How to calculate Beer's law?
Steps to calculate absorption from Beer-Lambert Law Determine the molar absorption coefficient of the solution. Find the concentration of the solution. Calculate the length of the path in which the light beam travels. Finally, calculate absorption using the formula mentioned above. Beer Lambert's Law finds diverse applications in various studies.
How to make a beer's law plot?
Beer’s Law Type in [K2CrO4] in the A1 cell (x-axis) and Absorbance in the B1 cell (y-axis). Enter the data for concentration and absorbance. Highlight the concentration and absorbance columns and click on Insert on the top menu and click on Chart on the drop down menu. Click on the xy scatter option and click Finish.
What is Beer's law equation?
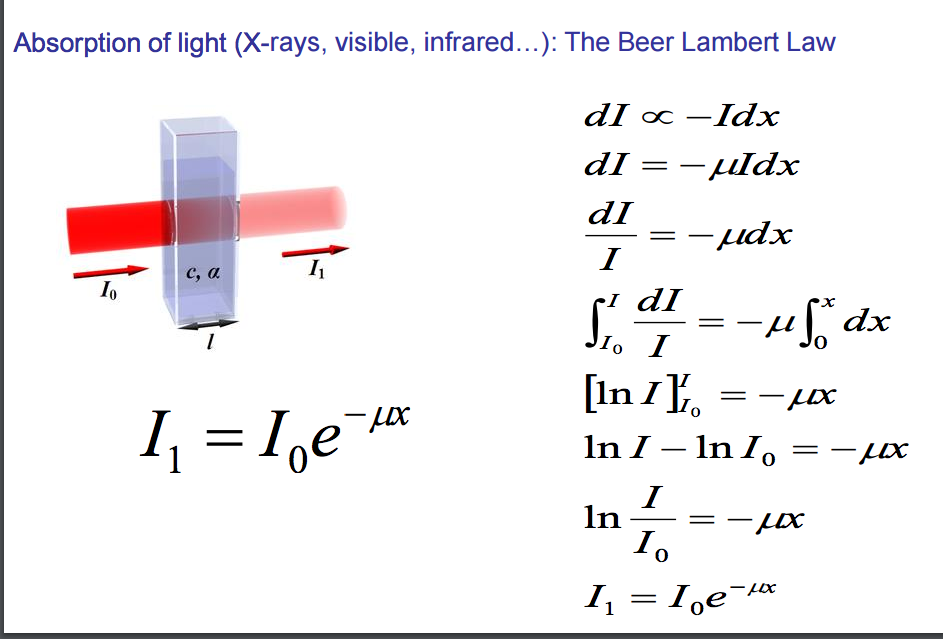
Beer-Lambert law Equation: I=Ioe- μ (x) Beer-Lambert's law can also be expressed as A= ε Lc, where A refers to the absorbance, ε denotes molar extinction coefficient, L denotes path length, and c denotes concentration. The law finds application in analytical chemistry and in the workings of the atmosphere.
What is the equation for the Beer Lambert law?
Beer-Lambert law Equation: I=Ioe- μ (x) Beer-Lambert's law can also be expressed as A= ε Lc, where A refers to the absorbance, ε denotes molar extinction coefficient, L denotes path length, and c denotes concentration. The law finds application in analytical chemistry and in the workings of the atmosphere.

How do you calculate Beer's law?
The equation for Beer's law is a straight line with the general form of y = mx +b.Beer's Law:A = (εl)c.with the general form.y = (m)x.
What is the equation for Beer's law and explain its meaning?
The relationship can be expressed as A = εlc where A is absorbance, ε is the molar extinction coefficient (which depends on the nature of the chemical and the wavelength of the light used), l is the length of the path light must travel in the solution in centimetres, and c is the concentration of a given solution.
What is Beer's Law simple?
Beer's Law (Beer-Lambert Law): The amount of energy absorbed or transmitted by a solution is proportional to the solution's molar absorptivity and the concentration of solute. In simple terms, a more concentrated solution absorbs more light than a more dilute solution does.
What is Beer's Law units?
Beer-Lambert Law It has units of M -1 cm -1 (M = molarity). The variation of ε with wavelength is characteristic of the substance. If you know the extinction coefficient of a species, you can measure the absorbance and the Beer-Lambert Law to calculate its concentration.
What is the formula of absorbance?
Absorbance (A) is the flip-side of transmittance and states how much of the light the sample absorbed. It is also referred to as “optical density.” Absorbance is calculated as a logarithmic function of T: A = log10 (1/T) = log10 (Io/I).
What is Beer-Lambert law statement?
The Beer-Lambert law states that there is a linear relationship between the concentration and the absorbance of the solution, which enables the concentration of a solution to be calculated by measuring its absorbance.
What is Beer-Lambert law BYJU's?
