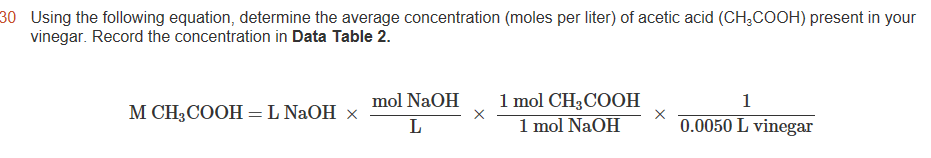
C (vinegar) = 60g/mol
Mole
The mole is the unit of measurement in the International System of Units (SI) for amount of substance. It is defined as the amount of a chemical substance that contains as many elementary entities, e.g., atoms, molecules, ions, electrons, or photons, as there are atoms in 12 grams of carbon-12 (C), the isotope of carbon with relative atomic mass 12 by definition.
Is acetic acid and vinegar the same thing?
Vinegar is 7-8% solution by volume Acetic Acid, while Acetic acid is pure Acetic acid itself. The IUPAC name of Acetic Acid is Ethanoic Acid. 100% pure Acetic acid is glacial Acetic acid. It solidifies just below normal room temperature. 5–8% Acetic acid in Water is called Vinegar. Vinegar is 5% acetic acid.
Is acetic acid stronger than citric acid?
Acetic acid, also named named ethanoic acid, is an organic acid with the chemical formula CH3COOH while citric acid, also weak organic acid has the chemical formula C6H8O7. Both of these are relatively weak acids, but citric acid is slightly stronger than acetic acid.
Does vinegar interact with acetic acid?
Yes, vinegar is a mixture of acetic acid and water. If you dissolve salt in vinegar, it is a three part mixture of water, acetic acid and sodium chloride. Since none of these components will react with one another, they will remain a mixture after they are combined. 525 views.
Is acetic acid the most volatile liquid?
Acetic acid is a colourless liquid; with a strong vinegar-like odour. It is flammable, and at temperatures warmer than 39°C, explosive vapour/air mixtures may be formed. Acetic acid is considered a volatile organic compound by the National Pollutant Inventory.

What is concentration of acetic acid in vinegar?
The acetic acid content of vinegar can vary widely, but for table vinegar it typically ranges from 4 to 8 % v/v. When used for pickling, the acetic acid content can be as high as 12 %.
How many grams of acetic acid are in each mL of vinegar?
The density of acetic acid is 1.05 g/mL.
What is the concentration of 5% acetic acid in vinegar?
5% means that for 1000 g of vinegar solution there is 50 g de acetic acid. Since molar mass is 60 g so 50/60 gives 0.83 mol/l as a molarity.
How do you calculate the amount of acetic acid in vinegar?
The number of moles of sodium hydroxide equals the number of moles of acetic acid in your vinegar sample (Ms). The sample volume was 1 mL, or 0.001 L (Vs). You can use a proportion to determine the number of moles of acetic acid (Mx) in a standard volume (Vx = 1 L) of vinegar: Ms/Vs = Mx/Vx.
How many grams of acetic acid are in a 250 mL bottle of vinegar?
13.125 g.Therefore, the mass of acetic acid present in 250.0 mL of vinegar is 13.125 g. Note: The volume/volume percentage is the measure of the substance present in the solution.
How do you find the molar concentration of acetic acid in a vinegar sample?
You will want to multiply the moles calculated by 4 to get moles of acetic acid in the 100mL of 10% solution. Then by dividing these moles by the volume of original acid that was diluted into 100 mL (because the moles of acetic acid all came from the 10 mL of vinegar), the molarity of the acetic acid can be found.
What is the density of vinegar g ml?
The molecular weight of acetic acid is 60.1 g/mol. Using these values and the data from table 2, calculate the average percentage of acetic acid in vinegar.
What concentration is distilled vinegar?
The distilled white vinegar you see in the condiment aisle contains around five percent acetic acid and 95 percent water. Cleaning vinegar contains around six percent acetic acid.
Is vinegar 5% by mass or volume?
