is solubility expression in physical pharmaceutics?
As defined by the United States Pharmacopeia (USP), the solubility of drugs is expressed as the amount of solvent needed per unit of solute. Additionally, solubility is quantified by the molality, molarity, and percentage of the solvent.
What is the expression product?
In mathematics, a product is the result of multiplication, or an expression that identifies objects (numbers or variables) to be multiplied, called factors. For example, 30 is the product of 6 and 5 (the result of multiplication), and is the product of and.
What is the unit for solubility?
The unit of solubility is generally in mg/L (milligrams per liter) or ppm (parts per million).
What are the 3 types of solubility?
Based on the concentration of solute dissolves in a solvent, solutes are categorized into highly Soluble, sparingly Soluble or inSoluble.
What is the symbol for solubility?
The solubility product constant is the equilibrium constant for the dissolution of a solid substance into an aqueous solution. It is denoted by the symbol Ksp.
How do you write an expression as a product?
0:241:50Write as an Algebraic Expression: the product of 5 and x - YouTubeYouTubeStart of suggested clipEnd of suggested clipThis the product of 5 and x so the product of 5 and x well product that means multiplication. So we'MoreThis the product of 5 and x so the product of 5 and x well product that means multiplication. So we're multiplying 5 and x so to do that well we just write 5.
What is the expression for KSP of AgCl?
Silver chloride, AgCl, has a Ksp = 1.77 x 10¯10.
What is the expression for KSP of Ag2CrO4?
The Ksp of Ag2CrO4 is 1.1 × 10 12 at 298 K.
What is the KSP expression for caco3?
Ksp=[Ca2+][CO2−3] = 1.4×10−8 .
What is the solubility of a substance?
As a quantitative property, the solubility of a substance is determined by its concentration in a saturated solution at a particular temperature, and as a qualitative property, it is regarded as the inevitable interaction between molecules to produce a homogeneous molecular dispersion. Dissolution is an extrinsic property affecting a wide range of chemical, physical and crystallographic factors including complexation, particle size, surface properties, and modifications to the solid-state or formulation strategies that improve solubility. A compound's solubility depends on several factors, including moisture content, temperature, pressure, pH, and other chemical and physical properties.
How to measure solubility?
Solubility can be measured in a variety of different ways. As defined by the United States Pharmacopeia (USP), the solubility of drugs is expressed as the amount of solvent needed per unit of solute. Additionally, solubility is quantified by the molality, molarity, and percentage of the solvent. As shown in the Table, solubility is categorized into seven groups. Six categories are provided by the European Pharmacopoeia (rather than the practically insoluble division).
How do nonpolar solvents differ from polar solvents?
A nonpolar solvent like hydrocarbons differs from a polar solvent because of its molecular structure. Low dielectric constants of nonpolar solvents prevent them from reducing the attraction between ions in both strong and weak electrolytes. Since the solvents belong to the group of aprotic solvents, they cannot ionize weak electrolytes nor can they break covalent bonds. They cannot form hydrogen bridges with non-electrolytes. Nonpolar solvents don't dissolve ionic or polar solutes, or only slightly dissolve them. Through induced dipole interactions, nonpolar compounds in essence dissolve nonpolar compounds with similar pressures. Molecular repulsion forces stop the solute molecules from escaping into the solution. All these solvents can dissolve oils and fats, including carbon tetrachloride, benzene, and mineral oil. Nonpolar solvents are also suitable for dissolving alkaloidal bases and fatty acids.
How does water affect solubility?
As a liquid, water can dissolve alcohols, aldehydes, ketones, amines, and other nitrogen-and oxygen-containing compounds that can form hydrogen bonds with the liquid. Solubility is determined, as we have already mentioned, by the relative proportion of nonpolar to polar groups in a substance's molecules. In aliphatic alcohols, as a nonpolar chain's length increases, a compound's solubility in water decreases. More than four or five carbons in a monohydroxy alcohol, aldehyde, ketone, or acid cannot penetrate the hydrogen bonding structure of water. As polar groups are added to water-soluble molecules, such as in propylene glycol, glycerine, or tartaric acid, the solubility is greatly increased. A carbon chain divided allows the nonpolar effect to be reduced, which leads to increased water solubility. n-Butyl alcohol, however, dissolves in water at a rate of approximately 8 g/100 mL of water at 20°C, whereas tertiary butyl alcohol dissolves at any ratio.
How many liquids are needed to make a pharmaceutical solution?
A pharmaceutical solution typically takes two or more liquids to achieve its desired characteristics. Example - Various alcoholic solutions can be made by mixing water and alcohol; volatile oils can be placed in water to produce aromatic waters; alcohol and volatile oils can be combined to create spirits and elixirs; and several fixed oils are blended into lotions, sprays, and medicated oils. A liquid-liquid system can be divided into two categories, depending on their solubility with one another: complete miscibility and partial miscibility. Liquid-liquid systems fit into the miscible category when both components are soluble in each other.