The Beer-Lambert law states that: for a given material sample path length and concentration of the sample are directly proportional to the absorbance of the light.
How do you calculate concentration in Beer-Lambert law?
Calculation of concentration (C = A/(L x Ɛ)) The Lambert-Beer law, which forms the physical basis for photometric applications, describes that the absorption of light by a sample is directly proportional to its concentration and its path length.
What is slope in Beer's law?
An example of a Beer's Law plot (concentration versus absorbance) is shown below. The slope of the graph (absorbance over concentration) equals the molar absorptivity coefficient, ε x l.
How do you calculate concentration from absorbance?
The equation should be in y=mx + b form. So if you substract your y-intercept from the absorbance and divide by the slope, you are finding the concentration of your sample.
Why absorbance has no unit?
Absorbance doesn’t have any unit because it is the ratio of the amount of light that passes through a solution compared to the amount of light that...
What are the limitations of Beer-Lambert law?
Following are the limitations of Beer-Lambert law: A diluted solution is used There shouldn’t be a scattering of the light beam Monochromatic elect...
Why does Beer-Lambert law fails at higher concentrations?
Beer-Lambert law fails at higher concentrations because the linearity of the law is limited to chemical and instrumental factors. When the solution...
What is Beer-Lambert’s law for absorption spectroscopy?
Beer-Lambert’s law for absorption spectroscopy is a linear relationship between the absorbance and the concentration of an absorbing species. The s...
State the situations when Beer’s law is not obeyed.
Following are the situations when Beer’s law is not obeyed: When different types of molecules are in equilibrium with each other. An association co...
What is the deviation of Beer Lamberts law?
The law also deviates if non-monochromatic light is used. The change in temperature also leads to the deviation of Beer-lamberts’ law. The deviation may also occur if the width of the instrument is not proper.
What is the absorption behavior of beer law?
Beer law and lambert law are only able to describe the absorption behavior of the solutions that contain relatively low amounts of solutes dissolved in it i.e. <10mm. the analyte starts behaving differently when the concentration of the analyte in the solution is high i.e. >10mm. this is due to the interaction of the analyte with the solvent and other solute molecules and sometimes even due to hydrogen bonding interactions.
Why is Beer Lambert law considered a limiting law?
Presently, the Beer lambert law is declared as a limiting law because the absorbance is only nearly linear depending on the concentration. This is the reason that the attenuation coefficient also depends on concentration and density even if there are no interactions.
What law describes the transmittance and absorbance of light?
The transmittance and absorbance of light by a substance was first introduced followed by an explanation of the Beer-Lambert Law.
Why is the Beer-Lambert law called the Beer-Lambert law?
The reason for so many names is because more than one law is involved in it. In 1729 Pierre Bouger discovered the law and published it in Essai d’optique sur la gradation de la lumiere. In 1760 Lambert quoted the Bouger’s discovery in his Photometria which states that the absorbance of a sample is directly proportional to the path length of light. Lambert did not claim any discovery, but he was often credited with it. In 1852, August Beer discovered that absorbance is proportional to the sample concentration. Generally, beers law relates only to concentration while Beer-Lambert law relates absorbance to both concentration and thickness of a sample.
What did Lambert discover about the law of absorbance?
Lambert did not claim any discovery, but he was often credited with it. In 1852, August Beer discovered that absorbance is proportional to the sample concentration. Generally, beers law relates only to concentration while Beer-Lambert law relates absorbance to both concentration and thickness of a sample.
What is the law of attenuation of solar radiation?
The attenuation of solar or stellar radiation is also described with the help of this law as it travels through the atmosphere. In this case, there is a scattering of radiation as well as absorption. The beer-lambert law for the atmosphere is written as:
What is the meaning of Beer Lambert law?
(Show more) Full Article. Beer’s law, also called Lambert-Beer law or Beer-Lambert law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. Formulated by German mathematician and chemist August Beer in 1852, it states that the absorptive capacity ...
Who created the absorptive capacity of a dissolved substance?
Formulated by German mathematician and chemist August Beer in 1852, it states that the absorptive capacity of a dissolved substance is directly proportional to its concentration in a solution.
What is Beer Lambert's law?
Beer-Lambert’s law for absorption spectroscopy is a linear relationship between the absorbance and the concentration of an absorbing species. The states imply that type, as well as the concentration of the molecules, are necessary.
Why does Beer Lambert law fail?
Beer-Lambert law fails at higher concentrations because the linearity of the law is limited to chemical and instrumental factors. When the solution has higher concentrations, the proximity between the molecules of the solution is so close that there are deviations in the absorptivity.
How much of the light is absorbed?
Therefore, we can say that 90% of the light is absorbed and 10% of light is transmitted.
Who said concentration and absorbance are directly proportional to each other?
Beer’s law was stated by August Beer which states that concentration and absorbance are directly proportional to each other.