Vinegar is an aqueous solution composed of acetic acid and water. Commercial vinegar sold in supermarkets usually contain 5 % acetic acid per volume.
How do you calculate the concentration of acetic acid?
Calculating the Concentration of Acetic Acid in Vingear in mol L -1Write the balanced chemical equation for the neutralisation reaction: ... Extract all the relevant data from the experiment. ... Check the data for consistency: ... Calculate the moles of NaOH(aq), n(NaOH) moles = concentration in mol L-1 x volume in L = n = c x V.More items...
How do you calculate concentration?
The standard formula is C = m/V, where C is the concentration, m is the mass of the solute dissolved, and V is the total volume of the solution. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.
How do you calculate acetic acid?
0:5712:21Lab Determining Percent of Acetic Acid in Vinegar - YouTubeYouTubeStart of suggested clipEnd of suggested clipThe amount of acetic acid in it. So just to review a few things in a titration one solution is thatMoreThe amount of acetic acid in it. So just to review a few things in a titration one solution is that slowly the other until the equivalence point is reached at the equivalence point of neutralization
How much does 25 ml of acetic acid weigh?
thus,given mass of CH3COOH =25 × 1.058 OR 26.45 gm.
How many grams are in acetic acid?
The molar mass of acetic acid is 60 g/mol.
How do you convert grams into ML?
1:475:41How To Convert Grams to Milliliters - g to mL - YouTubeYouTubeStart of suggested clipEnd of suggested clipIf you have one number on top and one number on the bottom you need to divide. So the operation thatMoreIf you have one number on top and one number on the bottom you need to divide. So the operation that we're going to perform is division it's 74 divided by 1.45.
Can you measure ml in grams?
How much is 1 ml in grams? For water, 1 milliliter equals 1 gram. For other ingredients, the weight varies. 1ml of milk weighs 1.04 grams, 1ml of flour weighs 0.53 grams and 1 ml of sugar equals 0.85 grams.
How do you find the percentage of acetic acid in vinegar?
To find the percentage of acetic acid in vinegar, you need to know the concentration of the acetic acid in the vinegar. The acetic acid concentrati...
What percent is acetic acid?
Acetic acid is a sour-tasting, colorless liquid with a strong, pungent odor. It is an organic compound with the chemical formula CH3COOH. Acetic ac...
What is the concentration of 5% acetic acid in vinegar?
The concentration of 5% acetic acid in vinegar is 0.5%. This is a relatively low concentration, which is why vinegar is safe to consume. However, i...
How to measure KHP?
Add approximately 0.5 grams of potassium hydrogen phthalate (KHP) to the beaker. Record the mass of the beaker and KHP to the nearest 0. 001 g. Calculate the mass of KHP by difference and record it. Add approximately 50 mL of distilled water to the beaker. Stir the solution until the KHP has dissolved completely.
How to find the equivalence point of a pH meter?
Using a pH meter and a graph of pH plotted versus the volume of base added, the equivalence point can be accurately found by finding the point that is in the middle of the vertical part of the curve. Once the equivalence point of the titration is known, the concentration of the sodium hydroxide can be determined. Sodium hydroxide is used to titrate vinegar so that the concentration of the vinegar is determined. The percentage of acetic acid in the solution can be determined from the concentration of the vinegar.
What is the name of the solution used to titrate vinegar?
Sodium hydroxide is used to titrate vinegar so that the concentration of the vinegar is determined. The percentage of acetic acid in the solution can be determined from the concentration of the vinegar.
What is the percent mass of CH3COOH?
Percent mass: 5. 115% CH3COOH From the plots determine the volume of NaOH required to neutralize the KHP solution in each titration.
What is titration in chemistry?
Titration is a method used in order to ascertain the amount of a constituent in a solution by measuring the volume of a known concentration of a reagent required to complete a reaction with it , typically using a burette. The equivalence point of a titration occurs when chemically equivalent amounts of acid and base are present.
What is the equivalence point of a titration?
The equivalence point of a titration occurs when chemically equivalent amounts of acid and base are present. In this experiment, the equivalence point occurs when the moles of acid in the solution equals the moles of base added in the titration. A rapid change in pH with the small addition of acid or base is the indicator of acid-base equivalency.