How does solubility change with temperature?
In the system, the temperatures under which two components are soluble in all proportions will be below and above which two distinct layers will exist. The other type of trajectory involves other mixtures of liquids such as nicotine and water, with an equilibrium temperature in the lower and upper ranges as well as an intermediate-range in which the liquid is only partially miscible. The final type of solution does not have a critical solution temperature. For example, ethyl ether and water are insoluble at both an upper and a lower solution temperature. When the mixture exists at all temperatures, only a small portion of the mixture is soluble.
Does water dissolve polar substances?
Accordingly, water dissolves sugars and other polyhydroxy compounds in all proportions with alcohol but dipole moments alone are insufficient to explain why polar substances dissolve in water. An acidic or basic constituent also contributes to specific interactions in solutions from the Lewis electron donor-acceptor point of view. In contrast to the dipole moment, hydrogen bonds are a more important aspect of the solute than polarity.
What is Solubility?
The maximum amount of solute that can dissolve in a known quantity of solvent at a certain temperature is its solubility.
What is the solubility product of salt?
Solubility Product. The term solubility product is generally applicable for sparingly soluble salts. It is the maximum product of the molar concentration of the ions (raised to their appropriate powers) which are produced due to dissociation of the compound. At a given temperature the solubility product is constant.
What is the difference between a saturated and supersaturated solution?
A saturated solution is a solution where a given amount of solute is completely soluble in a solvent at a given temperature. On the other hand, a supersaturated solution is those where solute starts salting out or precipitate after a particular concentration is dissolved at the same temperature.
What is the maximum concentration of a solute that dissolves in a known concentration of solvent at a given?
In terms of quantity, solubility is the maximum concentration of solute that dissolves in a known concentration of solvent at a given temperature. Based on the concentration of solute dissolves in a solvent, solutes are categorized into highly soluble, sparingly soluble or insoluble .
How does solubility affect pH?
The solubility of the solute can affect the pH of an aqueous solution. If the solution’s pH is such that no net electrical charge is borne by a specific molecule, the solution also has minimal solubility and precipitates out of the solution.
What is the property of sugar molecules to dissolve?
The property which helps sugar molecules to dissolve is known as solubility . Hence, the term solubility can be defined as a property of a substance (solute) to dissolve in a given solvent. A solute is any substance which can be either solid or liquid or gas dissolved in a solvent.
Why does solubility increase with temperature?
The solubility increases with temperature for certain solids that are dissolved in liquid water. The rise in higher temperature kinetic energy helps the solvent molecules to break apart the solute molecules that are kept together by intermolecular attractions more effectively.
What is the solubility of a substance?
The word solubility is all about calculating the amount of solute dissolved in a given solvent. The simple concept of solubility of a substance is the molarity of the material under excessive undissolved material in a solution at chemical equilibrium.
What is the molar solubility of tin iodide?
The molar solubility of tin iodide SnI2 is 1.28 x 10-2 mol/L. Calculate the Ksp of this compound.
What is the significance of the solubility product?
Let us learn about the significance of the solubility product in detail. Whenever a salt is dissolved in a solvent, the strong forces of attraction of solute that are the lattice enthalpy of its ions need to be overcome by the interactions between the ions and the solvent. The solvation enthalpy of ions is always negative and this means ...
What are the parameters of solubility?
The solubility depends on a several numbers of parameters which includes the lattice enthalpy of salt and solvation enthalpy of ions in the solution that are of most importance. Let us learn about the significance of the solubility product in detail.
What is the enthalpy of ions?
The solvation enthalpy of ions is always negative and this means that energy is released during the process. The nature of the solvent finds the amount of energy that is released during the solvation which is solvation enthalpy.
What is the product constant of a saturated solution?
The solubility product constant is used to describe the saturated solutions of ionic compounds having relatively low solubility. A saturated solution is said to be in a state of dynamic equilibrium between the ionic compound and the undissolved solid.
What is the solubility product constant?
The solubility product constant is a simplified equilibrium constant denoted as Ksp which is defined for equilibrium between a solid and its respective ions in a given solution. Its value shows the degree to which a compound can dissociate in water. The greater the solubility product constant, the more soluble is the compound. The Ksp expression for a given salt is the product of the concentrations of the ions. Each concentration is raised to a power which is equal to the coefficient of that ion in a balanced equation to get the solubility equilibrium. The solubility product constants are used for describing the saturated solutions of ionic compounds of relatively low solubility. A saturated solution is in a dynamic equilibrium state between the dissolved and dissociated ionic compound and the undissolved solid. In this article, we will learn about what is the solubility product, what is Ksp, the solubility product definition, and some solubility product numerical.
What is the KSP of salt?
The Ksp expression for a given salt is the product of the concentrations of the ions. Each concentration is raised to a power which is equal to the coefficient of that ion in a balanced equation to get the solubility equilibrium. The solubility product constants are used for describing the saturated solutions of ionic compounds ...
