
Some important points about noble gases are following: Noble gases or rare gases are placed in the zero groups. These have eight electrons in the valance shell of each element except helium. Noble gases are isolated from air by fractional distillation.
What is special about the noble gases quizlet?
The noble gases are the elements in group 0 of the periodic table. They all have a full outer electron she'll. This is the most stable arrangement of electrons which other elements only achieve by making ions or sharing electrons in covalent bonds. This is why the noble gases are inert, compared with other elements.
What makes noble gas different?
Noble gases are the least reactive of all elements. That's because they have eight valence electrons, which fill their outer energy level. This is the most stable arrangement of electrons, so noble gases rarely react with other elements and form compounds.
What are 3 facts about noble gases?
Noble gases share many similar properties including:A full outer shell of electrons. ... Because of their full outer shells, they are very inert and stable. ... They are gases under standard conditions.They are colorless and odorless.Their melting and boiling points are close together giving them a very narrow liquid range.
What do all noble gases have in common?
The noble gases have high ionization energies and negligible electronegativities. The noble gases have low boiling points and are all gases at room temperature.
Why are some gases called noble gases?
Why are noble gases called noble? The noble gases are helium, argon, krypton, xenon, and radon, in order of their mass. They are called noble gases because they are so majestic that they do not react with anything in general. They're also known as inert gases for this reason.
Why are noble gasses unreactive?
Because they're reluctant to share electrons from their filled outer electron shells, noble gases are generally considered unreactive.
Which of the following is a defining property of noble gas?
In general, noble gases are colorless, odorless, nonflammable, and have a low reactivity. Their low reactivity is because they have a complete set of electrons in their outer valence.
Why are noble gases chemically inert?
Noble gases are chemically inert because they have completely filled orbitals in the valence shell which leads to stable electronic configuration.
1. How many Noble Gases are there in the Periodic Table?
There are six noble gases in the periodic table: helium, neon, argon, krypton, xenon, and radon. Noble gases are a group of chemical elements with...
2. Write some properties of noble gases.
Some properties of noble gases are shown below. They are non-reactive and are used to create an inert atmosphere. They have a complete external ele...
3.What is the difference between octet and double?
The main difference between the octet and the double is that the octet is an atom or ion with up to 8 electrons in the outermost shell. A doublet,...
4. What are the properties of noble gases?
Noble gases have a low chemical reactivity and are odourless, colourless, non-flammable, and monotone. Noble gases contain entire valence electron...
What are the properties of noble gases?
The properties of the noble gases can be well explained by modern theories of atomic structure: Their outer shell of valence electrons is considered to be "full", giving them little tendency to participate in chemical reactions, and it has been possible to prepare only a few hundred noble gas compounds.
Why are noble gases considered a group 0?
They were once labeled group 0 in the periodic table because it was believed they had a valence of zero, meaning their atoms cannot combine with those of other elements to form compounds. However, it was later discovered some do indeed form compounds, causing this label to fall into disuse.
How did the discovery of noble gases help the development of atomic structures?
The discovery of the noble gases aided in the development of a general understanding of atomic structure . In 1895, French chemist Henri Moissan attempted to form a reaction between fluorine, the most electronegative element, and argon, one of the noble gases, but failed. Scientists were unable to prepare compounds of argon until the end of the 20th century, but these attempts helped to develop new theories of atomic structure. Learning from these experiments, Danish physicist Niels Bohr proposed in 1913 that the electrons in atoms are arranged in shells surrounding the nucleus, and that for all noble gases except helium the outermost shell always contains eight electrons. In 1916, Gilbert N. Lewis formulated the octet rule, which concluded an octet of electrons in the outer shell was the most stable arrangement for any atom; this arrangement caused them to be unreactive with other elements since they did not require any more electrons to complete their outer shell.
What was the name of the element that was discovered in 1868?
Pierre Janssen and Joseph Norman Lockyer had discovered a new element on 18 August 1868 while looking at the chromosphere of the Sun, and named it helium after the Greek word for the Sun, ἥλιος ( hḗlios ). No chemical analysis was possible at the time, but helium was later found to be a noble gas. Before them, in 1784, the English chemist and physicist Henry Cavendish had discovered that air contains a small proportion of a substance less reactive than nitrogen. A century later, in 1895, Lord Rayleigh discovered that samples of nitrogen from the air were of a different density than nitrogen resulting from chemical reactions. Along with Scottish scientist William Ramsay at University College, London, Lord Rayleigh theorized that the nitrogen extracted from air was mixed with another gas, leading to an experiment that successfully isolated a new element, argon, from the Greek word ἀργός ( argós, "idle" or "lazy"). With this discovery, they realized an entire class of gases was missing from the periodic table. During his search for argon, Ramsay also managed to isolate helium for the first time while heating cleveite, a mineral. In 1902, having accepted the evidence for the elements helium and argon, Dmitri Mendeleev included these noble gases as group 0 in his arrangement of the elements, which would later become the periodic table.
