A stable nucleus is a nucleus that has enough binding energy to hold the elements of the nucleus together permanently. In unstable nuclei, the strong nuclear forces do not generate enough binding energy to hold the elements of the nucleus together permanently. Hadrons (Mesons & Baryons) experience the strong nuclear force but Leptons do not.
Which type of nucleus will most likely be unstable?
The end of the stable elements in the periodic table occurs after lead, largely due to the fact that nuclei with 128 neutrons are extraordinarily unstable and almost immediately shed alpha particles. This also contributes to the very short half-lives of astatine , radon , and francium relative to heavier elements.
What nuclei would be the least stable?
• Nuclides containing odd numbers of both protons and neutrons are the least stable means more radioactive. • Nuclides containing even numbers of both protons and neutrons are most stable means less radioactive.
What are very large nuclei tend to be unstable?
Very large nuclei tend to be unstable because of the: repulsive forces between protons. In nuclear reactions: small amounts of mass are converted to large amount of energy. For the most common types of radioactive decay, the order of least penetrating to human tissue, to most penetrating to human tissue is: ...
How does an unstable nucleus achieve stability?
How does an unstable nucleus achieve stability? It emits particles or waves. Conversion to moles. From mass (grams) to moles requires the atomic weights of elements or molecular weights of compounds (the sum of the atomic weights of their elements) 100 grams of salt (NaC1)

What are unstable nuclei?
An unstable atom has excess internal energy, with the result that the nucleus can undergo a spontaneous change towards a more stable form. This is called 'radioactive decay'. Each element exists in the form of atoms with several different sized nuclei, called isotopes.
What are stable nuclei examples?
For example carbon, carbon-12 is stable and has six protons and six neutrons. However as the number of protons increases, more neutrons are needed to keep the nucleus stable. For example lead, lead-206 has 82 protons and has 124 neutrons.
What is stability of nuclei?
Definition. Nuclear stability means that the nucleus of an element is stable and thus it does not decay spontaneously emitting any kind of radioactivity. Among the ≈9,000 nuclei expected to exist, and the ≈3,000 presently known, only 195 are stable against spontaneous decay, because of energy conservation.
What causes unstable nuclei?
When the atoms of an element have extra neutrons or protons it creates extra energy in the nucleus and causes the atom to become unbalanced or unstable.
Where are unstable nuclei?
The unstable nuclei lie above and below the line of stability in the neutron – proton plot. This gives information of the type of radioactive decay they will undergo. Nuclei which lie above the line of stability contain too many neutrons to be stable. They are referred to as “neutron rich”.
What is the most unstable nucleus?
0:324:53Stable and Unstable Nuclei | Radioactivity | Physics | FuseSchoolYouTubeStart of suggested clipEnd of suggested clipThemselves by giving off the excess proton or neutron. This is radioactive decay unstable nuclei areMoreThemselves by giving off the excess proton or neutron. This is radioactive decay unstable nuclei are radioactive. And emit radiation.
How many nuclei are stable?
Most naturally occurring nuclides are stable (about 252; see list at the end of this article), and about 34 more (total of 286) are known to be radioactive with sufficiently long half-lives (also known) to occur primordially.
Are all nuclei stable?
Although most of the known elements have at least one isotope whose atomic nucleus is stable indefinitely, all elements have isotopes that are unstable and disintegrate, or decay, at measurable rates by emitting radiation. Some elements have no stable isotopes and eventually decay to other elements.
How do nuclei become stable?
A stable nucleus must have the right combination of protons and neutrons. Occurs if there are too many neutrons. A neutron to proton conversion occurs. This releases an electron or beta particle.
Why are protons stable?
According to the Standard Model, the proton, a type of baryon, is stable because baryon number (quark number) is conserved (under normal circumstances; see chiral anomaly for an exception).
Why Uranium is unstable?
Uranium-235 (U-235) is one of the isotopes that fissions easily. During fission, U-235 atoms absorb loose neutrons. This causes U-235 to become unstable and split into two light atoms called fission products.
What are the most stable nuclei?
The most common isotope of iron, iron-56, is thought to be the most stable nucleus since it has the lowest mass per nucleon of all nuclides. Iron-56 is also a strongly and effectively bound nucleus, having a binding energy of. 8 MeV per nucleon.
What is an example of a stable element?
A stable element by definition is a chemical element (found on the Periodic Table) that has atleast one stable, naturally occurring isotope. For example, Helium has 9 isotopes, but since two of them are stable (He-3 and He-4), Helium is considered to be a stable element.