What does KSP mean in chemistry?
Ksp in chemistry means the solubility product constant. The solubility product is an equilibrium constant whose value depends on the temperature. Ksp generally increases whenever there is an increase in the temperature due to the increased solubility. Hence, the Ksp meaning in chemistry refers to the solubility product constant.
Steps for Writing a Solubility Product (Ksp) Expression
Step 1: Read through the given information and note what ions the given salt will produce in solution.
Formulas and Definitions for Writing a Solubility Product (Ksp) Expression
Slightly Soluble Salt. Salts vary in their solubility in water. Some, such as {eq}NaCl {/eq}, are completely soluble; the solution consists entirely of their positive and negative ions without any of the undissociated salt.
Example Problem 1 - Writing a Solubility Product (Ksp) Expression
What is the solubility product expression for calcium phosphate, {eq}Ca_3 (PO_4)_2 {/eq}?
Example Problem 2 - Writing a Solubility Product (Ksp) Expression
What is the solubility product expression for barium sulfate, {eq}BaSO_4 {/eq}?
Why is KSP called the solubility product?
Ksp is called the solubility product because it is literally the product of the solubilities of the ions in moles per liter. The solubility product of a salt can therefore be calculated from its solubility, or vice versa. Photographic films are based on the sensitivity of AgBr to light.
What is the common mistake in solubility product calculations?
Another common mistake in solubility product calculations occurs when students are asked to write an equation that describes the relationship between the concentrations of the Ag + and S 2- ions in a saturated Ag 2 S solution. It is all too easy to look at the formula for this compound Ag 2 S and then write the following equation.
What does Cs mean in chemistry?
Remember that the symbol Cs in this equation stands for the solubility of Ag 2 S in moles per liter. Since we get two Ag + ions for each Ag 2 S formula unit that dissolves in water, the Ag + ion concentration at equilibrium is twice the solubility of the salt, or 2 Cs. We square the Ag + ion concentration term because the equilibrium constant expression for this reaction is proportional to the product of the concentrations of the three products of the reaction.
What is the KSP of salt?
Since this constant is proportional to the solubility of the salt, it is called the solubility product equilibrium constant for the reaction, or Ksp. Ksp = [Ag + ] [Cl -] The Ksp expression for a salt is the product of the concentrations of the ions, with each concentration raised to a power equal to the coefficient of that ion in ...
What happens when AgBR crystals do not absorb light?
AgBr crystals that do not absorb light are then removed from the film to "fix" the image. Example: Let's calculate the solubility of AgBr in water in grams per liter, to see whether AgBr can be removed by simply washing the film. We start with the balanced equation for the equilibrium.
When is the equilibrium of the salt at equilibrium?
When it is equal to the solubility product for the salt , the system is at equilibrium. The reaction eventually comes back to equilibrium after the excess ions precipitate from solution as solid AgCl. When equilibrium is reestablished, however, the concentrations of the Ag + and Cl - ions won't be the same.
When will the solution described by Point E come to equilibrium?
The solution described by Point E will eventually come to equilibrium after enough solid AgCl has precipitated.

What Is solubility?
Table of Contents
Solubility Product
- The term solubility product is generally applicable for sparingly soluble salts. It is the maximum product of the molar concentration of the ions (raised to their appropriate powers) which are produced due to dissociation of the compound. At a given temperature the solubility product is constant. Lesser the value of solubility product indicates lower solubility and higher value of sol…
Recommended Videos
- On the basis of solubility, the factors affecting solubility vary on the state of the solute: 1. Liquids In Liquids 2. Solids In Liquids 3. Gases In Liquids
Solubility of Liquids in Liquids
- Water is known as a universal solvent as it dissolves almost every solute except for a few. Certain factors can influence the solubility of a substance. Solubility is the new bond formation between the solute molecules and solvent molecules. In terms of quantity, solubility is the maximum concentration of solute that dissolves in a known concentration of solvent at a given temperatur…
Solubility of Solids in Liquids
- It has been observed that solid solubility depends on the nature of the solute as well as the solvent. We often see that substances like sugar, common salt (NaCl), etc readily dissolve in water while substances like naphthalene do not dissolve in water. From the various observations and experimental results, it has been seen that only polar solutes tend to dissolve in the polar so…
Solubility of Gases in Liquids
- Gas solubility in liquids deals with the concept of gas dissolving in a solvent. Let us first define solubility. For any substance, solubility is the maximum amount of solute that can be dissolved in a given solvent at a particular temperature. Now our concern is gas solubility in liquids. The gas solubility in liquids is greatly affected by temperature and pressure as well as the nature of the s…
Factors Affecting Solubility
- It has been found that the gas solubility in liquids increases with increase in pressure. To have a better understanding of the effect of pressure on gas solubility let us consider a system of a g...