How to use noble gas notation?
As a result of a full shell, the noble gases can be used in conjunction with the electron configuration notation to form the noble gas notation. To do this, the nearest noble gas that precedes the element in question is written first, and then the electron configuration is continued from that point forward. For example, the electron notation of phosphorus is 1s 2 2s 2 2p 6 3s 2 3p 3, while the noble gas notation is [Ne] 3s 2 3p 3. This more compact notation makes it easier to identify elements, and is shorter than writing out the full notation of atomic orbitals.
What is the color of a noble gas?
Atomic number color: red=gas. v. t. e. The noble gases (historically also the inert gases; sometimes referred to as aerogens) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity.
Which gas has the highest ionization potential?
The noble gases, which are labeled, have the largest ionization potential for each period. The noble gas atoms, like atoms in most groups, increase steadily in atomic radius from one period to the next due to the increasing number of electrons. The size of the atom is related to several properties.
What are the properties of noble gases?
Answer: Some properties of Noble gases are listed below: They are nonreactive and are used to form inert atmospheres. They have a complete outer electron or valence shell. They have high ionization energies.
Where do noble gases come from?
The term “noble gas” is derived from a translation of the German word Edelgas, which means “noble gas”. German chemist Hugo Erdmann termed this phrase in 1898.
What is the name of the group of 18 elements in the periodic table?
The noble gases are group 18 elements of the periodic table. Atoms of these elements have completely filled valence electron shells, making them relatively inert, colourless, odourless, monatomic gases at room temperature and pressure. Under the normal conditions of temperature and pressure, all the noble gases exist in the gaseous phase.
What is the atomic number of neon?
Neon is a second chemical element of group 18 and is denoted by the symbol Ne. The atomic number of neon is 10. Neon exists as a colourless monoatomic gas. This gas does not possess any colour, odour, taste and is lighter than air. The electron configuration of Neon is H e 2s22p6. It is used commonly in fluorescent lamps and electric signs. This gas occurs in minute quantities in Earth’s atmosphere and is trapped within the rocks of Earth’s crust. This element is more abundant in the cosmos as compared to Earth’s surface.
What is the first element in group 18?
Helium is the first chemical element of group 18 and is denoted by the symbol He. It is the first lightest noble gas and is known to possess the lowest boiling point. It is the second lightest element which is colourless, odourless, and tasteless. The atomic number of helium is 2. It is non-toxic in small concentrations, The electronic configuration of helium is 1s2. So, It has a total of 2 electrons in its valence shell. Helium cannot be solidified by sufficient cooling at normal atmospheric pressure, so it is necessary to apply pressure of 25 atmospheres at a temperature of −272 °C to convert it into a solid form.
Where is helium found?
Helium is mainly concentrated in stars, where it is synthesized from hydrogen by the nuclear fusion reaction. This gas is used as an inert-gas atmosphere for welding metals such as aluminium and is also used in rocket propulsion. Atomic Number: 2. Mass Number: 4. Melting point: -272 °C.
Who said noble gases do not react with other elements?
German chemist Hugo Erdmann termed this phrase in 1898. Like a nobleman consider it undignified to associate with common people, noble gases tend not to react with other elements. Other names for noble gases are rare gases, inert gases, and aerogens.
What is the noun for noble gas?
Noble gas is translated from German and was first used by Hugo Erdmann in 1898. The German noun for noble gas was Edelgas. [1] . In the periodic table, group 18 is the noble gases. All of the noble gases have a fragile interatomic force. They also all increase stably in atomic radius due to the raising number of electrons.
Why are noble gases used in refrigerant?
Noble gases have very low boiling and melting points, which would make them very useful in refrigerants. They are also commonly used in lighting. That is because of their ability to not react to most chemicals. That makes noble gases perfect in lighting.
What is the symbol for helium?
Helium is one of the noble gases. It is number two in the periodic table, which means it has two protons and two electrons. Its symbol is He. Helium’s boiling and melting point is the lowest in all of the elements. [2] . Helium is actually named after Helios, Greek god of the sun.
Why do noble gases not react to anything?
Helium, neon, argon, krypton, xenon, and radon are all noble gases. The reason why they don’t react to anything is because they have eight valence electrons, which makes them stable.
How many protons does neon have?
Having ten protons and electrons, eight valence electrons, neon is the second noble gas. Its symbol is Ne. Neon was discovered in 1898. It was recognized as a new element, when it emitted a bright red spectrum. [4] . It is also a very abundant element in the universe and solar system.