What are examples of nuclei?
For instance, in botany, the nucleus may also refer to the central kernel of a nut or seed, or the center of a starch granule. In neuroanatomy, a nucleus is a group of cell bodies of nerve cells in the brain or spinal cord.
Is Helium 3 a stable nuclei?
Helium-3 (3He see also helion) is a light, stable isotope of helium with two protons and one neutron (the most common isotope, helium-4, having two protons and two neutrons in contrast). Other than protium (ordinary hydrogen), helium-3 is the only stable isotope of any element with more protons than neutrons.
What is stable nucleus?
A stable nucleus is a nucleus that has enough binding energy to hold the elements of the nucleus together permanently. In unstable nuclei, the strong nuclear forces do not generate enough binding energy to hold the elements of the nucleus together permanently. Hadrons (Mesons & Baryons) experience the strong nuclear force but Leptons do not.
What is the decay of unstable nuclei?
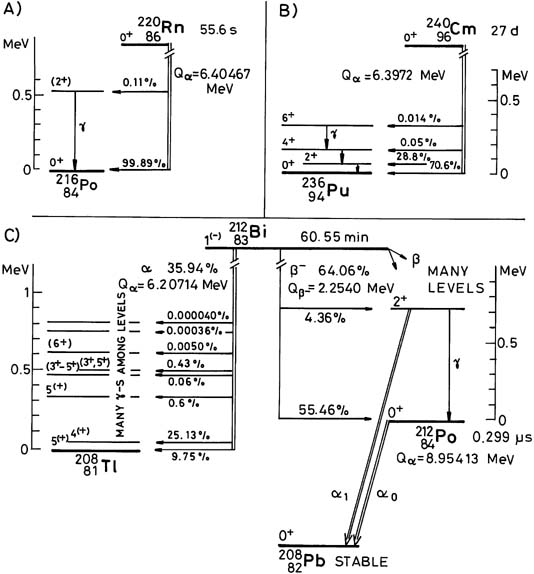
B. Decay of Unstable Nuclei. The unstable nuclei, in order to become stable nuclei, emit particles and/or electromagnetic radiation. These types of nuclei are said to be radioactive, and the emission is called radioactivity. The three type of emission that originate from the nucleus are: Gamma particles (γ).
What are the three types of nuclei?
The unstable nuclei, in order to become stable nuclei, emit particles and/or electromagnetic radiation. These types of nuclei are said to be radioactive, and the emission is called radioactivity. The three type of emission that originate from the nucleus are: 1 Alpha particles (α) 2 Beta particles (β) 3 Gamma particles (γ).
How does the nucleus return to its unexcited state?
The nucleus may return to its unexcited or stable state by emitting energy in the form of γ particles. γ−radiation is part of the electromagnetic spectrum with wavelengths between. For example, when uranium-238 decays by emitting an α particle, the resulting nucleus of thorium-234 contains excess energy and emits a photon ...
How many protons does the nucleus lose?
The nucleus loses two protons and two neutrons in this emission. Each element has a particular number of protons, and therefore α decay causes one element to change into another. This process is also called transmutation. The original nuclide is called the parent nuclide, and the new one is the daughter nuclide.
What type of nucleus is a negative electron?
A radioactive nucleus that undergoes alpha decay emits alpha particles. It contains two protons and two neutrons. A radioactive nucleus that undergoes beta decay may emit a negative or positive electron. The nucleus may return to its unexcited or stable state by emitting energy in the form of γ particles.
What is the attractive force that holds the elements of the nucleus together called?
The attractive force, similar in size to the electrostatic repulsion, which holds the elements of the nucleus together, is called as the strong nuclear force . An unstable nucleus, in order to become a stable nucleus, emits α, β or γ particles and/or electromagnetic radiation.
How do neutrons and protons contribute to nuclear stability?
These two forces compete, leading to various stability of nuclei. There are only certain combinations of neutrons and protons, which forms stable nuclei . Neutrons stabilize the nucleus, because they attract each other and protons , which helps offset the electrical repulsion between protons. As a result, as the number of protons increases, an increasing ratio of neutrons to protons is needed to form a stable nucleus. If there are too many ( neutrons also obey the Pauli exclusion principle) or too few neutrons for a given number of protons, the resulting nucleus is not stable and it undergoes radioactive decay . Unstable isotopes decay through various radioactive decay pathways, most commonly alpha decay, beta decay, or electron capture. Many other rare types of decay, such as spontaneous fission or neutron emission are known.