Can NaOH be prepared in a beaker?
The solution can be prepared in a beaker, it is not necessary to use a graduated cylinder or a volumetric flask because the NaOH solution will be standardized. Weigh a 150-mL beaker and record the mass to the nearest 0. 001 g. Add approximately 0.5 grams of potassium hydrogen phthalate (KHP) to the beaker.
How to titrate acetic acid?
Procedure to titrate the acetic acid in vinegar: 1 Rinse a clean 250 mL conical (erlenmeyer) flask with water. 2 Rinse a clean 25.00 mL pipette (pipet) with vinegar.#N#Pipette 25.00 mL of vinegar into the 250 mL conical (erlenmeyer) flask. 3 Add 2 drops of phenolphthalein indicator to the vinegar.#N#(The solution will remain colourless) 4 Rinse a clean 50.00 mL burette (buret) with standardised 1.00 mol L -1 aqueous sodium hydroxide solution.#N#Fill the burette (buret) with this standardised 1.00 mol L -1 NaOH (aq). 5 Set up the equipment as in the diagram on the right. 6 Run NaOH (aq) from the burette (buret) into the conical (erlenmeyer) flask until the solution changes colour from colourless to pink. 7 Repeat the titration carefully several times until concordant titres are achieved.
How to determine the concentration of acetic acid in vinegar?
It is therefore possible to determine the concentration of acetic acid in vinegar by titrating the vinegar with a strong base such as aqueous sodium hydroxide solution.
What is the best vinegar for titration?
For this experiment it is best to choose a "white vinegar" (that is, a colourless vinegar) rather than a " brown vinegar". The colour of a "brown vinegar" can mask the colour change at the end pointof the titration.
How is 20-25% vinegar made?
20-25% This is rarely sold to the public except as weed killer. It is made by centrifugation of lower acidity frozen vinegar crystals since vinegar bacteria can’t make vinegar this strong.
What is the acidity of vinegar?
5% acidity The standard acidity range for most vinegars. Good for use in canning if you are using good canning practices. Also can be used for any variety of cooking and most types of cleaning. Too weak to use for weed killer except in large amounts. 6-7% acidity Most wine and balsamic vinegars fall in this range.
What is the acidity of Essigessenz?
Germany’s Surig sells Essigessenz which is 25% acidity and can be used for cooking in only very small amounts. Gloves and eye protection are required in even small amounts. Gloves should be latex or nitrile and eye protection should be chemistry lab type goggles, not plastic impact safety glasses.
What is the minimum acidity level for vinegar?
4% acidity This is the minimum legal acidity level for vinegar in almost all countries. Most bargain and discount vinegar brands now are 4%. They don’t advertise this in large print so check the label. Only use for basic cooking and salad dressings. Do not use for canning.
What is 15% vinegar?
15% The mid-level of high strength vinegar. Only used in cooking in amounts of 1/3 typical vinegar and usually used for cleaning and weed killing. Gloves and eye protection should be used when handling. Rinse of any skin contact immediately. 20-25% This is rarely sold to the public except as weed killer.
Does vinegar have acidity?
We have talked about acidity a bit on this blog, from how to measure acidity and how it is different from pH to comparing the acidity of Coca-Cola and vinegar. Vinegar obviously comes in different acidities though. Which one do you need? How do they help? Below is a quick and informative guide to vinegar acidities and their usages.
Can nylon be used as a weed killer?
Also made by centrifugation it has almost no other use for the general public except as weed killer. If using this as weed killer, ABSOLUTELY use hand and eye protection (chemistry goggles, not loose plastic safety glasses). No substitutes for latex or nitrile. Nylon can be corroded.
What is the mass of acetic acid?
Mass of solute (acetic acid) is given as 5.0 g and the volume of solution is 100.0 mL.
What is the density of a 2.00 mL sample of a substance?
a 2.00 mL sample of a substance A has a density of 18.4g/mL and a 5.00 mL sample of substance B has a density of 35.5g/mL. do you have an equal mass o …
How to convert ml to L?
So, to convert mL to L we divide by 1000.