Is the path length of a sample directly proportional to the absorbance of the light?
for a given material sample path length and concentration of the sample are directly proportional to the absorbance of the light.
What is the equation for Beer's law?
Here is an example of directly using the Beer’s Law Equation (Absorbance = e L c) when you were given the molar absorptivity constant (or molar extinction coefficient). In this equation, e is the molar extinction coefficient. L is the path length of the cell holder. c is the concentration of the solution.
What is the law of beer?
Beer’s Law (Beer-Lambert Law): The amount of energy absorbed or transmitted by a solution is proportional to the solution’s molar absorptivity and the concentration of solute. In simple terms, a more concentrated solution absorbs more light than a more dilute solution does.
How to find the concentration of copper sulfate pent?
Determine the concentration by mass percent by dividing the formula weight of CuSO4 by the formula weight of CuSO4-5H2O and multiplying by 100 percent: 159.62 / 249.72 * 100 = 63.92 percent. This means that a 100-gram sample of copper sulfate pentahydrate will contain 63.92 grams of copper sulfate.
What is the slope of a Beer's law graph?
An example of a Beer’s Law plot (concentration versus absorbance) is shown below. The slope of the graph (absorbance over concentration ) equals the molar absorptivity coefficient, ε x l.
How to find the concentration of a solution?
Write out the equation C = m/V, where m is the mass of the solute and V is the total volume of the solution. Plug in the values you found for the mass and volume, and divide them to find the concentration of your solution.
What is the equation for concentration?
The equation should be in y=mx + b form. So if you substract your y-intercept from the absorbance and divide by the slope, you are finding the concentration of your sample.
How to determine the concentration of an unknown solution?
The concentration of an unknown solution can then be determined graphically by measuring its absorbance and comparing it to a standard plot (or using the experimentally determined ε value.) provided.) Calculate the dilutions necessary to make your concentration using volumetric glassware.
How to measure Beer Lambert law?
It is possible to measure the Beer Lambert law by calculating the concentration of a solution by making use of the absorbancies. Another way is to plot a graph of various concentrations and then align them according to their appropriate or correct absorbencies. Afterwards, one must use a colourimeter to calculate the concentration ...
What is the application of the Beer Lamberts law?
Answer 1: The application of the Beer-Lamberts law takes place to the analysis of a mixture by spectrophotometry. Furthermore, this application is without the need for extensive sample pre-processing. The Beer-Lambert law example includes the determination of bilirubin in blood plasma samples.
Why is Beer Lambert law inaccurate?
This is because the analyte’s molecules exhibit stronger electrostatics and intermolecular interactions.
How does Beer Lambert law derivation take place?
Beer-Lambert law derivation can take place from an approximation for the absorption coefficient for a molecule. This happens by carrying out an approximation of the molecule by an opaque disk whose cross-sectional area is representative of the effective area seen by a frequency w photon.
Who developed the law of concentration?
The development of the law first took place by Pierre Bouguer before 1729. After its attribution to Johann Heinrich Lambert, the law included path length as a variable that had an effect on absorbance. ...
What law states that light absorption is directly proportional to the concentration of a substance?
Beer Lambert law tells us that the absorption of a quantity of light by a substance that is dissolved in a fully transmitting solvent happens to be directly proportional to the substance’s concentration and the path length of the light via the solution.
Why is the Beer Lambert law important?
Now let us look at the Beer-Lambert law and explore it's significance. This is important because people who use the law often don't understand it - even though the equation representing the law is so straightforward:
Is the law obeyed at high concentrations?
Note that the Law is not obeyed at high concentrations. This deviation from the Law is not dealt with here.
What is the Beer Lambert Law?
The Beer-Lambert Law describes the relationship between the absorbance and intensity of incident light. It helps to understand the attenuation of light when it travels through a medium of varying concentrations and length of travel of light beam. This page explains what Beer-Lambert Law is and how to calculate absorbance, path length, concentration, and absorptivity coefficient.
Which law relates the concentration of the solution and the length of the light path with the absorbance?
Thus, A=ϵcl is the Beer-Lambert Law that relates the concentration of the solution and the length of the light path with the absorbance.
What does it mean when the absorbance of a solution is zero?
Absorbance can range anywhere between zero to infinity. The absorbance of zero means the solution or the medium did not absorb any light. Similarly, the absorbance of 2 means the solution absorbed 99 percent of the beam. Ideally, absorbance values below 1 are considered normal. Any absorbance above 1.0 indicates that the solution is highly concentrated.
Does the concentration of the solution cross 0.01M?
The concentration of the solution does not cross 0.01M.
Does Beer Lambert's law help with radiation?
Do you know that this law also helps in the calculation of earth’s radiation? Yes, the attenuation of radiation received by Earth’s atmosphere is calculated by Beer Lambert’s Law.