How is nuclear decay measured?
The rate of nuclear decay is also measured in terms of half-lives. The half-life is the amount of time it takes for a given isotope to lose half of its radioactivity. Half lives range from millionths of a second for highly radioactive fission products to billions of years for long-lived materials (such as naturally occurring uranium ). Notice that short half lives go with large decay constants. Radioactive material with a short half life is much more radioactive (at the time of production) but will obviously lose its radioactivity rapidly. No matter how long or short the half life is, after seven half lives have passed, there is less than 1 percent of the initial activity remaining.
What happens when there are too many neutrons?
If there are too many or too few neutrons for a given number of protons, the resulting nucleus is not stable and it undergoes radioactive decay. Unstable isotopes decay through various radioactive decay pathways. Periodic Table
How does radioactive decay occur?
Nuclear decay (Radioactive decay) occurs when an unstable atom loses energy by emitting ionizing radiation. Radioactive decay is a random process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. During radioactive decay an unstable nucleus spontaneosly and randomly decomposes to form a different nucleus (or a different energy state – gamma decay), giving off radiation in the form of atomic partices or high energy rays. This decay occurs at a constant, predictable rate that is referred to as half-life. A stable nucleus will not undergo this kind of decay and is thus non-radioactive. There are many modes of radioactive decay:
What is the alpha decay?
Alpha radioactivity. Alpha decay is the emission of alpha particles (helium nuclei). Alpha particles consist of two protons and two neutrons bound together into a particle identical to a helium nucleus. Because of its very large mass (more than 7000 times the mass of the beta particle) and its charge, it heavy ionizes material and has a very short range.
What is gamma radioactivity?
Gamma radioactivity consist of gamma rays. Gamma rays are electromagnetic radiation (high energy photons) of an very high frequency and of a high energy. They are produced by the decay of nuclei as they transition from a high energy state to a lower state known as gamma decay. Most of nuclear reactions are accompanied by gamma emission.
Do nuclei have quadrupole moments?
These nuclei appear to be perfectly spherical in shape; they have zero quadrupole electric moments.
How do neutrons and protons contribute to nuclear stability?
These two forces compete, leading to various stability of nuclei. There are only certain combinations of neutrons and protons, which forms stable nuclei . Neutrons stabilize the nucleus, because they attract each other and protons , which helps offset the electrical repulsion between protons. As a result, as the number of protons increases, an increasing ratio of neutrons to protons is needed to form a stable nucleus. If there are too many ( neutrons also obey the Pauli exclusion principle) or too few neutrons for a given number of protons, the resulting nucleus is not stable and it undergoes radioactive decay . Unstable isotopes decay through various radioactive decay pathways, most commonly alpha decay, beta decay, or electron capture. Many other rare types of decay, such as spontaneous fission or neutron emission are known.
How is nuclear decay measured?
The rate of nuclear decay is also measured in terms of half-lives. The half-life is the amount of time it takes for a given isotope to lose half of its radioactivity. Half lives range from millionths of a second for highly radioactive fission products to billions of years for long-lived materials (such as naturally occurring uranium ). Notice that short half lives go with large decay constants. Radioactive material with a short half life is much more radioactive (at the time of production) but will obviously lose its radioactivity rapidly. No matter how long or short the half life is, after seven half lives have passed, there is less than 1 percent of the initial activity remaining.
What happens when there are too many neutrons?
If there are too many or too few neutrons for a given number of protons, the resulting nucleus is not stable and it undergoes radioactive decay. Unstable isotopes decay through various radioactive decay pathways. Radiation Dosimetry
How does radioactive decay occur?
Nuclear decay (Radioactive decay) occurs when an unstable atom loses energy by emitting ionizing radiation. Radioactive decay is a random process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. During radioactive decay an unstable nucleus spontaneosly and randomly decomposes to form a different nucleus (or a different energy state – gamma decay), giving off radiation in the form of atomic partices or high energy rays. This decay occurs at a constant, predictable rate that is referred to as half-life. A stable nucleus will not undergo this kind of decay and is thus non-radioactive. There are many modes of radioactive decay:
What is the alpha decay?
Alpha radioactivity. Alpha decay is the emission of alpha particles (helium nuclei). Alpha particles consist of two protons and two neutrons bound together into a particle identical to a helium nucleus. Because of its very large mass (more than 7000 times the mass of the beta particle) and its charge, it heavy ionizes material and has a very short range.