How many valence electrons does a noble gas have?
However, they soon discovered that the noble gases do indeed form some compounds with some other elements and have eight valence electrons. [1] William Ramsay discovered most of noble gases. He discovered krypton, neon, and also xenon.
Why is helium used in balloons?
Helium is commonly used in blimps, airships, and balloons due to the fact that helium is lighter than air, itself. Helium is completely safe for these applications, because it doesn’t burn or react to other chemicals (since it’s a noble gas). [3] .
Overview
The noble gases (historically also the inert gases; sometimes referred to as aerogens ) make up a class of chemical elements with similar properties; under standard conditions, they are all odorless, colorless, monatomic gases with very low chemical reactivity. The six naturally occurring noble gases are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn).
History
Noble gas is translated from the German noun Edelgas, first used in 1898 by Hugo Erdmann to indicate their extremely low level of reactivity. The name makes an analogy to the term "noble metals", which also have low reactivity. The noble gases have also been referred to as inert gases, but this label is deprecated as many noble gas compounds are now known. Rare gases is anot…
Physical and atomic properties
The noble gases have weak interatomic force, and consequently have very low melting and boiling points. They are all monatomic gases under standard conditions, including the elements with larger atomic masses than many normally solid elements. Helium has several unique qualities when compared with other elements: its boiling point at 1 atm is lower than those of any other …
Chemical properties
The noble gases are colorless, odorless, tasteless, and nonflammable under standard conditions. They were once labeled group 0 in the periodic table because it was believed they had a valence of zero, meaning their atoms cannot combine with those of other elements to form compounds. However, it was later discovered some do indeed form compounds, causing this label to fall into dis…
Occurrence and production
The abundances of the noble gases in the universe decrease as their atomic numbers increase. Helium is the most common element in the universe after hydrogen, with a mass fraction of about 24%. Most of the helium in the universe was formed during Big Bang nucleosynthesis, but the amount of helium is steadily increasing due to the fusion of hydrogen in stellar nucleosynthesis (and, to a very slight degree, the alpha decay of heavy elements). Abundances on Earth follow diff…
Applications
Noble gases have very low boiling and melting points, which makes them useful as cryogenic refrigerants. In particular, liquid helium, which boils at 4.2 K (−268.95 °C; −452.11 °F), is used for superconducting magnets, such as those needed in nuclear magnetic resonance imaging and nuclear magnetic resonance. Liquid neon, although it does not reach temperatures as low as liquid helium, a…
Discharge color
The color of gas discharge emission depends on several factors, including the following:
• discharge parameters (local value of current density and electric field, temperature, etc. – note the color variation along the discharge in the top row);
• gas purity (even small fraction of certain gases can affect color);
See also
• Noble gas (data page), for extended tables of physical properties.
• Noble metal, for metals that are resistant to corrosion or oxidation.
• Inert gas, for any gas that is not reactive under normal circumstances.
Shared Physical Or Chemical Properties
Noble Gases
- Helium
Helium is one of the noble gases. It is number two in the periodic table, which means it has two protons and two electrons. Its symbol is He. Helium’s boiling and melting point are the lowest in all of the elements. Helium is actually named after Helios, the Greek god of the sun. That is beca… - Neon
Having ten protons and electrons, and eight valence electrons, neon is the second noble gas. Its symbol is Ne. Neon was discovered in 1898. It was recognized as a new element when it emitted a bright red spectrum. It is also a very abundant element in the universe and solar system. Howe…
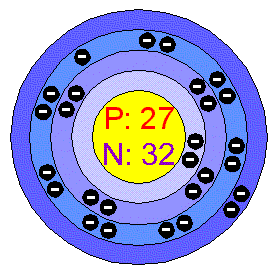
Different Ways to Show An Atom
- The Bohr Diagram
The Bohr Diagram is what scientists use to explain and show an atom’s subatomic particles. This technique was created by two scientists in 1913: Niels Bohr and Ernest Rutherford. This drawing is very simple and easy to do. The number of outer shells an atom has is the number of circles d… - The Lewis Dot-Diagram
The Lewis dot diagram is another way to explain an atom’s structure. More specifically, it represents the number of valence electrons an atom has. So, it only shows the last outer shell of an atom. The Lewis dot-diagram was created by Gilbert N. Lewis. In 1916, he showcased it in an …
Summary of The Diagrams
- Ultimately, there are many different ways scientists use to represent and explain atoms. The Lewis diagram is extremely helpful when one wants to see what will happen if two atoms come together (the sharing of atoms). The Bohr diagram shows the whole structure of an atom. Ultimately, there are many different simple ways to explain what an atom is.