
What is the nuclear charge of an atom?
What is the nuclear charge of an atom? Nuclear charge is a measure of the ability of protons in the nucleus to attract the negative electrons in orbit around the nucleus. Electrons are attracted to the nucleus as it is positively charged, but electrons in the inner shells can negate some of the attraction of the nucleus on the outermost electrons.
What does the nucleus of an atom contain?
The nucleus of an atom contains positively charged particles called protons. Electrons are attracted to the nucleus as they are negatively charged. However, negatively charged electrons around the nucleus are organized into layers called orbitals which repel each other, and negate some of the positive charge of the nucleus.
What is the difference between the electron and the effective nuclear charge?
The electron is the smallest sub atomic particle that make up the atom. Has a negative charge and is located in shells that orbit the nucleus The effective nuclear charge is the attraction of the nucleus to the valence electron taking into account the number of protons and the number of inner shell electrons.
How are negatively charged electrons arranged around the nucleus?
These negatively charged electrons are arranged into shells which form layers surrounding the nucleus. Elements in different groups on the periodic table have different numbers of electrons in their outermost shells. These outer electrons are also known as valence electrons. 1. How do you find effective nuclear charge?
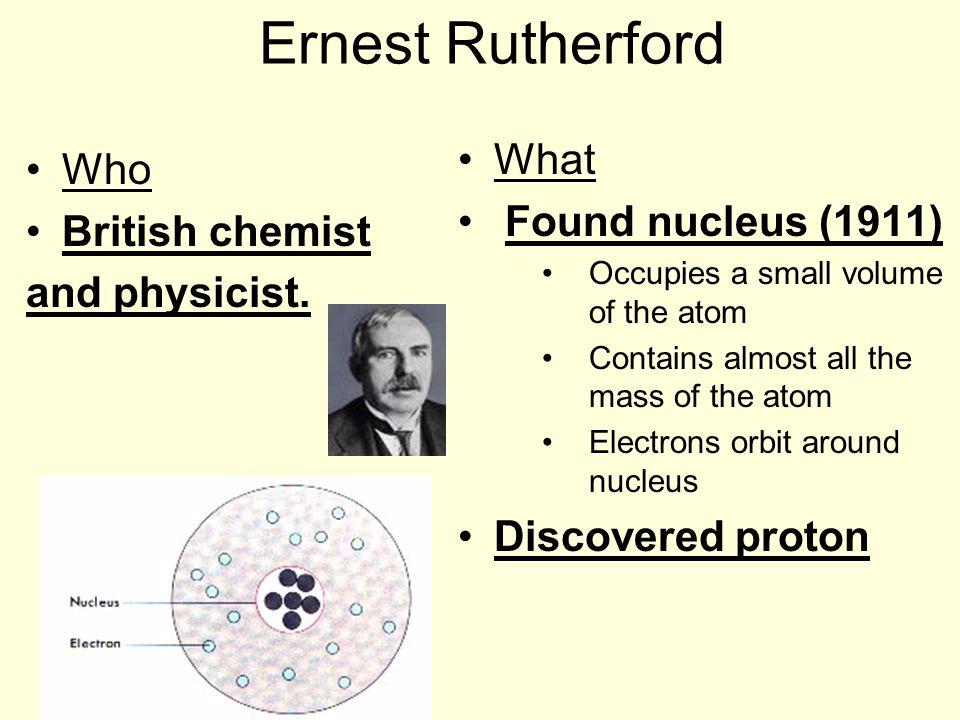
How do you find the charge of the nucleus of an atom?
0:081:46Calculating the Charge of an Atom - YouTubeYouTubeStart of suggested clipEnd of suggested clipBy comparing the number of protons to the number of electrons. We can determine whether the chargeMoreBy comparing the number of protons to the number of electrons. We can determine whether the charge of an atom is positive or negative and also can calculate the total charge of the atom.
Is the nucleus charge positive or negative?
Most electric charge is carried by the electrons and protons within an atom. Electrons are said to carry negative charge, while protons are said to carry positive charge, although these labels are completely arbitrary (more on that later).
What charge is a neutron?
no chargeUnlike protons and electrons, which are electrically charged, neutrons have no charge—they are electrically neutral. That's why the neutrons in the diagram above are labeled n0. The zero stands for "zero charge". The mass of a neutron is slightly greater than the mass of a proton, which is 1 atomic mass unit (amu).
What does the negative (-) charge indicate?
What Does Negative Charge Mean? A negative charge is an electrical property of a particle at the subatomic scale. An object is negatively charged if it has an excess of electrons, and is uncharged or positively charged otherwise.
Is the nucleus of a cell positively charged?
The nucleus is a small, dense region at the center of the atom. It consists of positive protons and neutral neutrons, so it has an overall positive charge.
What is nucleus positive and negative?
As nucleus is positively charged and electron is negatively charged in an atom , both of them having opposite charges then why they did not attract each other.
Is the nucleus of an atom positive?
Atomic nuclei consist of electrically positive protons and electrically neutral neutrons. These are held together by the strongest known fundamental force, called the strong force.
Is a neutron positive or negative?
electrically neutralMiller, a UW physics professor, has found that the neutron has a negative charge both in its inner core and its outer edge, with a positive charge sandwiched in between to make the particle electrically neutral.
What is nuclear charge?
Nuclear charge is a measure of the effect of the number of protons in the nucleus and their ability to attract the negative electrons in orbits around the nucleus. Refresher: Atoms are composed of a nucleus, containing positively charged protons and neutral neutrons, surrounded by a cloud of negatively charged electrons.
Why is nuclear charge important?
Knowing the effective nuclear charge allows us to determine how much energy is required to remove one electron, also known as ionizing the atom. Ionization energy is an important concept for reactivity of the elements.
What is Effective Nuclear Charge?
However, negatively charged electrons around the nucleus are organized into layers called orbitals which repel each other, and negate some of the positive charge of the nucleus. This results in a varying attraction of the nucleus on the electrons surrounding the nucleus, which is known as nuclear charge. The presence of electrons on the inner shells of an atom “shield” the outermost electron from feeling the full positive charge. Because of the varying charge on electrons in different orbitals, we typically refer to the effective nuclear charge, which is the effect of the nucleus experienced by the outermost electron of the atom, taking into account the shielding effect of inner electrons.
What is the effect of the nucleus on the outermost electron of an atom?
Because of the varying charge on electrons in different orbitals, we typically refer to the effective nuclear charge, which is the effect of the nucleus experienced by the outermost electron of the atom, taking into account the shielding effect of inner electrons.
What is the name of the attraction of the nucleus to the electrons surrounding the nucleus?
This results in a varying attraction of the nucleus on the electrons surrounding the nucleus, which is known as nuclear charge. The presence of electrons on the inner shells of an atom “shield” the outermost electron from feeling the full positive charge.
What is the net attraction of electrons to the nucleus?
The net attraction on these outer electrons is known as effective nuclear charge.
What are negatively charged electrons?
These negatively charged electrons are arranged into shells which form layers surrounding the nucleus. Elements in different groups on the periodic table have different numbers of electrons in their outermost shells. These outer electrons are also known as valence electrons. .
How to describe the nucleus?
The Composition of the Nucleus: 1 A physicist looks at a stone table and says that it is mostly empty space inside. How is this statement true? 2 Matter is made up of very small particles called atoms. 3 Atoms are composed of three fundamental particles: the proton, neutron and electron. 4 Figure shows the location of the protons, neutrons and electrons in an atom. 5 Protons and neutrons are the nucleons. The protons and neutrons form a very small, dense core known as the nucleus. Therefore, most of the mass of an atom is contained in its nucleus. 6 Electrons move in orbits around the nucleus. 7 The diameter of the atom is about 100 000 times bigger than the diameter of the nucleus. 8 This means that there is a lot of empty space within an atom. 9 (a) A proton is a positively-charged particle. It carries a charge of +e, where e = 1.6 × 10 -19 C.#N#(b) A neutron carries no charge.#N#(c) An electron carries a negative charge of the same magnitude as the proton.#N#(d) Therefore, a proton and an electron are oppositely charged.
How many nucleons are in the nucleus?
The nuclide notation gives the symbol of the element, X, the proton number, Z and the nucleon number, A. The nuclide notation is written as follows: K = Potassium, A = 40 and Z = 19 Therefore, the nucleus contains 40 nucleons. There are 19 protons and 21 neutrons in the nucleus.
How many protons are in a titanium nucleus?
Nucleus of an Atom Examples. A nuclide of titanium (Ti) contains 22 protons and 26 neutrons. (a) State the proton number and nucleon number of this nuclide. (b) Write the nuclide notation. Solution: (a) Proton number = Number of protons = 22. Nucleon number = Number of proton and neutrons. = 22 + 26 = 48.
What is the symbol for a nuclide?
What is the symbol for nuclide? The proton number, Z is the number of protons in a nucleus. The nucleon number, A gives the total number of protons and neutrons in a nucleus. The number of neutrons, N = A – Z. A nuclide is a nucleus with a particular proton number and a particular nucleon number.
Which element has a number of neutrons equal to the number of protons?
Table gives the number of protons, number of neutrons and the nuclide notations of some common elements. For lighter elements such as helium, carbon and oxygen, it is common for the number of protons to be equal to the number of neutrons. Lithium is one example where the number of neutrons is not equal to the number of protons.
How do electrons move?
Electrons move in orbits around the nucleus. The diameter of the atom is about 100 000 times bigger than the diameter of the nucleus. This means that there is a lot of empty space within an atom. (a) A proton is a positively-charged particle.