What is gamma radioactivity?
Gamma radioactivity consist of gamma rays. Gamma rays are electromagnetic radiation (high energy photons) of an very high frequency and of a high energy. They are produced by the decay of nuclei as they transition from a high energy state to a lower state known as gamma decay. Most of nuclear reactions are accompanied by gamma emission.
Do nuclei have quadrupole moments?
These nuclei appear to be perfectly spherical in shape; they have zero quadrupole electric moments.
What is unstable nucleus?
Unstable Nuclei. In unstable nuclei the strong nuclear forces do not generate enough binding energy to hold the nucleus together permanently. It is unstable nuclei that are radioactive and are referred to as radioactive nuclei and in the case of their isotopes called radioisotopes.
Which nuclei are on the stability line?
Nuclei which lie on the stability line are stable nuclei. From the plot it can be seen that many of the stable nuclei have equal number of protons and neutrons. These are usually the elements in the lighter section of the periodic table. For the heavier stable nuclei the there are approximately 50% more neutrons to protons.
What is the force between protons and neutrons called?
Strong Nuclear Force. A strong force must exist between the protons and neutrons in a nucleus to overcome the electrostatic repulsion between the protons. This additional force is called the strong nuclear force. This force is attractive over short distances, but this is not all.
Why is the combined gravitational force from the protons and neutrons in a nucleus insignificant?
The combined gravitational force from the protons and neutrons in a nucleus is insignificant as an attractive force because their masses are so tiny. This implies there must be an additional attractive force similar in size to the electrostatic repulsion which holds the nucleus together.
What is neutron rich?
They are referred to as “neutron rich”. Those that lie below the line of stability contain too many protons to be stable and are called “proton rich”. In summary it is the balance of protons and neutrons in a nucleus which determines whether a nucleus will be stable or unstable.
What is the force that holds the nucleus together called?
This is the force that holds the nucleus together and the energy associated with this force is called the binding energy. It is the amount of strong nuclear force and the associated binding energy in ...
Is the nucleus stable?
Many nucle i in nature are very stable, most of the nuclei formed at the creation of the universe or after supernovae explosions many millions of years ago are still in existence now. The graph below is a plot of neutron number against proton number. It is used as rule to determine which nuclei are stable or unstable.
Which nucleus is more stable?
It is found that nuclei with even numbers of protons and neutrons are more stable than those with odd numbers. Nuclei which have both neutron number and proton number equal to one of the magic numberscan be called “doubly magic“, and are found to be particularly stable. There are further special propertis of nu clei, ...
How is nuclear stability determined?
A nuclear stabilityis determined by the competition between two fundamental interactions. Atomic nuclei consist of protons and neutrons, which attract each other through the nuclear force, while protons repel each other via the electromagnetic force due to their positive charge. These two forces compete, leading to various stability of nuclei. ...
What is the role of neutrons in nuclear reactors?
This type of radiation plays key role in nuclear reactor control, because these neutrons are delayed neutrons.
Why do neutrons stabilize the nucleus?
Neutrons stabilize the nucleus, because they attract each other and protons , which helps offset the electrical repulsion between protons. As a result, as the number of protons increases, an increasing ratio of neutrons to protons is needed to form ...
What is the closing of shells?
These correspond to the closing of shells (or sub-shells). Nuclei with closed shells are more tightly bound than the next higher number. The closing of shells occurs at Z or N = 2, 8, 20, 28, (40), 50, 82, 126. It is found that nuclei with even numbers of protons and neutrons are more stable than those with odd numbers.
Which nuclei have a magic number?
The stable elements at the end of the decay series all have a “magic number” of neutrons or protons. The nuclei He-4, O-16, and Pb-208 (82 protons and ...
How does nuclear decay occur?
Nuclear decay (Radioactive decay) occurs when an unstable atom loses energy by emitting ionizing radiation. Radioactive decay is a random process at the level of single atoms, in that, according to quantum theory, it is impossible to predict when a particular atom will decay. During radioactive decay an unstable nucleus spontaneosly and randomly decomposes to form a different nucleus (or a different energy state – gamma decay), giving off radiation in the form of atomic partices or high energy rays. This decay occurs at a constant, predictable rate that is referred to as half-life. A stable nucleus will not undergo this kind of decay and is thus non-radioactive. There are many modes of radioactive decay:
